La denervación renal percutánea debe considerarse en el tratamiento de la hipertensión arterial
AGONIST
“Controversy is the fuel of progress; only by facing opposing ideas can we move forward”.
John Stuart Mill
The present controversy refers to the clinical implications of the percutaneous renal denervation (PRD) strategy in hypertensive patients. Although beforehand my role as PRD “agonist” could be termed “the dark side of the moon” (in relation to the antagonist), I will try to postulate in the following paragraphs the potential benefits and the clinical scenarios where we should not neglect considering this treatment to optimize the care of our patients.
INTRODUCTION
Hypertension (HTN) is still one of the main cardiovascular risk factors worldwide, with a prevalence of 34% according to data of the most recent review of the International Society of Hypertension, (1) and it is associated with a clear deleterious clinical effect. (2)
Even though the prevalence of HTN continues to be constant, the absolute number of people diagnosed with this disease has markedly increased, from 648 million in 1990 to 1.28 billion in 2019, representing a constant challenge for public health. (3)
Currently, the achievement of blood pressure (BP) targets in patients with HTN is still low in most regions of the world. (4) One of the main reasons is the low adherence to pharmacological treatment, identified as one of the main causes of HTN management failure. (5,6) Thus, we should consider “alternative” therapeutic strategies for HTN treatment (though from now on we could term them “additional”), among which PRD is postulated as a promising tool.
PHYSIOLOGICAL BASIS OF PRD
When considering a therapeutic strategy, it is essential to understand its mechanism of action in order to obtain the desired benefit. In the context of the complex pathophysiology of HTN, the renal sympathetic nervous system, composed of both afferent and efferent sympathetic nervous fibers, plays a crucial role in the development of this entity. (7) The activation of the sympathetic efferent nerves in the renal arteries may cause vasoconstriction of the kidney arterioles, reduced renal blood flow, increased renin secretion, and finally activation of the renin-angiotensin-aldosterone system, which leads to increased sodium and water absorption, and as a result, increased intravascular volume and the perpetuation of systemic HTN. (8) On the other hand, the activation of the sympathetic afferent nerves from the renal arteries can produce an increase in systemic sympathetic activity and the release of hypothalamic hormones, leading to increased systemic vascular resistance and, consequently, to the maintenance of HTN. (9) This is the pathophysiological basis of PRD therapy, which interrupts through the implementation of various mechanisms (radiofrequency [RF], ultrasound [US], neurolysis) the pathways of both afferent and efferent sympathetic nerves in the renal artery, thus achieving an “additional” (and not “alternative”) therapeutic approach to the treatment of HTN.
SCIENTIFIC EVIDENCE
Symplicity HTN-3 Study
In our setting, PRD is still a rarely prescribed procedure. It could be assumed that this position is based on the results of the Symplicity HTN-3 study. (10) This was the first sham-controlled, randomized study to demonstrate a high safety profile of the PRD procedure using a device based on an RF-emitting monoelectrode (Symplicity renal-denervation catheter, Medtronic), but that did not succeed in demonstrating superiority in terms of 24-hour outpatient systolic BP (SBP) and office BP reduction, compared with the sham subgroup. It is worth highlighting how striking these negative findings are, considering that early studies in the first half of the twentieth century had demonstrated the effectiveness of surgical sympathectomy as a tool for treating severe HTN. (11)
Further analyses have identified multiple causes of negative results in the Symplicity HTN-3 study, including changes in pharmacological adherence during the course of the study, poor experience of operators with the PRD device, high variability in the results of the procedures and the inability of the first-generation device to perform simultaneous ablations, among others. (12) Identifying these points was essential to improve further studies.
SPYRAL HTN-ON MED EXPANSION AND SPYRAL HTN-OFF MED PIVOTAL STUDIES
These were the first international, randomized, sham-controlled studies that included patients with HTN evaluating the benefit of PRD in terms of reducing 24-hour SBP both in the presence of antihypertensive pharmacological treatment (SPYRAL HTN-ON MED Expansion), (13) and in its absence (SPYRAL HTN-OFF MED Pivotal). (14) With a 3-month follow-up, the SPYRAL HTN-OFF MED Pivotal study succeeded in demonstrating a statistically significant decrease in 24-hour SBP (Δ -4.0 [-6.2 to -1.8] mmHg), and office SBP (Δ-6.6 [-7.9 to -1.9) mm Hg in relation to the sham subgroup; (14) whereas, conversely, SPYRAL HTN-ON MED Expansion did not identify a statistically significant difference between the two subgroups in terms of 24 hour SBP, which could be explained by an exaggerated reduction in pressure values within the sham subgroup secondary to an unbalanced prescription of pharmacological treatment and also by the impact of the SARS-CoV-2 pandemic. (13) It is also highlighted that the PRD subgroup showed a significant reduction in office SBP and diastolic BP, and a further reduction of blood pressure levels during nighttime, which represents an additional therapeutic benefit. (13) Both studies demonstrated a high safety profile, with an occurrence of adverse periprocedural events close to 1-2% and a rate of major adverse events of 0.4% at one year of the index procedure. (15)
RADIANCE-HTN STUDY
The RADIANCE-HTN study was a multicenter, randomized, sham-controlled study that identified a benefit of the US Paradise-based DRP system (ReCor Medical, Palo Alto, CA, USA) in terms of reducing BP in patients with HTN, in relation to the sham procedure, both in the subgroup with no concomitant antihypertensive treatment (RADIANCE-HTN SOLO) (16) and in those with resistant HTN treated with a fixed dose of a triple pharmacological combination. (RADIANCE-HTN TRIO). (17) It is important to note that the benefit of this therapy in terms of BP reduction remained constant for a period of 36 months since the index procedure (Table 1). (18)
Table 1
Randomized, sham-controlled clinical studies and real-world evidence supporting percutaneous renal denervation
| SPYRAL HTN-ON MED (13) | SPYRAL HTN-OFF MED (14) | RADIANCE HTN-SOLO (16) | RADIANCE HTN-TRIO (17) | RADIANCE II (PIVOTAL) (26) | Global SYMPLICITY Registry (27) | NETROD HTN Study (28) | Iberis-HTN Trial (29) | SMART Trial (30) | |
|---|---|---|---|---|---|---|---|---|---|
| Design | Multicenter, randomized, sham-controlled study. | Multicenter, randomized, sham-controlled study. | Multicenter, randomized, sham-controlled study. | Multicenter, randomized, sham-controlled study. | Multicenter, randomized, sham-controlled study. | Observational, prospective, open-label, single-arm registry. | Multicenter, randomized, sham-controlled study. | Multicenter, randomized, sham-controlled study. | Multicenter, randomized, sham-controlled study. |
| Sample size | 337 patients. | 331 patients. | 146 patients. | 136 patients. | 224 patients. | >3000 patients. | 205 patients. | 217 patients. | 220 patients. |
| Objective | Assess the efficacy of PRD in patients under antihypertensive treatment. | Assess the efficacy of PRD in patients without antihypertensive treatment. | Assess the efficacy of US PRD in patients without antihypertensive treatment. | Assess the efficacy of US PRD in patients with antihypertensive treatment. | Assess the efficacy of US PRD in patients with stage 2 HTN under treatment with 0-2 antihypertensive drugs. | Assess the safety and efficacy of PRD in “real-world” patients. | Assess the safety and efficacy of PRD in hypertensive patients under treatment with two antihypertensive drugs. | Assess the safety and efficacy of PRD in hypertensive patients under treatment with triple pharmacological therapy. | Assess the safety and efficacy of a mapping system and selective PRD in hypertensive patients under dual pharmacological therapy treatment. |
| Primary endpoints | Change in average 24-hour ambulatory SBP with respect to baseline, at 6-month follow-up. | Change in average 24-hour ambulatory SBP with respect to baseline, at 3-month follow-up. | Change in average daytime SBP with respect to baseline, at 2-month follow-up. | Change in average daytime SBP with respect to baseline, at 2-month follow-up. | Change in average daytime SBP with respect to baseline, at 2-month follow-up. | N/A | Change in average office SBP with respect to baseline, at 6-month follow-up. | Change in average 24-hour ambulatory SBP with respect to baseline, at 6-month follow-up. | Asses the proportion of patients with office SBP ≤140/90 mmHg (non-inferiority). Difference in antihypertensive medication load |
| Results | Change in 24-hour ambulatory SBP: PRD -6.5 ±10.7 mmHg vs. sham -4.5±10.3 mmHg (p=0.12). | Change in 24-hour ambulatory SBP: PRD -4.7 mmHg vs. sham -0.6 mmHg (Δ -4.0 mmHg; p<0.001). | Change in daytime SBP: PRD -8.5±9.3 mmHg vs. sham -2.2±10.0 mmHg (p< 0.001). | Change in daytime SBP: PRD -8.0 (-16.4 to 0) mmHg vs. sham -3.0 (-10.3 to - 1.8) (p=0.022). | Change in daytime SBP: PRD -7.9±11.6 mmHg vs. sham -1.8±9.5 mmHg (Δ -6.3 mmHg; p<0.001). | Change in office SBP: PRD -12.8±26.2 mmHg vs. baseline (p<0.001); 24-hour ambulatory SBP: PRD -7.2±17.8 mmHg vs. baseline (p<0.001). | Change in office SBP: PRD -25.2±13.9 mmHg vs. sham -6.2±12.5 mmHg (p<0.001). | Change in 24-hour ambulatory SBP: PRD-13.0±12.1 mmHg vs. sham -3.0±13.0 mmHg (Δ -9.4 mmHg; p<0.001). | Office SBP control percentage: PRD 95% vs. sham 93% (p=0.429, non-inferiority p <0.001). Less antihypertensive drugs (or lower dose) in the PRD subgroup vs. sham (Δ -3.3, superiority p =0.003). |
| Conclusions | Negative study, without significant difference in the primary endpoint. | Positive study, showing significant RF PRD benefit relative to sham. | Positive study, showing significant US PRD benefit relative to sham. | Positive study, showing significant US PRD benefit relative to sham. | Positive study, showing significant US PRD benefit relative to sham. | Positive study, showing significant RF PRD benefit sustained throughout time with high safety profile. | Positive study, showing significant RF PRD benefit relative to sham. | Positive study, showing significant RF PRD benefit relative to sham. | Positive study, showing feasibility, efficacy and safety of selective PRD. |
PATIENT SELECTION AND RECOMMENDATION OF CLINICAL PRACTICE GUIDELINES
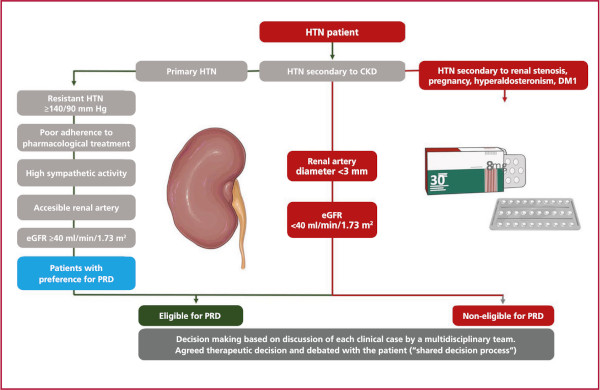
There are currently underrepresented subpopulations of HTN patients in which this therapeutic approach strategy is not recommended, namely: (19)
Renal transplant recipients.
Patients with severe renal impairment (glomerular filtration rate ≤40 ml/min/1.73 m2).
Patients requiring hemodialysis therapy.
Patients diagnosed with fibromuscular dysplasia.
Patients with untreated secondary HTN.
Patients with a single functional kidney (functionally monorenal).
Conversely, clinical practice guidelines recommend considering PRD in patients with “resistant HTN” (defined as office BP ≥140/90 mmHg, confirmed by 24-hour outpatient pressure monitoring with SBP ≥130 mmHg or daytime SBP ≥135 mm Hg, treated with ≥3 antihypertensive drugs, including a diuretic at the highest tolerated dose) and with an estimated glomerular filtration rate of ≥40 ml/min/1.73m2 (class of recommendation [COR] II, level of evidence [LOE] B). It is also recommended in patients with “uncontrolled HTN” (under treatment with ≥4 antihypertensive drugs) and in those who have severe adverse events or quality of life impairment secondary to pharmacological treatment. It is also worth pointing out that this procedure should be carried out in specialized medical centers (COR I, LOE C), with a multidisciplinary team for decision-making and that patient preference should be considered (COR I, LOE C). (19,20) Patients not adhering to or intolerant to multiple pharmacological treatments should also be considered as candidates for PRD (Figure 1).
CONCLUDING CONSIDERATIONS
The benefit of PRD systems in patients with HTN is not negligible and has proven to be a safe and lasting strategy. In this regard, by analyzing the evidence as a whole, identifying an approximate decrease of 10 mmHg in office SBP and 6 mm Hg in 24-hour outpatient SBP monitoring could result in 20% reduction in major cardiovascular events, 28% of heart failure events, 17% of atherosclerotic coronary disease and 13% decrease of mortality. (21)
In our setting, and considering the invasive nature of the PRD strategy, the costs required by the health system and the above-mentioned “modest” benefit observed, it is clear that this strategy should not be used systematically. Despite the “invasiveness” of the method, it has been demonstrated that it is a safe procedure and does not exceed the risk of adverse events inherent in the natural evolution of the study population, while in terms of cost-effectiveness it appears to be a cost-effective strategy in selected patients. (22)
Given that the clinical benefit of PRD is equivalent to the reduction of only one antihypertensive drug, (23) it is worth noting that current evidence on PRD suggests that this therapy produces a constant reduction in daytime and nighttime BP, which is known as the ‘always-on’ effect.(24) This contrasts with the pharmacokinetic profiles and dosage regimes of antihypertensive drugs, and also overcomes the challenges of pharmacological adherence, so their pharmacologic equivalence should not be considered a valid argument for its non-implementation.
The decision shared with the patient should not be underestimated. In both patients with intolerance to antihypertensive drugs and in those with a history of poor pharmacological adherence, PRD therapy is postulated as a valuable therapeutic option. Although this therapeutic alternative is commonly considered in our setting for patients with “refractory HTN”, (25) evidence has shown that its prescription at earlier stages of the disease implies substantial clinical benefit.
CONCLUSIONS
Percutaneous renal denervation therapy is a safe and effective therapeutic strategy for selected hypertensive patients, which achieves an additional benefit to pharmacological medical treatment and reduces the number of associated adverse events, so we should consider it in our therapeutic arsenal based on the clinical judgement of a multidisciplinary team, and offer patients this option to participate in decision-making.
Conflicts of interest Conflicts of interest
None declared. (See conflicts of interest forms on the website).
REFERENCES
1. Beaney T, Schutte AE, Stergiou GS, Borghi C, Burger D, Charchar F, et al. May Measurement Month 2019: The Global Blood Pressure Screening Campaign of the International Society of Hypertension. Hypertens 2020;76:333-41. https://doi.org10.1161/HYPERTENSIONAHA.120.14874
2. Abbafati C, Abbas KM, Abbasi-Kangevari M, Abd-Allah F, Abdelalim A, Abdollahi M, et al. Global burden of 87 risk factors in 204 countries and territories, 1990-2019: a systematic analysis for the Global Burden of Disease Study 2019. Lancet 2020;396:1223-49. https://doi.org10.1016/S0140-6736(20)30752-2
3. Zhou B, Carrillo-Larco RM, Danaei G, Riley LM, Paciorek CJ, Stevens GA, et al. Worldwide trends in hypertension prevalence and progress in treatment and control from 1990 to 2019: a pooled analysis of 1201 population-representative studies with 104 million participants. Lancet 2021;398:957-80. https://doi.org10.1016/S0140-6736(21)01330-1
4. Sakulsupsiri A, Chattranukulchai P, Siwamogsatham S, Boonchayaanant P, Naeowong W, Ariyachaipanich A, et al. Home Blood Pressure Control and Drug Prescription Patterns among Thai Hypertensives: A 1-Year Analysis of Telehealth Assisted Instrument in Home Blood Pressure Monitoring Nationwide Pilot Project. Int J Hypertens 2021:2021:8844727. https://doi.org10.1155/2021/8844727
5. Burnier M, Egan BM. Adherence in Hypertension. Circ Res 2019;124:1124-40. https://doi.org10.1161/CIRCRESAHA.118.313220
6. Lawson AJ, Hameed MA, Brown R, Cappuccio FP, George S, Hinton T, et al. Nonadherence to antihypertensive medications is related to pill burden in apparent treatment-resistant hypertensive individuals. J Hypertens 2020;38:1165-73. https://doi.org10.1097/HJH.0000000000002398
7. Grassi G, Mark A, Esler M. The sympathetic nervous system alterations in human hypertension. Circ Res 2015;116:976-90. https://doi.org10.1161/CIRCRESAHA.116.303604
8. DiBona GF. Neural control of the kidney: functionally specific renal sympathetic nerve fibers. Am J Physiol Regul Integr Comp Physiol 2000;279. https://doi.org10.1152/ajpregu.2000.279.5.R1517
9. Ciriello J, De Oliveira CVR. Renal afferents and hypertension. Curr Hypertens Rep 2002;4:136-42. https://doi.org10.1007/s11906-002-0038-x
10. Bhatt DL, Kandzari DE, O'Neill WW, D'Agostino R, Flack JM, Katzen BT, et al. A Controlled Trial of Renal Denervation for Resistant Hypertension. N Engl J Med 2014;370:1393-401. https://doi.org10.1056/NEJMoa1402670
11. Smithwick RH, Thompson JE. Splanchnicectomy for essential hypertension; results in 1,266 cases. J Am Med Assoc 1953;152:1501-4. https://doi.org10.1001/jama.1953.03690160001001
12. Epstein M, De Marchena E. Is the failure of SYMPLICITY HTN-3 trial to meet its efficacy endpoint the ï¿1/2end of the roadï¿1/2 for renal denervation? J Am Soc Hypertens 2015;9:140-9. https://doi.org10.1016/j.jash.2014.12.002
13. Kandzari DE, Townsend RR, Kario K, Mahfoud F, Weber MA, Schmieder RE, et al. Safety and Efficacy of Renal Denervation in Patients Taking Antihypertensive Medications. J Am Coll Cardiol 2023;82:1809-23. https://doi.org10.1016/j.jacc.2023.08.045
14. Böhm M, Kario K, Kandzari DE, Mahfoud F, Weber MA, Schmieder RE, et al. Efficacy of catheter-based renal denervation in the absence of antihypertensive medications (SPYRAL HTN-OFF MED Pivotal): a multicentre, randomised, sham-controlled trial. Lancet 2020;395:1444-51. https://doi.org10.1016/S0140-6736(20)30554-7
15. Townsend RR, Walton A, Hettrick DA, Hickey GL, Weil J, Sharp ASP, et al. Review and meta-analysis of renal artery damage following percutaneous renal denervation with radiofrequency renal artery ablation. EuroIntervention 2020;16:89-96. https://doi.org10.4244/EIJ-D-19-00902
16. Azizi M, Schmieder RE, Mahfoud F, Weber MA, Daemen J, Davies J, et al. Endovascular ultrasound renal denervation to treat hypertension (RADIANCE-HTN SOLO): a multicentre, international, single-blind, randomised, sham-controlled trial. Lancet 2018;391:2335-45. https://doi.org10.1016/S0140-6736(18)31082-1
17. Azizi M, Sanghvi K, Saxena M, Gosse P, Reilly JP, Levy T, et al. Ultrasound renal denervation for hypertension resistant to a triple medication pill (RADIANCE-HTN TRIO): a randomised, multicentre, single-blind, sham-controlled trial. Lancet 2021;397:2476-86. https://doi.org10.1016/S0140-6736(21)00788-1
18. Rader F, Kirtane AJ, Wang Y, Daemen J, Lurz P, Sayer J, et al. Durability of blood pressure reduction after ultrasound renal denervation: three-year follow-up of the treatment arm of the randomised RADIANCE-HTN SOLO trial. EuroIntervention 2022;18:E677-85. https://doi.org10.4244/EIJ-D-22-00305
19. Barbato E, Azizi M, Schmieder RE, Lauder L, Bï¿1/2hm M, Brouwers S, et al. Renal denervation in the management of hypertension in adults. A clinical consensus statement of the ESC Council on Hypertension and the European Association of Percutaneous Cardiovascular Interventions (EAPCI). EuroIntervention 2023;18:1227-43.
20. Mancia G, Kreutz R, Brunström M, Burnier M, Grassi G, Januszewicz A, et al. 2023 ESH Guidelines for the management of arterial hypertension The Task Force for the management of arterial hypertension of the European Society of Hypertension: Endorsed by the International Society of Hypertension (ISH) and the European Renal Associat. J Hypertens 2023;41:1874-2071. https://doi.org10.1097/HJH.0000000000003480
21. Ettehad D, Emdin CA, Kiran A, Anderson SG, Callender T, Emberson J, et al. Blood pressure lowering for prevention of cardiovascular disease and death: a systematic review and meta-analysis. Lancet. 2016;387:957-67. https://doi.org10.1016/S0140-6736(15)01225-8
22. Kandzari DE, Cao KN, Lobo MD, Sharp ASP, Schmieder RE, Esler MD, et al. C-13 | Cost-Effectiveness of Renal Denervation for Uncontrolled Hypertension in the Presence of Antihypertensive Drugs: An Analysis for the U.S. Based on 6-Month Primary Results from the SPYRAL HTN-ON MED Randomized Trial. J Soc Cardiovasc Angiogr Interv 2023;2:100799. https://doi.org10.1016/j.jscai.2023.100799
23. Kjeldsen SE, Esler MD. Take a blood pressure pill or undergo renal denervation? Lancet 2018;391:2298-300. https://doi.org10.1016/S0140-6736(18)31126-7
24. Azizi M. Catheter-based renal denervation for treatment of hypertension. Lancet 2017;390:2124-6. https://doi.org10.1016/S0140-6736(17)32293-625.
26. Azizi M, Saxena M, Wang Y, Jenkins JS, Devireddy C, Rader F, et al. Endovascular Ultrasound Renal Denervation to Treat Hypertension: The RADIANCE II Randomized Clinical Trial. JAMA 2023;329:651-61. https://doi.org10.1001/jama.2023.0713
27. Mahfoud F, Böhm M, Schmieder R, Narkiewicz K, Ewen S, Ruilope L, et al. Effects of renal denervation on kidney function and long-term outcomes: 3-year follow-up from the Global SYMPLICITY Registry. Eur Heart J 2019;40:3474-82. https://doi.org10.1093/eurheartj/ehz118
28. Li Y, Nawabi AQ, Feng Y, Dai Q, Ma G, Liu N, et al. Safety and Efficacy of a New Renal Denervation Catheter in Hypertensive Patients in the Absent of Antihypertensive Medications: A Pilot Study. Int J Hypertens 2019;7929706. https://doi.org10.1155/2019/7929706
29. Kario K, Rumoroso JR, Okawara Y, Pérez de Prado A, García Fernández E, Kagitani H, et al. Renal Sympathetic Denervation in Patients with Resistant Hypertension: A Feasibility Study. Pulse (Basel) 2019;6: 137-143. https://doi.org10.1159/000490620
30. Wang J, Sun N, Ge J, Jiang H, Yin Y, Chen M, et al. Rationale and Design of Sympathetic Mapping/Ablation of Renal Nerves Trial (SMART) for the Treatment of Hypertension: a Prospective, Multicenter, Single-Blind, Randomized and Sham Procedure-Controlled Study. J Cardiovasc Transl Res 2023;16:358-70. https://doi.org10.1007/s12265-022-10307-z
ANTAGONIST
Percutaneous renal denervation (PRD) has reemerged as a promising option for the treatment of resistant or refractory HTN. (1,2,3) However, beyond the efficacy that can be attributed in terms of BP reduction, better structured scenarios would be needed for its optimal indication.
In the current scenario, two main factors contribute to a still insufficient recommendation: low patient adherence to non-invasive antihypertensive treatment, and medical inertia or limited availability of resources. (4),,5,6,7)
A provocative analogy that exemplifies this view might be to compare the need to prescribe parenteral feeding to a person who could eat by himself if provided with adequate food, a plate and cutlery.
Some of the main challenges that we consider to optimize the scenario and define the prominence of PRD are:
a) Challenge 1: Pseudo-resistance
Let us consider a possible situation: on the one hand, a patient with resistant HTN, and on the other his family doctor, who does not have the resources to rule out the causes of pseudo- resistance, such as lack of adherence to prescribed medications, secondary causes of hypertension, hemodynamic mechanisms, and dysautonomia.
In this case, if PRD were an available treatment, would it be an appropriate option?
The answer should be negative; it would be preferable to improve access to non-invasive options with a better risk/benefit and cost/benefit equation with respect to PRD.
This is especially relevant if one takes into account that pseudo or apparent resistance may be present in 10 to 40% of patients considered resistant. (8,9)
b) Challenge 2: The paradox of dysautonomia treatment
A relevant and little explored aspect in the literature concerning renal denervation is simply the paradox of considering the invasive intervention of the autonomic nervous system, without having previously tried other non-invasive interventions. It is true that the treatment of dysautonomia may be less known and addressed in everyday medical practice compared with other more common conditions. (10) However, some of the causes of certain medical inertia in its diagnostic and therapeutic approach could be:
-
Low prevalence: although dysautonomia may be prevalent in certain populations (such as in people with diabetes, Parkinson's disease, or after a stroke), its frequency, compared with other more common diseases, can make it not a priority of continuous medical training and education, which gives rise to a significant low level of research, and limited centers where the diagnosis can be adequately addressed in situations such as HTN, since it is generally centralized in specialists in neurology or electrophysiology.
-
Complex diagnosis: accurate diagnosis of dysautonomia disorders can be challenging due to the variety of symptoms and the overlap with other medical conditions.
-
Lack of resources and specialization: assessment and management of dysautonomia may require specific resources, such as specialized autonomic function testing and collaboration with specialists in neurology or rehabilitation medicine.
-
Focus on specific symptoms: physicians may often focus on treating individual symptoms (such as hypertension or tachycardia) without recognizing dysautonomia as the underlying cause.
To strengthen this concept, the frequency of use of medications that affect the autonomic nervous system (for example, alpha 1 adrenergic antagonists, central alpha 2 adrenergic agonists) can be analyzed in our caseload of patients with resistant HTN. Given that PRD is an invasive treatment aimed at reducing sympathetic stimulation in HTN, it would have been logical to explore non-invasive interventions aimed at the same mechanism prior to its indication, to determine if a favorable result could be obtained with less intrusive resources when addressing this underlying mechanism in the patient's condition. (9)
c) Challenge 3: The absence of phenotyping
In the main studies supporting the efficacy of PRD, the patient recruited for participation is stabilized for a few months with drugs that do not include direct effects on the autonomic nervous system. (12,13) This situation does not contribute to providing information about their phenotyping before and after the procedure. Patients with different degrees of dysautonomia may present significant alterations in autonomic control, which may influence BP regulation and the response to therapeutic interventions such as renal denervation. The efficacy of the procedure is different and not uniform in its magnitude or in its persistence in the follow-up among the included patients, so future studies are required on the possibility of stratifying the response, as is done in other clinical studies.
Assessing sympathetic nervous system function before renal denervation is crucial for several reasons:
-
Appropriate patient selection: allows identifying those patients whose HTN may be significantly influenced by sympathetic activity. Renal denervation may be more beneficial in these cases, since its main mechanism of action is to reduce sympathetic activity at the renal level.
-
Prediction of treatment response: evaluation of the sympathetic nervous system can help predict the probability that a patient will respond favorably to renal denervation. Those with elevated sympathetic activity may be more likely to benefit from the procedure.
-
Optimization of therapeutic management: before opting for an invasive treatment such as renal denervation, it is important to ensure that non-invasive treatments targeting the sympathetic nervous system have been exhausted and optimized. This may include medication and lifestyle changes, as well as other therapeutic interventions.
-
Reducing therapeutic inertia: identifying elevated sympathetic activity can help avoid therapeutic inertia, that is, the delay in escalating treatment when BP goals are not achieved with conventional medications.
d) Challenge 4: Inconsistent research results
Overall, the arguments underline the need for a critical and evidence-based evaluation of renal denervation as a treatment for resistant HTN, considering both the potential benefits and the possible risks and limitations of the intervention. The trajectory of research and the results obtained have had contradictions, but the most recent data are encouraging and bring renal denervation back into scientific analysis. (14) However, some points remain to be resolved:
-
Limited evidence of long-term efficacy.
-
Methodology and design of the studies.
-
Variability in results.
-
Therapeutic alternatives.
-
Potential risks and safety.
A design that could provide answers in the future, and be welcomed in the medical community, would consist of randomly assigning patients with resistant HTN to compare PRD with traditional pharmacological treatment, together with drugs that directly modulate the autonomic nervous system. In addition, evaluation of PRD efficacy compared with an intensive healthy lifestyle strategy, and monitoring adherence in both groups, could be considered, among other possible options.
e) Challenge 5: Is the expectation greater than the result?
The average SBP reduction achieved in 24-hour ambulatory monitoring is approximately 6 mm Hg, as reported in new randomized clinical trials. This value, undoubtedly significant in terms of risk and event reduction, is equivalent to that of an antihypertensive medication.
A favorable point of view would assume that it is a curative treatment, but in fact it is not, since it is necessary to continue with pharmacological treatment after PRD, though at lower doses and perhaps fewer drugs. From a pessimistic point of view, it is still a potentially replaceable invasive intervention with better patient adherence.
Moreover, in pharmacological studies we demand evidence of event reduction, which have not yet been reported in renal denervation studies, that only have modeling of the possible decrease in events associated with a drop in BP. (15)
Conclusions
To conclude, the context on which decision-making is based regarding the efficacy and indication of PRD for the treatment of HTN is constantly evolving and shows clear potential. However, there are some relevant considerations that still need to be addressed for greater generalization of this intervention:
-
Data are required on the direct impact of PRD on the reduction of cardiovascular events, as required from pharmacological studies.
-
It is crucial to implement resources that allow evaluating the patient's adherence to sodium intake restriction by determining 24-hour natriuresis. Available evidence suggests that this measure can achieve a similar or even greater reduction in SBP compared with renal sympathetic denervation.
-
Strategies to evaluate the patient's response to non-invasive treatments that modulate the autonomic nervous system are necessary before considering invasive treatment. This premise should be applied both in daily clinical practice and in research studies.
-
Propose predictive scores for the response to PRD, allowing a more precise selection of candidate patients.
Finally, the perspective of PRD acceptance and the most recent data is still divergent. While in the United States no PRD device has yet been authorized by the FDA (Food and Drug Administration), in Europe the strategy is gaining ground and raises the possibility of treating in a similar way patients with non-resistant HTN, unlike the original indications of the technique limited to rescue cases for those with resistant HTN. Interestingly, a study conducted in Germany with 1000 patients with HTN revealed that 25% would prefer to undergo PRD rather than use a drug. (16) These considerations are crucial to advance in the understanding and effective application of PRD in the management of HTN, ensuring more personalized and evidence-based care for patients.
Conflicts of interest Conflicts of interest
None declared. (See conflicts of interest forms on the website).
REFERENCES
1. Pisano A, Iannone LF, Leo A, Russo E, Coppolino G, Bolignano D. Renal denervation for resistant hypertension. Cochrane Database Syst Rev 2021;11:CD011499. https://doi.org10.1002/14651858.CD011499.pub3
2. Azizi M, Sanghvi K, Saxena M, Gosse P, Reilly JP, Levy T, et al. Ultrasound renal denervation for hypertension resistant to a triple medication pill (RADIANCE-HTN TRIO): a randomised, multicentre, single-blind, sham-controlled trial. Lancet 2021;397:2476-86. https://doi.org10.1002/14651858.CD011499.pub3
3. Schmieder RE, Mahfoud F, Azizi M, Pathak A, Dimitriadis K, Kroon AA, et al; Members of the ESH Working Group on Interventional Treatment of Hypertension. J Hypertens 2018; 36:2042-8. https://doi.org10.1097/HJH.0000000000001858
4. Kearney PM, Whelton M, Reynolds K, Muntner P, Whelton PK, He J. Global burden of hypertension: analysis of world- wide data. Lancet 2005;365:217-23. https://doi.org10.1016/S0140-6736(05)17741-1
5. Carey RM, Calhoun DA, Bakris GL, Brook RD, Daugherty SL, Dennison-Himmelfarb CR, et al. Resistant hypertension: detec- tion, evaluation, and management: a scientific statement from the American Heart Association. Hypertension 2018;72:e53-90. https://doi.org10.1161/HYP.0000000000000084
6. Burnier M, Egan BM. Adherence in hypertension: a review of prevalence, risk factors, impact, and management. Circ Res 2019;124:1124-40. https://doi.org10.1161/CIRCRESAHA.118.313220
7. Pathan MK, Cohen DL. Resistant hypertension: where are we now and where do we go from here? Integr Blood Press Control. 2020;13:83-93. https://doi.org10.2147/IBPC.S223334
8. Flack JM, Buhnerkempe MG, Moore KT. Resistant Hypertension: Disease Burden and Emerging Treatment Options. Curr Hypertens Rep 2024;26:183-199. https://doi.org10.1007/s11906-023-01282-0
9. López-Jaramillo P, Barbosa E, Molina DI, Sanchez R, Diaz M, Camacho PA, et al; Latin American Society of Hypertension Consensus Expert Group. Latin American Consensus on the management of hypertension in the patient with diabetes and the metabolic syndrome. J Hypertens. 2019;37:1126-1147. https://doi.org10.1097/HJH.0000000000002072
11. Kotliar C, Obregón S, Koretzky M, Botto F, Di Leva A, Boscaro M, et al. Improved identification of secondary hypertension: use of a systematic protocol. Ann Transl Med 2018;6:293. https://doi.org10.21037/atm.2018.06.25
12. Esler M. Illusions of truths in the Symplicity HTN-3 trial: generic design strengths but neuroscience failings. J Am Soc Hypertens 2014;8:593-8. https://doi.org10.1016/j.jash.2014.06.001
13. Kandzari DE, Bhatt DL, Brar S, Devireddy CM, Esler M, Fahy M, et al. Predictors of blood pressure response in the SYMPLICITY HTN-3 trial. Eur Heart J 2015;36:219-27. https://doi.org10.1093/eurheartj/ehu441
14. Weber MA, Kirtane A, Mauri L, Townsend RR, Kandzari DE, Leon MB. Renal Denervation for the Treatment of Hypertension: Making a New Start, Getting It Right. Clin Cardiol 2015;38:447-54. https://doi.org10.1002/clc.22424
15. Fengler K, Reimann P, Rommel KP, Kresoja KP, Blazek S, Unterhuber M, et al Comparison of Long-Term Outcomes for Responders Versus Non-Responders Following Renal Denervation in Resistant Hypertension. J Am Heart Assoc. 2021;10:e022429. https://doi.org10.1161/JAHA.121.022429
16. Schmieder RE, Högerl K, Jung S, Bramlage P, Veelken R, Ott C. Patient preference for therapies in hypertension: a cross-sectional survey of German patients. Clin Res Cardiol 2019;108:1331-42. https://doi.org10.1007/s00392-019-01468-0
AGONIST REPLY
First of all, I congratulate Dr. Kotliar for the strength and clarity of her presentation. At the end of the reading, the concepts of "poor adherence", "medical inertia" and "dysautonomia" were prominent. I fully agree that poor adherence to pharmacological treatment is an area for improvement, but I disagree that the strategy of percutaneous renal denervation (PRD) should be considered only when others have failed, which would position it as an "alternative" rather than a "complementary" strategy. Not only does this remind me of the previously mentioned "medical inertia" (probably due to limited knowledge of the technique), but it also fails to consider the additional advantages of PRD, such as the 'always-on' phenomenon, and its potentially greater benefit when implemented at earlier stages in selected patients.
I agree that the pseudo-resistance mentioned by Dr Kotliar is a frequent phenomenon in clinical practice. However, it is important to note that there is scientific evidence to support PRD as a highly cost-effective strategy in patients with resistant hypertension, especially when implemented in early stages. In addition, I would like to point out that the global acceptance of PRD differs from that mentioned by Dr Kotliar. Currently, devices such as the Symplicity Spyral (Medtronic) and Paradise (ReCor) have been approved by the FDA (US Food and Drug Administration), and many others are CE (Conformité Européenne) marked, allowing their use in the treatment of resistant hypertension. Although more clinical evidence is still required to demonstrate a reduction in adverse clinical events associated with this therapy, there is abundant evidence on its safety profile. Therefore, we should not discard this option from our therapeutic arsenal, and it would be prudent to discuss it with our patients, considering their individual preferences.
ANTAGONIST REPLY
The agonist argumentation is certainly a valuable contribution that lists and describes the main results and trajectory of the available research. I certainly agree with the points that will hopefully be clarified in future studies.
However, in my view, I do not consider that we are in a position to consider PRD as an "additional treatment" for the management of resistant HTN. I base this on the fact that in none of the studies mentioned in the agonist position have all the necessary diagnostic requirements been fulfilled to "adequately" determine the condition of true resistance: for example, natriuresis to verify adherence to sodium intake restriction, evaluation of the correct intake of the indicated medications, among others.
On the other hand, international recommendations include non-invasive hemodynamic study in the management of resistant HTN, if available. In a study of the magnitude of those conducted to evaluate the effectiveness of different PRD catheters, it would be expected that more accurate and comprehensive information on hemodynamic and autonomic activation patterns would be available, in order to determine whether the patient's pharmacological treatment is the most appropriate before opting for invasive treatment.