INTRODUCTION
The left atrium (LA) plays an important role in cardiac physiology, and its volume has been associated with various clinical conditions such as atrial fibrillation, heart failure, and increased cardiovascular risk. (1-3) Therefore, its volumetric measurement is an essential parameter in imaging studies in clinical practice.
The most commonly used echocardiographic methods for estimating LA volume are area-length (AL) and Simpson methods. (4,5) Cardiac magnetic resonance (CMR) imaging allows global and multiplanar assessment of the LA, and indirect estimates or direct quantification can be performed, the latter being the most accurate but also the most labor-intensive. The AL method has been the most widely used in CMR imaging from the beginning and remains the method of choice in our setting. (6-10) Despite its greater simplicity, it may not accurately reflect LA volume. Direct quantification of LA volume requires a greater number of slices for CMR imaging acquisition, resulting in a longer study and its subsequent analysis. (10-12)
Objectives
To propose and validate an alternative method for estimating LA volume using CMR. To compare accuracy and agreement of this new method with the AL method, using direct quantification by complete volumetric tracing of the LA in CMR imaging studies as the gold standard.
METHODS
Study population
Twenty-five patients aged over 18 years were randomly selected from a database of CMR imaging at our center. Patients were excluded if they had complex atrial anatomy, such as congenital heart disease with Fontan-Kreutzer circulation, poor image quality due to technical artifacts, or if, for any reason, they had not completed acquisition of the basic images required for analysis.
CMR imaging protocol
The images were acquired using a Siemens Magneton Aera 1.5 T scanner. Image acquisition was performed in the supine position, with retrospective electrocardiography gating. The center basic CMR imaging acquisition protocol includes cine images obtained using steady-state free precession (SSFP) sequences in 2-chamber (2C), 3-chamber (3C), 4-chamber (4C), and complete short-axis (SA) views. During acquisition of the complete ventricular short-axis stack, the center protocol routinely includes the atria.
Image analysis
All images were analyzed using the free software Segment v4.0 (Medvisto, Lund, Sweden). Two independent analyses were performed by two researchers experienced in the method. All measurements were performed at end-systole. The LA volume was directly quantified by manual tracing, excluding the left atrial appendage and pulmonary veins on all slices, and this was assumed to be the reference value. The estimation was performed using the usual biplane area-length (AL) method and the newly proposed irregular base cylinder (CBI) method.
When estimating the volume using the AL method, the area and height (length) of the LA were measured in the 2C and 4C cine views. The following formula was used: 8/3 II (2C area x 4C area) / length.
When estimating the volume using the CBI method, the base of the cylinder was measured. The atrial area was traced in a perpendicular slice to its major axis, obtained from the complete short-axis acquisition between the orifice of the left atrial appendage and the orifices of the pulmonary veins; the atrial height was measured from the mitral annulus to the atrial roof in the 4C view. The volume was calculated using the area x height formula.
Statistical analysis
Continuous variables are presented as mean ± standard deviation (SD) or median and interquartile range (IQR) according to their distribution. Categorical variables are presented as frequencies and percentages. Both estimates were compared with the gold standard (direct volumetric) using Pearson's correlation coefficient (r). A Bland-Altman analysis was performed to assess mean differences and dispersion between both methods and the gold standard.
RESULTS
Baseline characteristics
From our center imaging database, we analyzed previously acquired images from 25 patients with a mean age of 44± 15 years. Fifty-two percent were male, and 84% had been studied on an outpatient basis. The most frequent diagnoses included hypertrophic cardiomyopathy (20%) ,ventricular arrhythmia (16%) , ischemic heart disease (16%) , and dilated cardiomyopathy (12%) .The mean left ventricular ejection fraction LVEF) was 59± 8% and the mean left atrial volume was 97± 38 mL . The remaining baseline characteristics are presented in Table 1.
Table 1.
Population characteristics
| Variables | Value |
|---|---|
| Age, years, mean (SD) | 45 (15) |
| Male sex, n (%) | 13 (52) |
| Weight, kg, mean (SD) | 80 (19) |
| Height, cm - mean (SD) | 169 (24) |
| Body surface area, m², mean (SD) | 1.94 (0.29) |
| LV EDV, mL, median (IQR) | 161 (134-187) |
| LV ESV, mL, median (IQR) | 63 (42-85) |
| LVEF, %, mean (SD) | 59 (8) |
| LV mass, g, mean (SD) | 115 (43) |
| LA volume, mL, mean (SD) | 97 (38) |
| Main pathology | |
| Hypertrophic cardiomyopathy, n (%) | 5 (20) |
| Ventricular arrhythmia, n (%) | 4 (16) |
| Ischemic heart disease, n (%) | 4 (16) |
| Dilated cardiomyopathy, n (%) | 3 (12) |
| Other, n (%) | 9 (36) |
EDV: end-diastolic volume; ESV: end-systolic volume; IQR: interquartile range; LA: left atrium; LV: left ventricle; LVEF: left ventricular ejection fraction; SD: standard deviation
Quantification methods
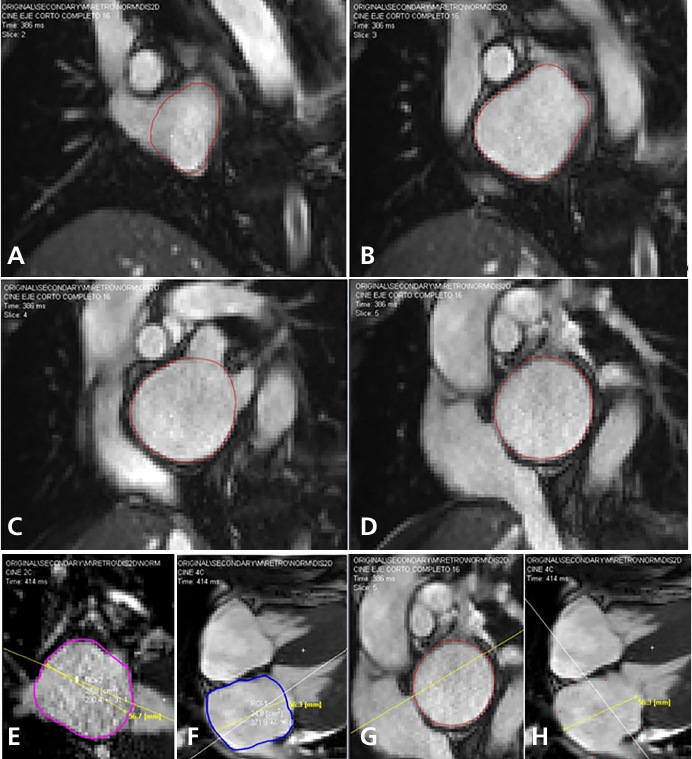
Figure 1 shows images of direct LA volume measurement using the AL and CBI methods.
Fig. 1.
Left atrial measurements performed in the complete short-axis, 4C, and 2C views with manual endocardial tracing at end-systole. Direct volumetry (A-B-C-D). AL method (E-F). CBI method (G-H)
The correlation measured against direct volumetric quantification was very high for both methods (Figure 2). Pearson's correlation coefficient was higher for the CBI method (r = 0.93 p < 0.001 ) than for the AL method (r = 0.83 p < 0.001 ). The Bland-Altman analysis showed low bias for both methods, with less dispersion for the CBI method [- 5.73 mL (18.6 to - 30.11)] than for the AL method [-1.18 mL (43.82 to -46.17)]. Interobserver variability was lower for the CBI method [4.94 mL (44.76 to - 34.88)] than for the AL method [19.7 mL (65.91 to - 26.51)] (Figure 3).
Fig. 2.
Correlation between measurements by direct volumetry and area-length method (blue) and CBI method (red).
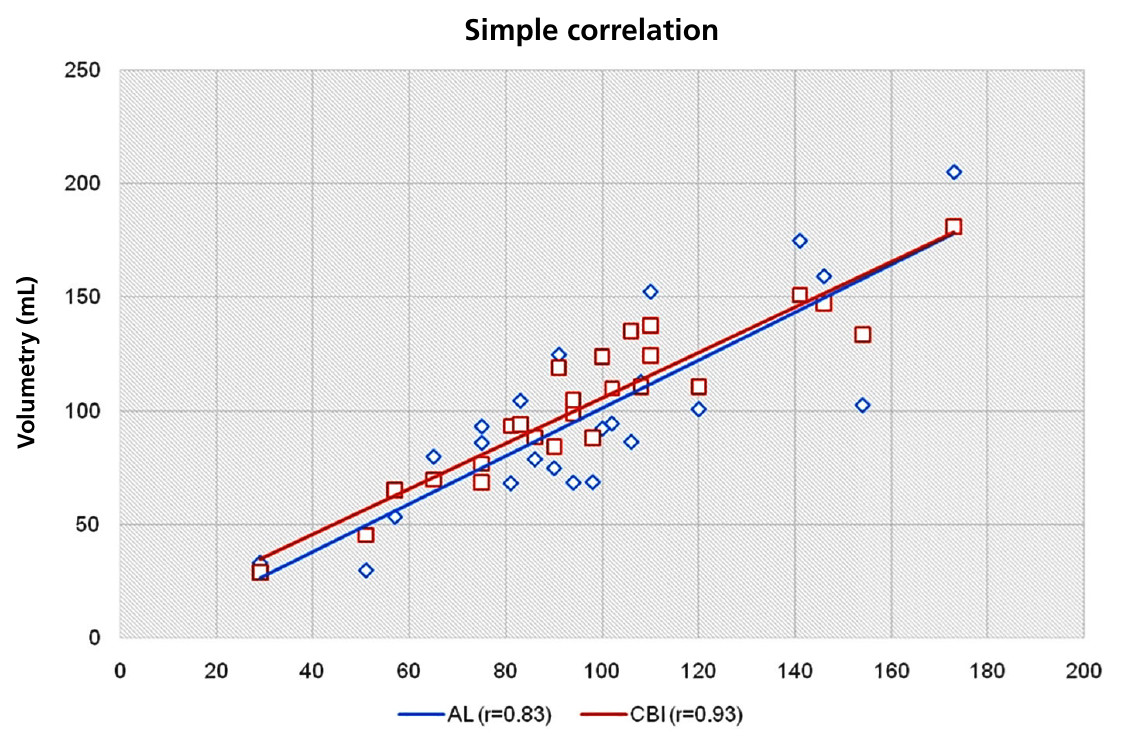
Fig. 3.
Mean differences and dispersion in the measurement of left atrial volume: (A) Direct volumetry and area-length method. (B) Direct volumetry and irregular base cylinder method. Interobserver variability: (C) area-length method, (D) irregular base cylinder method.
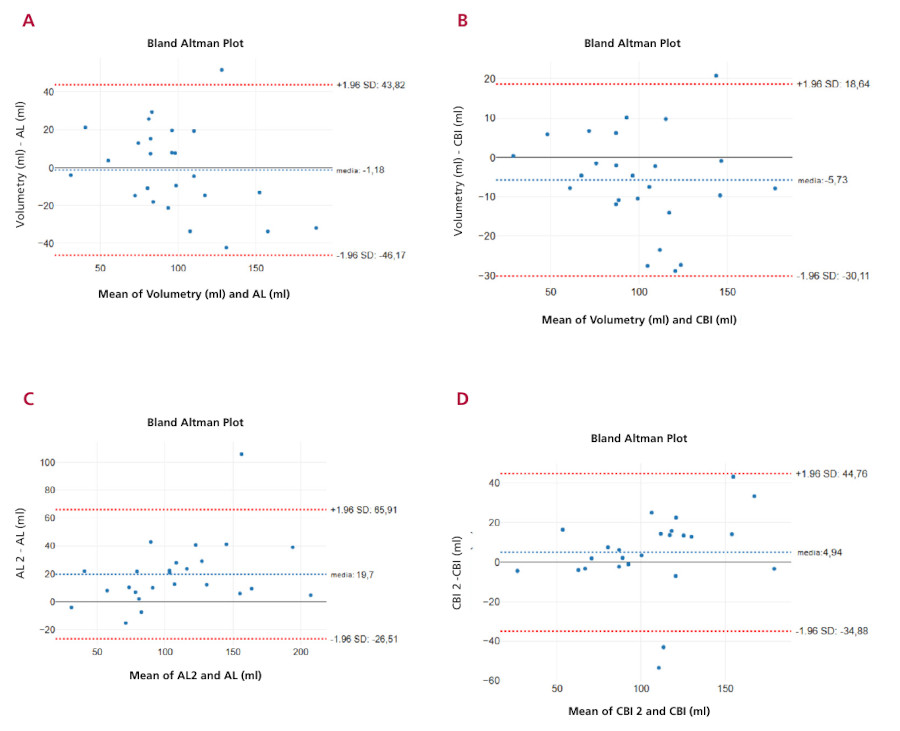
AL: area-lenght; CBI: irregular base cylinder
DISCUSSION
Our results show an excellent correlation between both methods and direct volumetric quantification, with a higher correlation for the newly proposed method (CBI). In addition, dispersion with the new method is lower than with the AL method, and its interobserver reproducibility is higher.
It should be noted that acquiring the entire atrial volume is not necessary, since the base of the cylinder can be obtained with a slice beyond the mitral annulus plane. This makes the method practical and does not require additional time for image acquisition.
The AL method is excellent, and its use in two-dimensional echocardiography has probably been the best option for approximating the actual volume, given the impossibility of direct quantification or the interposition of strictly orthogonal short-axis planes. For years, this method, derived from echocardiography, was adopted in CMR imaging because of its good correlation with direct quantification and the ease of image acquisition and processing.
From a clinical perspective, accurate estimation of LA volume is particularly important. In the context of remodeling, LA dilation is a marker of what is occurring in a patient's chronic hemodynamics. Volumetric measurement can provide a parameter for longitudinal assessment and is an independent predictor for major cardiovascular events, atrial fibrillation, heart failure, and embolic events. (13-20) In some cases, small differences in LA volumetric estimation may lead to a different diagnostic interpretation when values are close to the cutoff point defined by clinical guidelines or consensus statements. Therefore, greater precision in estimation would be expected to improve diagnostic accuracy and consequently benefit clinical practice.
Furthermore, according to our observations, the lower interobserver variability makes CBI a more robust method that may be particularly valuable in multicenter studies or serial evaluations, where methodological stability is essential to detect real changes in atrial remodeling.
In summary, CMR has the advantage over two-dimensional echocardiography of allowing image slices at any desired orientation and facilitates the use of geometric figures and shapes that better fit volumetric estimation. We believe that the CBI method, with its slightly more accurate approximation than the method commonly used, without compromising its practicality, may improve the interpretation of the study.
Limitations
A small number of cases were used assuming a coefficient r > 0.8 and although the selection in the database was random, this geometric model and the AL method may not be extrapolated to all atrial morphologies.
CONCLUSIONS
The CBI method showed a better correlation with the direct measurement standard than that the AL method. It therefore represents a valid alternative that is equally simple and more accurate than the method commonly used to estimate LA volume by CMR.
Conflicts of interest
None declared. (See authors' conflict of interests forms on the web).
Funding
This work received no financial support.
REFERENCES
1. Le TT, Tan RS, De Deyn M, Goh EP, Han Y, Leong BR, et al. Cardiovascular magnetic resonance reference ranges for the heart and aorta in Chinese at 3T. J Cardiovasc Magn Reson 2016;18:21. https://doi.org/10.1186/s12968-016-0236-3
2. Petersen SE, Aung N, Sanghvi MM, Zemrak F, Fung K, Paiva JM, et al. Reference ranges for cardiac structure and function using cardiovascular magnetic resonance (CMR) in Caucasians from the UK Biobank population cohort. J Cardiovasc Magn Reson 2017;19:18. https://doi.org/10.1186/s12968-017-0327-9
3. Zemrak F, Ambale-Venkatesh B, Captur G, Chrispin J, Chamera E, Habibi M, et al. Left Atrial Structure in Relationship to Age, Sex, Ethnicity, and Cardiovascular Risk Factors: MESA (Multi-Ethnic Study of Atherosclerosis). Circ Cardiovasc Imaging 2017;10:9. https://doi.org/10.1161/CIRCIMAGING.116.005379
4. Lang RM, Badano LP, Mor-Avi V, Afilalo J, Armstrong A, Ernande L, et al. Recommendations for cardiac chamber quantification by echocardiography in adults: an update from the American Society of Echocardiography and the European Association of Cardiovascular Imaging. Eur Heart J Cardiovasc Imaging. 2015;16:233-70. https://doi.org/10.1093/ehjci/jev014
5. Deschle HA, Allende N, Miranda A, Lakowsky A, Sanchez Luceros D, Carbajales J, et al. Left atrial volume indexed by body surface area in normal subjects divided by sex and age. Rev Argent Cardiol. 2010;77:39-42.
6. Nacif MS, Barranhas AD, Turkbey E, Marchiori E, Kawel N, Mello RA, et al. Left atrial volume quantification using cardiac MRI in atrial fibrillation: comparison of the Simpson's method with biplane area-length, ellipse, and three-dimensional methods. Diagn Interv Radiol 2013;19:213-20. https://doi.org/10.5152/dir.2012.002
7. Maceira AM, Cosin-Sales J, Roughton M, Prasad SK, Pennell DJ. Reference left atrial dimensions and volumes by steady state free precession cardiovascular magnetic resonance. J Cardiovasc Magn Reson 2010;12:65. https://doi.org/10.1186/1532-429X-12-65
8. Funk S, Kermer J, Dogganuezel S, Schwenke C, von Knobelsdorff-Brenkenhoff F, Schulz-Menger J. Quantification of the left atrium applying cardiovascular magnetic resonance in clinical routine. Scand Cardiovasc J 2018;52:85-92. https://doi.org/10.1080/14017431.2017.1423107
9. Rohner A, Brinkert M, Kawel N, Buechel RR, Leibundgut G, Grize L, et al. Functional assessment of the left atrium by real-time three-dimensional echocardiography using a novel dedicated analysis tool: initial validation studies in comparison with computed tomography. Eur J Echocardiogr 2011;12:497-505. https://doi.org/10.1093/ejechocard/jer066
10. Hudsmith LE, Petersen SE, Francis JM, Robson MD, Neubauer S. Normal human left and right ventricular and left atrial dimensions using steady state free precession magnetic resonance imaging. J Cardiovasc Magn Reson 2005;7:775-82. https://doi.org/10.1080/10976640500295516
11. Maceira AM, Cosin-Sales J, Roughton M, Prasad SK, Pennell DJ. Reference left atrial dimensions and volumes by steady state free precession cardiovascular magnetic resonance. J Cardiovasc Magn Reson 2010;12:65. https://doi.org/10.1186/1532-429X-12-65
12. Rodevand P, Bjornerheim R, Ljosland M, Maehle J, Smith HJ, Ihlen H. Left atrial volumes assessed by three- and two-dimensional echocardiography compared to MRI estimates. International Journal of Cardiac Imaging 1999;15:397-410. https://doi.org/10.1023/A:1006276513186
13. Vaziri SM, Larson MG, Benjamin EJ, Levy D. Echocardiographic predictors of nonrheumatic atrial fibrillation. The Framingham Heart Study. Circulation 1994;89:724-30. https://doi.org/10.1161/01.CIR.89.2.724
14. Tsang TS, Barnes ME, Bailey KR, Leibson CL, Montgomery SC, Takemoto Y, et al. Left atrial volume: important risk marker of incident atrial fibrillation in 1655 older men and women. Mayo Clin Proc 2001;76:467-75. https://doi.org/10.4065/76.5.467
15. Tsang TS, Barnes ME, Gersh BJ, Bailey KR, Seward JE. Left atrial volume as a morphophysiologic expression of LV diastolic dysfunction and predictor of cardiovascular risk burden. Am J Cardiol 2002;15:90:1284-9. https://doi.org/10.1016/S0002-9149(02)02864-3
16. Benjamin EJ, D'Agostino RB, Belanger AJ, Wolf PA, Levy D. Left atrial size and the risk of stroke and death. Circulation 1995;15:92:835-41. https://doi.org/10.1161/01.CIR.92.4.835
17. Barnes ME, Miyasaka Y, Seward JB, Gersh BJ, Rosales AG, Bailey KR, et al. Left atrial volume in the prediction of first ischemic stroke in an elderly cohort without atrial fibrillation. Mayo Clin Proc 2004;79:1008-14. https://doi.org/10.4065/79.8.1008
18. Tsang TS, Gersh BJ, Appleton CP, Tajik AJ, Barnes ME, Bailey KR, et al. Left ventricular diastolic dysfunction as a predictor of the first diagnosed nonvalvular atrial fibrillation in 840 elderly men and women. J Am Coll Cardiol 2002;40:1636-44. https://doi.org/10.1016/S0735-1097(02)02373-2
19. Tsang TS, Barnes ME, Gersh BJ, Bailey KR, Seward JB. Risks for atrial fibrillation and congestive heart failure in patients > = 65 years of age with abnormal left ventricular diastolic relaxation. Am J Cardiol 2004;93:54-8. https://doi.org/10.1016/j.amjcard.2003.09.012
20. Sabharwal N, Cemin R, Rajan K, Hickman M, Lahiri A, Senior R. Usefulness of left atrial volume as a predictor of mortality in patients with ischemic cardiomyopathy. Am J Cardiol 2004;94:760-3. https://doi.org/10.1016/j.amjcard.2004.05.060
HTML generado desde un xml-jats a través de la plantilla jats-rac.xsl por RevisCiencia. El motor utilizado es Apache Software Foundation (Xalan XSLTC).
