Belén Davico1, Maximiliano Martín1, Ezequiel Lozano Chiappe1, Rodrigo Esparza Iraola2 MTSAC, Marina Marchesini2 MTSAC, María Pérez2, Leonardo Gómez Rosso1, Laura Boero1, Gustavo Giunta2 MTSAC, Fernando Brites1
1 Laboratory of Lipids and Atherosclerosis, Department of Clinical Biochemistry, School of Pharmacy and Biochemistry, University of Buenos Aires. Buenos Aires, Argentina.
2 Department of Clinical Cardiology, Fundación Favoloro. Buenos Aires, Argentina.
Address for reprints: Belén Davico. E-mail: belendavico@hotmail.com
Rev Argent Cardiol 2024;92:124-129. http://dx.doi.org/10.7775/rac.v92.i2.20753
ABSTRACT
Background: Post-COVID syndrome (PCS), characterized by symptoms that persist for more than 4 weeks after initial infection, could increase cardiovascular risk. High-density lipoproteins (HDL) have antiatherogenic functions, such as the ability to promote reverse cholesterol transport (RCT) and antioxidant activity. In this regard, paraoxonase 1 (PON 1) plays a key role.
Objective: The aim of this study was to evaluate HDL functions in patients with PCS and compare them with asymptomatic patients (AP) and controls.
Methods: The study included 9 patients with PCS, 18 AP and 10 controls. Complete blood count, basic lipoprotein profile, apolipoproteins A-I and B, and inflammatory markers were measured using automated methods. PON 1 activity was evaluated by a spectrophotometric assay, and the 3 steps of RCT, cellular cholesterol (efflux CCE), lecithin-cholesterol acyltransferase (LCAT) activity and cholesteryl ester transfer protein (CETP) activity were evaluated by radiometric assays.
Results : There were no differences in sex, age, or general parameters. The AP group had higher PON activity than the control group (94±76 vs. 183±111 vs. 148±58 nmol/mL.min, in controls, AP and PCS, respectively; p=0.049). There were no differences in RCT. Cellular cholesterol efflux (r=-0.45; p=0.049) and CETP (r=- 0.38; p=0.028) had a negative correlation with neutrophil-to-lymphocyte ratio. LCAT had an inverse correlation with ferritin (r=-0.34; p=0.046).
Conclusions: Increased antioxidant activity of PON 1 would represent a defensive mechanism against oxidative stress after infection. All the RCT steps had a negative correlation with inflammatory markers. Our findings may explain, at least in part, the link between COVID-19 and atherosclerosis.
Key words: Post COVID Syndrome - Cholesterol HDL - Lipoproteins - Oxidative stress
RESUMEN
Introducción: El síndrome post COVID (SPC), que se caracteriza por síntomas que se extienden superando las 4 semanas post-infección, podría desencadenar aumento en el riesgo cardiovascular. Las lipoproteínas de alta densidad (HDL) presentan funciones antiaterogénicas, como su capacidad para promover el transporte inverso del colesterol (TIC) y su actividad antioxidante, en la que es clave la enzima paraoxonasa 1 (PON 1).
Objetivo: Evaluar funcionalidad de HDL en pacientes con SPC comparados con pacientes asintomáticos (PA) y controles.
Material y métodos: Se incluyeron 9 individuos con SPC, 18 PA y 10 controles. Se midieron el hemograma, el perfil lipoproteico básico, las apolipoproteínas A-I y B, y marcadores inflamatorios por métodos automatizados. La actividad de PON 1 se evaluó empleando un método espectrofotométrico y los 3 pasos del TIC, eflujo de colesterol (ECC), y actividades de lecitina:colesterol aciltransferasa (LCAT) y proteína transportadora de colesterol esterificado (CETP), por métodos radiométricos.
Resultados: No se observaron diferencias en sexo, edad, ni parámetros generales. El grupo PA presentó mayor actividad PON que los controles (94±76 vs. 183±111 vs. 148±58 nmol/mL.min, en controles, PA y SPC, respectivamente; p=0,049). No se observaron diferencias en el TIC. El ECC (r=-0,45; p=0,049) y CETP (r=-0,38; p=0,028) correlacionaron negativamente con el índice neutrófilos/linfocitos. LCAT correlacionó inversamente con la ferritina (r=-0,34; p=0,046).
Conclusiones: El incremento de PON 1 en el grupo PA representaría un mecanismo de defensa frente al estrés oxidativo post-infección. Todos los pasos del TIC mostraron una correlación negativa con marcadores inflamatorios. Nuestros resultados podrían explicar, en parte, el vínculo entre COVID y ateroesclerosis.
Palabras clave: Síndrome post COVID - HDL - Lipoproteínas - Estrés oxidativo
Received: 11/6/2023
Accepted: 02/272024
INTRODUCTION
The coronavirus disease described in December 2019 (COVID-19) is an infection caused by severe acute respiratory syndrome coronavirus 2 (SARS-Cov-2). (1) Post-COVID syndrome (PCS) is a multisystemic disease characterized by signs and symptoms that develop during or after an acute infection and persist for more than 4 weeks after symptom onset. (2) The most common clinical symptoms are fatigue, dyspnea, chest pain, cough, myalgia, headache, and palpitations. (3) This condition can cause a significant decline in the quality of life of those affected. (4) It is estimated that at least 10% of infections result in PCS, affecting 65 million people worldwide. (5)
SARS-CoV-2 enters the host cells due to the interaction of the viral spike protein with the angiotensin-converting enzyme 2 (ACE2) receptor. (6) As a result, ACE2 is less available to catalyze the conversion of angiotensin II to angiotensin 1-7 and angiotensin II levels increase. Under this condition, angiotensin II binds the angiotensin II type 1 receptor (AT1R), which stimulates oxidized nicotinamide dinucleotide phosphate (NADPH) activity. This results in reducing O2 to superoxide and increasing the production of other reactive oxygen species (ROS). (7,8). This is the main mechanism leading to increased oxidative stress in patients with COVID-19 and is facilitated by the pro-inflammatory context, as the identification of the virus promotes the release of cytokines that recruit macrophages and neutrophils. These cells are responsible for the production of ROS and other mediators that contribute further to oxidative stress and cytokines synthesis. Cytokine release is prolonged and disproportionate and is known as cytokine storm, which is closely related to disease severity. ((9),(10)) It can cause tissue damage and symptoms that persist beyond the acute stage of infection and may be responsible, at least in part, for the development of PCS. Increased oxidative stress impairs mitochondrial function and affects cell proliferation, extracellular matrix remodeling, and lung defensive mechanisms. (11) In this context, the role of antioxidants has become relevant and even proposed as therapy for PCS. (12,13)
Some patients, while they were suffering from COVID-19, had lower levels of high-density lipoprotein cholesterol (HDL-C), the only lipoprotein fraction with antiatherogenic properties. This decrease was associated with disease severity and exacerbated cytokine storm. (14,15) In addition, HDL-C levels were significantly associated with prolonged virus clearance time. (16,17) This highlights the potential antiviral role of HDL, which, in addition to promoting reverse cholesterol transport, has antithrombotic, antiapoptotic, anti-inflammatory, and antioxidant functions. The antioxidant activity of HDL is due to the paraoxonase (PON) 1 which is located on it and prevents low-density lipoprotein (LDL) from oxidation. (18) For these reasons, the evaluation of HDL function becomes relevant in a context of high oxidative stress and inflammation with high risk of cardiovascular disease such as PCS.
The aim of the present study was to assess the primary cardioprotective functions of HDL, including its antioxidant activity and its ability to facilitate reverse cholesterol transport. This process encompasses three steps: a) cellular cholesterol efflux, b) esterification of free cholesterol by the enzyme lecithin-cholesterol acyltransferase (LCAT), and c) cholesteryl ester transfer protein (CETP) transfer of cholesterol from HDL to apoB-containing lipoproteins in exchange for triglycerides, a process with therapeutic relevance. (19) Impairment of some HDL functions may be associated with slow clearance of SARS-CoV-2 from the body and progression to PCS.
METHODS
Study design and population
We conducted a cross-sectional, observational and collaborative study between Favaloro Foundation University Hospital and the Laboratory of Lipids and Atherosclerosis, School of Pharmacy and Biochemistry, University of Buenos Aires, between June 2021 and February 2022. The inclusion criteria included men and women between 20 and 60 years with a history of COVID-19 confirmed by polymerase chain reaction (PCR) between 4 and 12 weeks prior to inclusion and who signed the informed consent form. This period corresponds to the symptomatic PCS defined in the National Institute for Health and Care Excellence (NICE) 2020 guidelines. (2) Based on the presence of persistent symptoms, a group of patients with PCS (PCS group, n=9) was compared with a group of patients who had experienced the disease and subsequently evolved without any symptoms (AP group, n=18). A control group of patients with no diagnosis of COVID-19 was incorporated in the last year (control group, n=10). The following exclusion criteria were considered: symptoms prior to COVID-19, body mass index (BMI) > 35 kg/m2, presence of comorbidities (chronic kidney disease, type 1 or type 2 diabetes mellitus, chronic liver disease, chronic inflammation, structural or functional heart disease, lung disease), patients treated with ACE inhibitors, angiotensin II receptor antagonists, statins or corticosteroids, and pregnancy. The study protocol was approved by the Committee on Ethics of Favaloro Foundation and followed the recommendations of the Declaration of Helsinki and subsequent amendments.
Evaluation of the clinical and anthropometric characteristics
All the selected subjects underwent clinical examination, and their medical history was recorded. Weight and height were measured to calculate BMI. Blood pressure (BP) was measured three times with a certified sphygmomanometer (Welch-Allyn, USA), and the average of the last two determinations was considered. To verify the persistence of symptoms and to assess functional capacity, a 6-minute walk test was conducted, and the rating of perceived exertion was measured using the Borg scale. ((20),21)
Samples
Blood samples were obtained from the antecubital vein after a 12-hour fast. Samples were collected in tubes containing clot accelerator and gel serum separator and in Na2EDTA tubes and centrifuged at 1500 rpm for 15 minutes. Serum and/or plasma samples was separated, as appropriate, and aliquots were stored at 4 ºC and -70 ºC.
Determination of general and specific biochemical parameters
Plasma levels of blood urea nitrogen and creatinine and complete blood count were determined in accordance with standard methods.
Characterization of the inflammatory status
High-sensitivity C-reactive protein (hs-CRP) and ferritin concentrations were determined by immunoturbidimetric assays on a COBAS c 501 analyzer (Roche S.A.Q. e I., Switzerland). Neutrophil-to-lymphocyte ratio was calculated as an inflammatory marker. (22)
Determination of lipids, liproproteins, and apolipoproteins
Plasma levels of triglycerides, total cholesterol, and HDL-C were measured according to standard methods. LDL-C was calculated using the Friedewald formula or the Sampson formula according to triglyceride levels. Apo B and apo A-I concentrations were determined by immunoturbidimetric assays on a COBAS c 501 analyzer (Roche, Basel, Switzerland).
Paraoxonase 1 activity
PON 1 activity was evaluated by a spectrophotometric assay using two substrates: paraoxon (paraoxonase activity itself, PON) and phenylacetate (arylesterase activity, ARE). Both activities were measured following a previously described method with minimal modifications. (23) The phenotypes for the Q192R genetic variant of the PON1 gene were estimated by the double substrate method. (24)
Evaluation of the ability of HDL to promote cellular cholesterol efflux
HDL was isolated by selective precipitation using 45% polyethylene glycol (6000) and 0.2 M Tris-HCl buffer (pH = 8.2). The ability of HDL to promote cellular cholesterol efflux was analyzed using the human monocyte cell line THP-1. (25)
Determination of lecithin cholesterol acyltransferase activity
LCAT activity was determined using a radiometric assay based on a modified version of the protocol of Aguilar-Espinoza et al. (26)
Determination of cholesteryl ester transfer protein
CETP was measured with a radiometric assay previously described with minimal modifications. (27)
Statistical analysis
The sample size was estimated using Open Epi software (MIT, USA) considering 80% power and an alpha level of 0.05. We chose PON activity as the dependent variable and used previous unpublished data from the group in similar populations. The sample size was estimated in 8 subjects per group. Quantitative variables were expressed as mean and standard deviation, or median and interquartile range (Q1-Q3), according to their distribution. The assumption of normal distribution of the population for each variable was evaluated using descriptive statistics (comparison of means and medians), graphics (histogram and quantile-quantile plot) and goodness-of-fit tests (Kolmogorov-Smirnov test and Shapiro-Wilk test). ANOVA or Kruskal-Wallis test was used to compare continuous variables according to data distribution. The correlations were evaluated using Pearson's correlation coefficient for parametric variables or Spearman's correlation coefficient for non-parametric variables. The differences in categorical variables were compared using the chi-square test. All the statistical calculations were performed using Infostat (National University of Cordoba, Cordoba, Argentina) and SPSS 26.0 software packages (Chicago, Illinois, USA) A two-tailed p value < 0.05 was considered statistically significant.
RESULTS
There were no differences in the distribution of sex, age, height, weight, BMI, and BP among the 3 groups evaluated in the study (Table 1). Patients in the PCS group presented dyspnea (69%), palpitations (54%) and asthenia (46%) as the main symptoms. Consistent with these symptoms, subjects in the PCS group reported a higher score on the Borg scale in the 6-minute walk test (1.6±1.0 vs. 1.2±1.0 vs. 3.5±1.9; control group, AP, and PCS, respectively; p=0.017). There were no significant differences in any of the general biochemical parameters or in the complete blood count (Table 1).
Furthermore, plasma levels of total cholesterol, LDL-C, HDL-C, triglycerides, Apo A-I or B did not differ significantly (Table 2). The inflammatory markers (hs-CRP, ferritin and neutrophil-to-lymphocyte ratio) were similar among the 3 groups (Table 3).
When the three steps of reverse cholesterol transport were analyzed, none of them showed significant differences (Figure 1). However, all three presented associations with other parameters. The results indicated a negative correlation between cellular cholesterol efflux and the neutrophil-to-lymphocyte ratio (r = -0.45; p=0.049), an inverse correlation between LCAT activity and the acute phase reactant ferritin (r = -0.34; p = 0.046), while CETP activity demonstrated a positive correlation with triglyceride levels (r = 0.47; p = 0.006) and a negative correlation with the neutrophil-to-lymphocyte ratio (r = -0.38; p =0.028).
The AP group had higher PON activity than the control group (94±76 vs. 183±111 vs. 148±58 nmol/mL.min, in controls, AP and PCS, respectively; p=0.049), with no significant differences in ARE activity between the three groups (Figure 2). In addition, all groups showed a similar phenotypic distribution of the Q192R genetic variant of the PON1 gene, allowing for comparison between groups.
DISCUSSION
The results show that PCS is not associated with changes in reverse cholesterol transport in the groups analyzed. However, we observed a change in HDL antioxidant function represented by higher PON activity in patients who resolved symptoms compared to healthy controls. This function is particularly important during viral infections, since the performance and survival of immune cells is under redox control and depends on ROS levels (28) which are increased in PCS. Therefore, the increased PON activity could be explained as a defensive mechanism against increased oxidative stress. In fact, this has already been described in other conditions associated with increased oxidative stress such as physical activity. (29,30) In addition to PON activity, ARE activity, which is not affected by genetic variants and is considered a marker of PON 1 concentration, was also analyzed. (24) Therefore, the presence of increased PON activity with preserved ARE activity would indicate a specific increase in the intrinsic activity of the enzyme without changes in its concentration. This response, observed in patients whose symptoms have resolved and not in patients with PCS, may explain, at least in part, the persistence of symptoms in PCS patients. Indeed, previous studies have indicated that HDL may play an antiviral role, particularly in the context of COVID-19. (17) In addition, the antioxidant activity of HDL attributable to PON 1, as well as its general antiatherogenic properties, may have an influence in suppressing the replication of SARS-Co-V2. (17) Consistent with this, a previous study showed that PON1 Q192R polymorphism was associated with less severe COVID-19. (31)
It is worth noting that the inflammatory markers measured in our population were not elevated. However, there is a negative correlation between each step of reverse cholesterol transport and some of these markers. This would confirm the previously reported impact of persistent inflammation on HDL function and the development of atherosclerotic cardiovascular disease. ((32),(33)) In fact, in a previous paper we showed the negative association between the presence of vascular-specific inflammation and reverse cholesterol transport in obese children and adolescents. (34)
Persistent symptoms in the PCS group were associated with a higher score reported on the Borg scale for the 6-minute walk test. This test has been widely used to measure functional exercise capacity, (35) which has been found to be impaired in patients after acute SARS-CoV-2 infection. (36) The discrepancy between symptoms and inflammatory markers may be attributed to the heterogeneity of these markers reported in different studies. As we have previously explained, persistent inflammation may play an important role in the development of PCS. (9,10) However, the evidence is inconclusive. A recent study demonstrated that persistent inflammation was present in approximately 60% of patients with PCS. (37) Furthermore, the persistence of inflammation would be determined by several factors, such as the severity of the acute infection, age, or BMI. (37) A meta-analysis analyzing 113 inflammatory markers in PCS reported that hs-CRP and ferritin were not elevated even in the presence of persistent inflammation. (38) The inflammatory status of patients with PCS would be defined by markers of low-grade inflammation such as interferon. (3738) Thus, the diversity present in patients with PCS may explain the lack of differences in inflammatory markers found in our study. However, the presence of low-grade inflammation cannot be excluded, which may partly explain the negative association between inflammatory markers and HDL function observed in PCS patients.
Table 1. Anthropometric and biochemical parameters
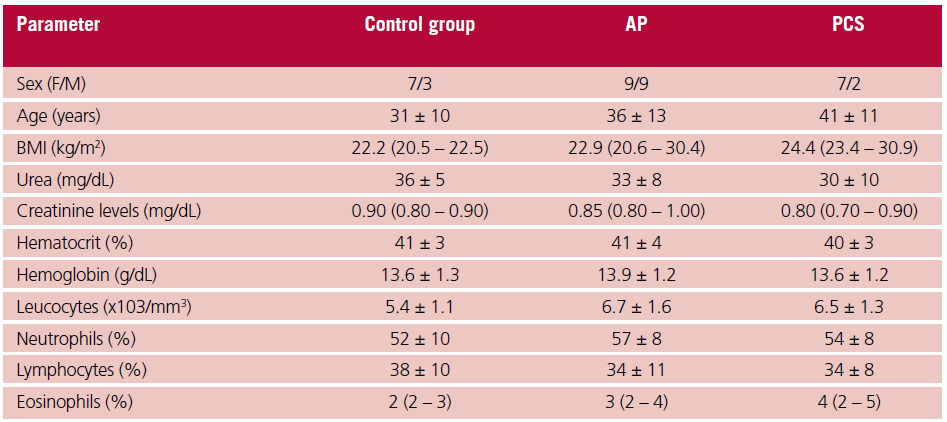
AP: asymptomatic patients; BMI: body mass index; F: female; M: male; PCS: post-COVID syndrome. Variables are expressed as mean ± SD, or median and interquartile range (Q1-Q3), according to their distribution.
Table 2. Determination of lipids, lipoproteins and apolipoproteins
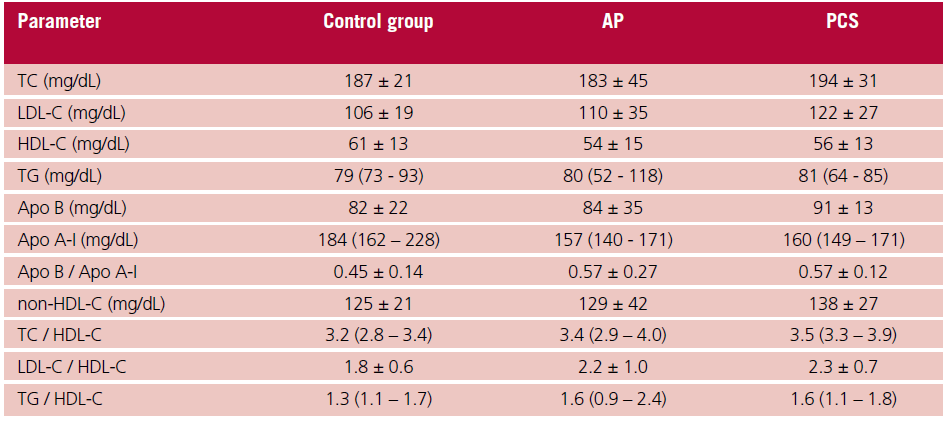
AP: asymptomatic patients; Apo: apolipoprotein; HDL-C: high-density lipoprotein cholesterol; LDL-C: lowdensity lipoprotein; PCS: post-COVID syndrome; TC: total cholesterol; TG: triglycerides. Variables are expressed as mean ± SD, or median and interquartile range (Q1-Q3), according to their distribution.
Table 3. Inflammatory mark- Parameter Control group AP PCS ers. Determination of lipids, lipoproteins and apolipoproteins
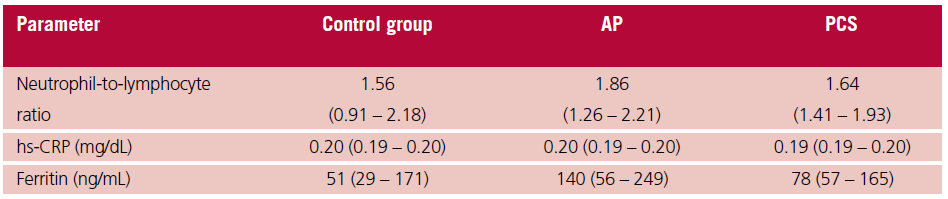
AP: asymptomatic patients; hs-CRP: high-sensitivity C-reactive protein; PCS: post-COVID syndrome. Variables are expressed as mean ± SD, or median and interquartile range (Q1-Q3), according to their distribution
Fig. 1. Evaluation of the three steps of reverse cholesterol transport. Panel A: cellular cholesterol efflux. Panel B: LCAT activity. Panel C: CETP activity

AP: asymptomatic patients; CETP: cholesteryl ester transfer protein; LCAT; lecithin-cholesterol acyltransferase; PCS: post-COVID syndrome.
Fig. 2. Paraoxonase 1 activity. Panel A: PON activity. Panel B: ARE activity.
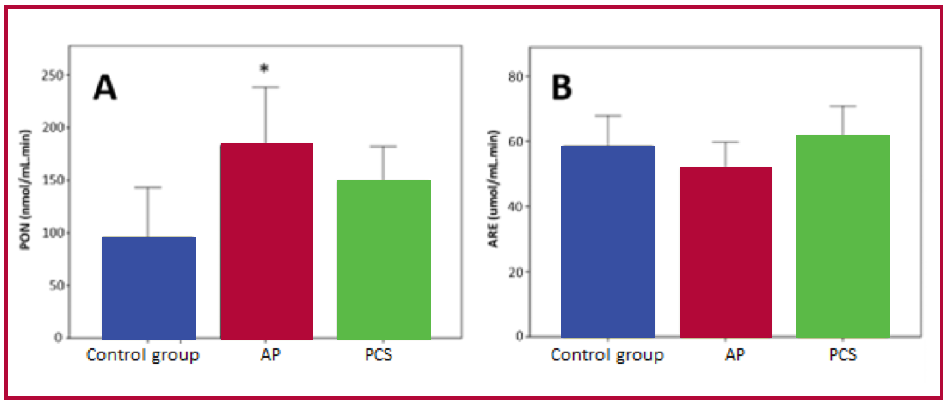
AP: asymptomatic patients; ARE: arylesterase; PCS: Post-COVID syndrome; PON: paraoxonase. *p=0.049 between AP and control group
CONCLUSIONS
Increased antioxidant activity of PON1 would represent a defensive mechanism against oxidative stress that only reaches statistical significance in the AP group. Consequently, the negative correlation between inflammatory markers and each step of reverse cholesterol transport indicates the potential for a detrimental effect of the inflammatory process on this crucial function of HDL. Our findings may explain, at least in part, the link between PCS and atherosclerosis.
Conflicts of interest
None declared.
(See conflicts of interest forms on the website).
Sources of funding
Project PIP N° 11220200102363 and project UBACyT N° 20020190100075BA.
https://creativecommons.org/licenses/by-nc-sa/4.0/

©Revista Argentina de Cardiología
REFERENCES
- https://www.who.int/es/news/item/27-04-2020-who-timeline-covid-19
- COVID-19 rapid guideline: managing the long-term effects of COVID-19. London: National Institute for Health and Care Excellence (NICE); 2020 Dec 18.
- Oronsky B, Larson C, Hammond TC, Oronsky A, Kesari S, Lybeck M, et al. A Review of Persistent Post-COVID Syndrome (PPCS). Clin Rev Allergy Immunol. 2023;64:66-74. https://doi.org/10.1007/s12016-021-08848-3
- Ayoubkhani D, Khunti K, Nafilyan V, Maddox T, Humberstone B, Diamond I, et al. Post-covid syndrome in individuals admitted to hospital with covid-19: retrospective cohort study. BMJ. 2021;372:n693. https://doi.org/10.1136/bmj.n693
- Davis HE, McCorkell L, Vogel JM, Topol EJ. Long COVID: major findings, mechanisms and recommendations. Nat Rev Microbiol. 2023;21:133-46. https://doi.org/10.1038/s41579-022-00846-2
- Beyerstedt S, Casaro EB, Rangel ÉB. COVID-19: angiotensin-converting enzyme 2 (ACE2) expression and tissue susceptibility to SARS-CoV-2 infection. Eur J Clin Microbiol Infect Dis. 2021;40:905- 19. https://doi.org/10.1007/s10096-020-04138-6
- Suhail S, Zajac J, Fossum C, Lowater H, McCracken C, Severson N, et al. Role of Oxidative Stress on SARS-CoV (SARS) and SARS-CoV-2 (COVID-19) Infection: A Review. Protein J. 2020;39:644-56. https://doi.org/10.1007/s10930-020-09935-8
- Contreras-Briceño F, Espinosa-Ramírez M, Rozenberg D, Reid WD. Eccentric Training in Pulmonary Rehabilitation of Post-COVID-19 Patients: An Alternative for Improving the Functional Capacity, Inflammation, and Oxidative Stress. Biology (Basel). 2022;11:1446. https://doi.org/10.3390/biology11101446
- Sorokin AV, Karathanasis SK, Yang ZH, Freeman L, Kotani K, Remaley AT. COVID-19-Associated dyslipidemia: Implications for mechanism of impaired resolution and novel therapeutic approaches. FASEB J. 2020;34:9843-53. https://doi.org/10.1096/fj.202001451
- Tang L, Yin Z, Hu Y, Mei H. Controlling Cytokine Storm Is Vital in COVID-19. Front Immunol. 2020;11:570993. https://doi.org/10.3389/fimmu.2020.570993.
- Siekacz K, Kumor-Kisielewska A, Miłkowska-Dymanowska J, Pietrusińska M, Bartczak K, Majewski S, et al. Oxidative Biomarkers Associated with the Pulmonary Manifestation of Post-COVID-19 Complications. J Clin Med. 2023;12:4253. https://doi.org/10.3390/jcm12134253
- Vollbracht C, Kraft K. Oxidative Stress and Hyper-Inflammation as Major Drivers of Severe COVID-19 and Long COVID: Implications for the Benefit of High-Dose Intravenous Vitamin C. Front Pharmacol. 2022;13:899198. https://doi.org/10.3389/fphar.2022.899198
- Jarrott B, Head R, Pringle KG, Lumbers ER, Martin JH. “LONG COVID”-A hypothesis for understanding the biological basis and pharmacological treatment strategy. Pharmacol Res Perspect. 2022;10:e00911. https://doi.org/10.1002/prp2.911
- Hu X, Chen D, Wu L, He G, Ye W. Declined serum high density lipoprotein cholesterol is associated with the severity of COVID-19 infection. Clin Chim Acta. 2020;510:105-10. https://doi.org/10.1016/j.cca.2020.07.015
- Wang G, Zhang Q, Zhao X, Dong H, Wu C, Wu F, et al. Low high-density lipoprotein level is correlated with the severity of COVID-19 patients: an observational study. Lipids Health Dis. 2020;19:204. https://doi.org/10.1186/s12944-020-01382-9
- Ding X, Zhang J, Liu L, Yuan X, Zang X, Lu F, et al. High-density lipoprotein cholesterol as a factor affecting virus clearance in covid-19 patients. Respir Med. 2020;175:106218. https://doi.org/10.1016/j.rmed.2020.106218
- Cho KH, Kim JR, Lee IC, Kwon HJ. Native High-Density Lipoproteins (HDL) with Higher Paraoxonase Exerts a Potent Antiviral Effect against SARS-CoV-2 (COVID-19), While Glycated HDL Lost the Antiviral Activity. Antioxidants (Basel). 2021;10:209. https://doi.org/10.3390/antiox10020209
- Brites F, Martin M, Guillas I, Kontush A. Antioxidative activity of high-density lipoprotein (HDL): Mechanistic insights into potential clinical benefit. BBA Clin. 2017;8:66-77. https://doi.org/10.1016/j.bbacli.2017.07.002
- Nurmohamed NS, Ditmarsch M, Kastelein JJP. Cholesteryl ester transfer protein inhibitors: from high-density lipoprotein cholesterol to low-density lipoprotein cholesterol lowering agents? Cardiovasc Res. 2022;118:2919-31. https://doi.org/10.1093/cvr/cvab350.
- Enright PL. The six-minute walk test. Respir Care 2003;48:783–5.
- Borg GA. Psychophysical bases of perceived exertion. Med Sci Sports Exerc 1982;14:377–81.
- Buonacera A, Stancanelli B, Colaci M, Malatino L. Neutrophil to Lymphocyte Ratio: An Emerging Marker of the Relationships between the Immune System and Diseases. Int J Mol Sci. 2022;23:3636. https://doi.org/10.3390/ijms23073636
- Furlong CE, Richter RJ, Seidel SL, Costa LG, Motulsky AG. Spectrophotometric assays for the enzymatic hydrolysis of the active metabolites of chlorpyrifos and parathion by plasma paraoxonase/arylesterase. Anal Biochem. 1989;180:242-7. https://doi.org/10.1016/0003-2697(89)90424-7
- Nevin DN, Zambon A, Furlong CE, Richter RJ, Humbert R, Hokanson JE, et al. Paraoxonase genotypes, lipoprotein lipase activity, and HDL. Arterioscler Thromb Vasc Biol. 1996;16:1243-9. https://doi.org/10.1161/01.atv.16.10.1243
- Martin M, Condori AI, Davico B, Gómez Rosso L, Gaete L, Tetzlaff W, et al. Impaired Reverse Cholesterol Transport is Associated with Changes in Fatty Acid Profile in Children and Adolescents with Abdominal Obesity. J Nutr. 2024;154:12-25. https://doi.org/10.1016/j.tjnut.2023.08.037
- Aguilar-Espinosa SL, Mendoza-Espinosa P, Delgado-Coello B, Mas-Oliva J. Lecithin cholesterol acyltransferase (LCAT) activity in the presence of Apo-AI-derived peptides exposed to disorder-order conformational transitions. Biochem Biophys Res Commun. 2013;441:469-75.
- Lagrost L, Gandjini H, Athias A, Guyard-Dangremont V, Lallemant C, Gambert P. Influence of plasma cholesteryl ester transfer activity on the LDL and HDL distribution profiles in normolipidemic subjects. Arterioscler Thromb. 1993;13:815-25. https://doi.org/10.1161/01.atv.13.6.815
- Morris G, Gevezova M, Sarafian V, Maes M. Redox regulation of the immune response. Cell Mol Immunol. 2022;19:1079-101. https://doi.org/10.1038/s41423-022-00902-0
- Otocka-Kmiecik A, Orłowska-Majdak M, Stawski R, Szkudlarek U, Kosielski P, Padula G, et al. Repetitions of Strenuous Exercise Consistently Increase Paraoxonase 1 Concentration and Activity in Plasma of Average-Trained Men. Oxid Med Cell Longev. 2021;2021:2775025. https://doi.org/10.1155/2021/2775025
- Brites F, Zago V, Verona J, Muzzio ML, Wikinski R, Schreier L. HDL capacity to inhibit LDL oxidation in well-trained triathletes. Life Sci. 2006;78:3074-81. https://doi.org/10.1016/j.lfs.2005.12.015
- Ghoreshi ZA, Abbasi-Jorjandi M, Asadikaram G, Sharif-Zak M, Seyedi F, Khaksari Haddad M, et al. Paraoxonase 1 rs662 polymorphism, its related variables, and COVID-19 intensity: Considering gender and post-COVID complications. Exp Biol Med (Maywood). 2023;248:2351-62. https://doi.org/10.1177/15353702221128563
- Tall AR, Yvan-Charvet L. Cholesterol, inflammation and innate immunity. Nat Rev Immunol. 2015;15:104-16. https://doi.org/10.1038/nri3793
- McGillicuddy FC, de la Llera Moya M, Hinkle CC, Joshi MR, Chiquoine EH, Billheimer JT, et al. Inflammation impairs reverse cholesterol transport in vivo. Circulation. 2009;119:1135-45. https://doi.org/10.1161/CIRCULATIONAHA.108.810721
- Martin M, Gaete L, Tetzlaff W, Ferraro F, Lozano Chiappe E, Botta EE, Osta V, et al. Vascular inflammation and impaired reverse cholesterol transport and lipid metabolism in obese children and adolescents. Nutr Metab Cardiovasc Dis. 2022;32:258-68. https://doi.org/10.1016/j.numecd.2021.09.025
- ATS Committee on Proficiency Standards for Clinical Pulmonary Function Laboratories. ATS statement: guidelines for the six-minute walk test. Am J Respir Crit Care Med. 2002;166:111-7. https://doi.org/10.1164/ajrccm.166.1.at1102
- Qorolli M, Beqaj S, Ibrahimi-Kaçuri D, Murtezani A, Krasniqi V, Mačak Hadžiomerović A. Functional status and quality of life in post-COVID-19 patients two to three weeks after hospitalization: A cross-sectional study. Health Sci Rep. 2023;6:e1510. https://doi.org/10.1002/hsr2.1510
- Talla A, Vasaikar SV, Szeto GL, Lemos MP, Czartoski JL, MacMillan H, et al. Persistent serum protein signatures define an inflammatory subcategory of long COVID. Nat Commun. 2023;14:3417. https://doi.org/10.1038/s41467-023-38682-4
- Lai YJ, Liu SH, Manachevakul S, Lee TA, Kuo CT, Bello D. Biomarkers in long COVID-19: A systematic review. Front Med (Lausanne). 2023;10:1085988. https://doi.org/10.3389/fmed.2023.1085988.