I would like to express my gratitude to my friend and academic master, Marcelo V. Elizari, for his assistance in writing and critiquing this work. This work is dedicated to our unforgotten master, Mauricio B. Rosenbaum (RIP), with whom it all began.
To the editor:
The first manifestations of acute ischemia are high-peaked hyperacute T waves attributed to a sudden increase in potassium (K+) in the epicardium. It has been suggested that this phenomenon protects against the development of necrosis in this region of the myocardium caused by ischemia. (1)
The relationship between T wave voltage and extracellular K+ has been known for several decades. Hyperkalemia between 5.5 and 6.5 mEq/L generates high-peaked hyperacute T waves in precordial leads. Experiments have shown that blockade of K+ channels or knockout of the genes encoding K+ channels prevent the development of high-peaked hyperacute T waves in myocardial ischemia. (2,3) K+ influx produces taller and more narrow T waves in the epicardium, and this effect is less evident in the endocardium. (4) Notably, these T waves are present in sudden segmental, regional, transmural and subendocardial ischemia (Figures 1 and 2). (5) In the obstructive phase of anteroseptal myocardial infarctions, well protected by a good collateral circulation, only high-peaked hyperacute T waves are recorded in the electrocardiogram. (6) An experimental study in canine hearts reproduced changes similar to those observed in clinical practice with balloon occlusion of the left anterior descending coronary artery. Occlusion of the artery for 2 minutes produced a typical pattern of anteroseptal ischemia with ST-segment elevation of 9 mm and high-peaked hyperacute T waves of 15 mm (Figure 3). This experiment demonstrates the effect of diazoxide in increasing adenosine-triphosphate (ATP) dependent K+ levels in the epicardium. (7)
Figure 1.
Electrocardiographic changes of a sudden total occlusion of the left anterior descending coronary artery. The T wave changes between 10:55 and 10:58, becoming high-peaked hyperacute (13 mm wave) as the first manifestation of the ATP-dependent K+ influx to protect the epicardium until ST-segment elevation develops. High-peaked hyperacute T waves are due to the sudden influx of K+ into the epicardium. ST-segment elevation is related to calcium, which acts as a buffer in the center of ischemia with very low pH.

Figure 2.
Electrocardiographic changes of a sudden subtotal occlusion of left the anterior descending coronary artery, expressed with high-peaked hyperacute T waves and ST-segment-depression, suggesting acute subendocardial ischemia. See how the 15-mm high hyperacute T wave evolves. Although ischemia is subendocardial, the ATP-dependent K+ influx protects the epicardium. (5) Two hours after the onset of ischemia, a minimal subendocardial Q wave develops.

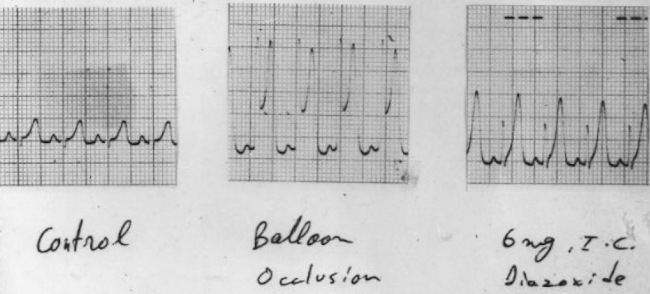
Figure 3.
Canine experiment. Left image: baseline recording. Central image: introduction of a balloon catheter and sudden occlusion of the left anterior descending coronary artery for 2 minutes. The typical 15-mm high T wave is observed. Right image: intracoronary injection of 5 mg of diazoxide. A T wave similar to an ischemic T wave is recorded. Diazoxide stimulates adenosine release, which increases epicardial K+ concentration.
Biological processes that reduce the effects of the ischemic storm
Mitochondria are independent organisms within the cells that have their own membranes and exhibit DNA similar to that of bacteria. (8) Their main function is to provide energy to cells. They produce ATP from fatty acids, glucose and oxygen via a complex enzyme system. (9) Phosphorus is released through a fission process and generates heat and energy. This is how mitochondria produce energy in all normally functioning cells. (10) The mechanism is similar to the one used by nuclear energy plants where uranium undergoes a fission process that produces heat and energy. Phosphorus is the second most abundant element in organisms and has been used as the main source of energy during evolution.(11) Consequently, it can be reasonably assumed that this source of energy is the most effective and cost-efficient option. The myocyte requires large amounts of energy to function and contains large numbers of mitochondria. During exercise, mitochondria in myocytes replicate into thousands, like bacterial binary fission, to provide the energy needed. (12) After mitochondrial fission, adenosine diphosphate (ADP) is formed, which apparently has no biological function. Part of it returns to the mitochondria to synthetize ATP, and another part loses phosphates. In acute ischemia, another phosphate is lost due to lack of oxygen and forms adenosine monophosphate (AMP). (13) This process plays an essential role in cellular biology by facilitating the influx of calcium through calcium channels into the sarcoplasmic reticulum (a calcium reservoir within the myocyte). Following phosphorus degradation, adenosine is released. The latter plays a crucial role in myocardial protection during ischemic storm. (14) Adenosine 1 blocks the effects of adrenaline on the sinus node preventing sinus tachycardia, which is a negative factor in acute ischemia. (15) An increase in energy demands during tachycardia is a high-risk factor in the event of a sudden reduction in coronary flow. Adenosine 1 is responsible for maintaining sinus rhythm in acute ischemia at a rate of 55 to 70 beats per minute despite the patient’s anxiety and suffering. In patients with acute myocardial infarction who did not receive medical care within the first 72 hours, sinus tachycardia may occur and is typically related to adenosine depletion. Adenosine 2 is present in the left ventricle and plays a vital role in the epicardium, as it increases K+ concentration. (16) This invasion of K+ in the epicardium is expressed by high-peaked (> 15 mm) hyperacute T waves in the anterior epicardial leads in anteroseptal myocardial infarctions and in some posteroinferior infarctions. These ischemic T waves are the expression of a shorter phase 2 of the epicardial action potential, during which calcium enters the myocyte. The decrease in calcium influx reduces epicardial contraction and thus decreases oxygen uptake (preconditioning). (16)
Anteroseptal transmural infarction involves necrosis of 2/3 of the myocardial wall (17,18,19). The third corresponding to the epicardium is protected by the increase in K+ concentration facilitated by adenosine 2. Anteroseptal infarctions complicated with cardiac rupture present T waves of approximately 5 mm or less, along with a reduced ST-T gradient. This suggests that the epicardium may not be protected by the mechanism of adenosine 2 and increased K+ concentrations. (20) Adenosine A2A has also an effect on the brain that is different from that of caffeine, suggesting that patients with acute infarction avoid moving to reduce the cardiac demands for energy, another effect to protect myocardial infarction in the obstructive stage. (21)
At the beginning of this century, several studies have been published on the mitochondrial function and dysfunction, especially in heart failure and hypertrophy. (22)
But we have not found publications in the medical literature addressing the importance of mitochondria and the biological cascade that begins with ATP and continues with adenosine upregulation and ATP-dependent K+ influx to protect the anteroseptal epicardium. In case of cardiac rupture in the context of acute myocardial infarction, we may speculate that it is a manifestation of mitochondrial dysfunction. Figure 3 shows the effect of intracoronary diazoxide injection in a canine experiment, stimulating adenosine production and inducing ATP-dependent K+ influx into the epicardium. The T wave obtained in the experiment is very similar to the T wave in occlusion of the left anterior descending artery (both waves measure 15 mm).
REFERENCES
2. Lukas A, Antzelevitch C. Differences in the electrophysiological response of canine ventricular epicardium and endocardium to ischemia. Role of the transient outward current. Circulation 1993;88:2903-15. https://doi.org/10.1161/01.cir.88.6.2903. PMID: 8252704.
3. Li RA, Leppo M, Miki T, Seino S, Marbán E. Molecular basis of electrocardiographic ST-segment elevation. Circ Res 2000;87:837-9. https://doi.org/10.1161/01.res.87.10.837.
4. Furukawa T, Kimura S, Furukawa N, Bassett AL, Myerburg RJ. Role of cardiac ATP-regulated potassium channels in differential responses of endocardial and epicardial cells to ischemia. Circ Res 1991;68:1693-702. https://doi.org/10.1161/01.res.68.6.1693.
5. Sclarovsky S, Rechavia E, Strasberg B, Sagie A, Bassevich R, Kusniec J, et al. Unstable angina: ST segment depression with positive versus negative T wave deflections--clinical course, ECG evolution, and angiographic correlation. Am Heart J 1988;116:933-41. https://doi.org/10.1016/0002-8703(88)90143-3.
6. Sagie A, Sclarovsky S, Strasberg B, Kracoff O, Rechavia E, Bassevich R, et al. Acute anterior wall myocardial infarction presenting with positive T waves and without ST segment shift. Electrocardiographic features and angiographic correlation. Chest 1989;95:1211-5. https://doi.org/10.1378/chest.95.6.1211.
7. Fedorov VV, Glukhov AV, Ambrosi CM, Kostecki G, Chang R, Janks D, et al. Effects of KATP channel openers diazoxide and pinacidil in coronary-perfused atria and ventricles from failing and non-failing human hearts. J Mol Cell Cardiol 2011;51:215-25. https://doi.org/10.1016/j.yjmcc.2011.04.016.
8. Boguszewska K, Szewczuk M, Kazmierczak-Baranska J, Karwowski BT. The Similarities between Human Mitochondria and Bacteria in the Context of Structure, Genome, and Base Excision Repair System. Molecules 2020;25:2857. https://doi.org/10.3390/molecules25122857.
9. Bertram R, Gram Pedersen M, Luciani DS, Sherman A. A #simplified model for mitochondrial ATP production. J Theor Biol 2006;243:575-86. https://doi.org/10.1016/j.jtbi.2006.07.019.
10. Bird RP, Eskin NAM. The emerging role of phosphorus in human health. Adv Food Nutr Res 2021;96:27-88. https://doi.org/10.1016/bs.afnr.2021.02.001.
12. Mishra P, Chan DC. Metabolic regulation of mitochondrial dynamics. J Cell Biol 2016;212:379-87. https://doi.org/10.1083/jcb.201511036.
13. Mubagwa K, Flameng W. Adenosine, adenosine receptors and myocardial protection: an updated overview. Cardiovasc Res 2001;52:25-39. https://doi.org/10.1016/s0008-6363(01)00358-3.
15. Crimm A, Severance HW Jr, Coffey K, McKinnis R, Wagner GS, Califf RM. Prognostic significance of isolated sinus tachycardia during first three days of acute myocardial infarction. Am J Med. 1984;76:983-8. https://doi.org/10.1016/0002-9343(84)90846-5.
16. Tseng CJ, Ho WY, Lin HC, Tung CS, Kuan CJ. Modulatory effects of endogenous adenosine on epinephrine secretion from the adrenal medulla of the rat. Hypertension 1994;24:714-8. https://doi.org/10.1161/01.hyp.24.6.714.
17. Murry CE, Jennings RB, Reimer KA. Preconditioning with ischemia: a delay of lethal cell injury in ischemic myocardium. Circulation. 1986;74:1124-36. https://doi.org/10.1161/01.cir.74.5.1124.
18. O'Rourke B. Myocardial K(ATP) channels in preconditioning. Circ Res. 2000;87:845-55. https://doi.org/10.1161/01.res.87.10.845.
19. Liu Y, Sato T, O'Rourke B, Marban E. Mitochondrial ATP-dependent potassium channels: novel effectors of cardioprotection? Circulation. 1998;97:2463-9. https://doi.org/10.1161/01.cir.97.24.2463.
21. Lazarus M, Shen HY, Cherasse Y, Qu WM, Huang ZL, Bass CE, et al. Arousal effect of caffeine depends on adenosine A2A receptors in the shell of the nucleus accumbens. J Neurosci. 2011;31:10067-75. https://doi.org/10.1523/JNEUROSCI.6730-10.2011.
22. Zhou B, Tian R. Mitochondrial dysfunction in pathophysiology of heart failure. J Clin Invest. 2018;128:3716-26. https://doi.org/10.1172/JCI120849.
22. Zhou B, Tian R. Mitochondrial dysfunction in pathophysiology of heart failure. J Clin Invest. 2018;128:3716-26. https://doi.org/10.1172/JCI120849.
