Alejandro Peirone1, MTSAC, FPICS, Alejandro Contreras1, MTSAC, Marcelo Cabrera1, Edgardo Banille1, Antonio Guevara1, Ignacio Juaneda1, MTSAC, Christian Kreutzer1, MTSAC, Carlos Pedra2, FPICS
1 Hospital Privado Universitario de Córdoba. University Biomedical Science Institute of Córdoba. Argentina.
2 Dante Pazzanese Institute of Cardiology. Secretariat of Public Health, San Pablo. Brazil.
Address for reprints: Alejandro Peirone. Valparaíso 4339 (5016) Córdoba, Argentina. E-mail: alepeirone@yahoo.com
Rev Argent Cardiol 2023;91:423-428. http://dx.doi.org/10.7775/rac.v91.i6.20713 http://dx.doi.org/10.7775/rac.es.v91.i6.20713
Financing: None.
This work received the Dr. Rodolfo Kreutzer Award at the 49th Argentine Congress of Cardiology.
ABSTRACT
Background: Surgical replacement using bioprostheses is a frequently used strategy to treat right-side heart valves dysfunction in congenital heart disease
Objectives: The aim of this study was to present the initial experience in Argentina with right-side bioprosthetic valve ring fracture/remodeling and subsequent transcatheter valve-in-valve replacement in patients with congenital heart disease.
Methods: This was a descriptive and observational study including all patients treated with transcatheter replacement due to bioprosthetic valve dysfunction.
Results: From August 2021 to May 2023, 5 patients (3 female) with right-side bioprosthetic dysfunction underwent transcatheter intervention as an alternative to de novo surgical replacement. Mean age was 21.2±9.20 years and mean weight 56.2±22.2 kg. Five balloon expandable valves were implanted: 3 in pulmonary position and 2 in tricuspid position, in 4 cases with previous fracture of the valve ring. In all cases, valve function was restored without complications. During follow-up, moderate right valve incompetence was observed in one case.
Conclusion: Transcatheter “valve-in-valve” replacement in dysfunctional right-side heart bioprostheses is an attractive and
safe strategy in selected cases, which restores competence and eliminates obstructions. This approach is a reasonable option
as alternative to de novo surgical valve replacement.
Key words: Heart defects - Congenital - Bioprosthesis - Valve dysfunction - Transcatheter replacement - Valve in valve
RESUMEN
Introducción: El reemplazo quirúrgico con colocación de bioprótesis es una estrategia utilizada frecuentemente para tratar valvulopatías en cardiopatías congénitas.
Objetivos: Presentar la experiencia inicial en Argentina con fractura/remodelación del anillo valvular en bioprótesis y posterior reemplazo percutáneo “válvula en válvula” en pacientes con cardiopatías congénitas.
Material y métodos: Estudio descriptivo y observacional. Se incluyeron todos los pacientes tratados con reemplazo percutáneo debido a disfunción de prótesis valvular biológica
Resultados: Desde agosto de 2021 hasta mayo de 2023, 5 pacientes (3 de sexo femenino) con disfunción de bioprótesis derechas recibieron tratamiento percutáneo como alternativa a la realización de un nuevo reemplazo quirúrgico. La edad media fue 21,2 ± 9,2 años, el peso medio fue 56,2 ± 22,2 kg. Fueron implantadas cinco válvulas balón expandibles: 3 en posición pulmonar y 2 en posición tricúspide y en 4 casos, con fractura previa del anillo valvular. En todos los pacientes se restauró la función valvular, sin complicaciones. En el seguimiento, en un sólo paciente se objetivó insuficiencia valvular moderada derecha
Conclusión: El reemplazo percutáneo “válvula en válvula” en bioprótesis disfuncionantes derechas es una estrategia atractiva y segura en casos seleccionados, que restaura la competencia y elimina las obstrucciones. Esta estrategia es una opción razonable como alternativa a la realización de un nuevo recambio valvular quirúrgico.
Palabras clave: Cardiopatías congénitas - Bioprótesis - Disfunción valvular - Reemplazo percutáneo - Válvula en válvula
Received: 11/03/2023
Accepted: 11/24/2023
INTRODUCTION
Bioprosthetic valves (BPV) with ring are commonly used for right-side surgical valve replacement in congenital heart disease. Satisfactory short and midterm clinical and hemodynamic outcomes have been shown, though frequently valve dysfunction develops during evolution. Data of BPV durability in tricuspid position are scarce. It is estimated that freedom from reoperation reaches 81% at 10 years post-implantation, though 42% presents echocardiographic valve dysfunction in the same time period. In addition, in patients requiring tricuspid valve implantation before 16 years of age, most bioprostheses are dysfunctional at 5 years post intervention, with valve regurgitation being the main cause in up to 89% of cases. (1)
In BPV in pulmonary position, freedom from de novo valve replacement and valve failure/dysfunction at 10 years is estimated at 51.7% and 20.2%, respectively. Young age, diagnosis of pulmonary atresia with ventricular septal defect and valve implantation without stent are among the de novo surgery risk factors. (2)
Transcatheter “valve-in-valve” technique is available for the treatment of valve dysfunction. However, it may reduce even more the internal BPV diameter, especially in prostheses of small caliber, which can potentially lead to functional stenosis or patient-prosthesis mismatch. (3)
The intentional fracture or remodeling of the BPV ring in pulmonary and tricuspid position is an attractive technique to achieve a larger internal diameter and better hemodynamic outcome in this strategy, avoiding patient-prosthesis mismatch. Published data on transcatheter “valve-in-valve” replacement (TVVR) in right-side valve bioprostheses for congenital heart disease are very limited.
The objective of the present study was to present the initial experience in Argentina with BPV ring fracture/remodeling and subsequent TVVR in patients with congenital heart disease.
METHODS
This was a descriptive, observational study including all patients treated with TVVR due to BPV dysfunction. The BPV had been previously implanted as part of the initial correction of a congenital heart disease. Valve dysfunction in pulmonary position was defined as peak pressure gradient ≥40 mmHg or the presence of at least moderate valve incompetence demonstrated by color Doppler echocardiography. Tricuspid valve dysfunction was defined as the presence of moderate to severe incompetence, and/or mean gradient ≥9 mmHg. (4)
Color Doppler echocardiograms before, during hospitalization/ discharge and during follow-up were performed by a single experimented cardiac physiologist (AC) to standardize the interpretation of the findings.
The valve ring rupture/remodeling strategy and subsequent “valve-in-valve” implantation was performed according to usual standardized techniques. (5)
The ultra-high pressure non-compliant balloon selected for the fracture was of the same size or up to 1.5 mm larger than the nominal diameter of the bioprosthesis to be fractured. The rupture pressure applied was as needed to achieve the objective and did not exceed 12 atmospheres (Figures 1 and 2). The diameter of the balloon-expandable valve to be implanted was selected between 0.5 and 2 mm larger than the nominal/commercial BPV diameter. All the patients received aspirin 100 mg/day indefinitely after valve implantation.
Statistical analysis
Data collection and statistical analysis was performed according to guidelines for reporting morbidity and mortality after cardiac valve interventions. (6)
Categorical variables are expressed as percentages and continuous variables as mean and standard deviation (SD) or median and interquartile range (IQR), as appropriate. The distribution of continuous variables was analyzed using the Kolmogorov-Smirnov test. SPSS 24 software package was used for the statistical analysis.
Ethical considerations
Intentional BPV fracture and subsequent balloon-expandable valve implantation with “valve-in-valve” technique as an alternative to de novo surgical replacement was approved by the institution’s ethical committee. All adult patients or their parents/tutors in case of underage patients signed a health information act before the procedure. This study was performed following the Declaration of Helsinki recommendations for observational studies reviewed in 1989.
RESULTS
From August 2021 to May 2023, 5 patients with rightside valve bioprosthetic dysfunction received TVVR as alternative to de novo surgical valve replacement. Mean age was 21±9.20 years, mean weight 56.2±22.2 kg and 60% of patients were female. Two patients had initial diagnosis of tetralogy of Fallot, two of Ebstein’s disease and the remaining patient idiopathic dilation of the pulmonary artery trunk with severe valve incompetence. All the patients had functional class (FC) III/IV symptoms of heart failure. In one case, BPV infective endocarditis had been diagnosed in tricuspid position 6 months before the percutaneous intervention.
Regarding valve dysfunction, three patients presented prosthetic dysfunction with predominant pulmonary valve stenosis, one with pure tricuspid stenosis and the remaining patient with double tricuspid anomaly (Table 1). Mean time between the surgical implant and the percutaneous intervention was 6.1±2.6 years.
During TVVR, four patients underwent intentional bioprosthesis ring fracture using ultra-high pressure non-compliant balloons [Atlas Gold ™ or True Balloon™ (Bard, USA)] with diameter equal to or up to 1.5 mm greater than the nominal/commercial size of the ring to be fractured and mean rupture pressure of 9.7±2.6 atmospheres. In all cases rupture was achieved during the first high pressure inflation and then balloon-expandable valves were implanted during the same intervention. In no case was concomitant transient pacemaker stimulation necessary during valve implantation. In the four patients who received intentional ring fracture, the final size of the implanted valve was 1.2 mm larger (0.5-2 mm) than the initial bioprosthesis nominal/commercial diameter. In all cases, the transvalvular gradients were resolved (peak pulmonary gradients decreased from 74 mmHg to 13.6 mmHg and mean tricuspid gradients were reduced from 11.2 mmHg to 3.2 mmHg) with absence of immediate post implant valve incompetence (Table 2). Mean radioscopy time was 19.3±10.3 minutes. Patients remained hospitalized for a median of 24 hours and mean follow-up time was 13.8±6.2 months. No immediate complications or during short or mid-term follow-up were recorded.
Follow-up color Doppler echocardiogram showed mean pulmonary gradient of 16±3.6 mmHg and tricuspid gradient of 6.6±1.2 mmHg. Only one patient developed moderate tricuspid valve incompetence. Clinically, improvement to FC I was observed in four patients and to FC II in the remaining case, and no deaths, valve explants, events of infective endocarditis or need for another reintervention were encountered.
Fig. 1.
(A) Angiography of Epic Supra™ (St Jude Medical, USA) dysfunctional bioprosthesis in pulmonary position in strict 90 °left lateral oblique projection. (B) Ultrahigh pressure non-compliant balloon to produce intentional valve ring fracture. (C) Fractured valve ring (arrow). . (D) Angiography in the same initial projection: Myval™ balloon-expandable valve (Meril, India) implanted with “valve-in-valve” technique in pulmonary position.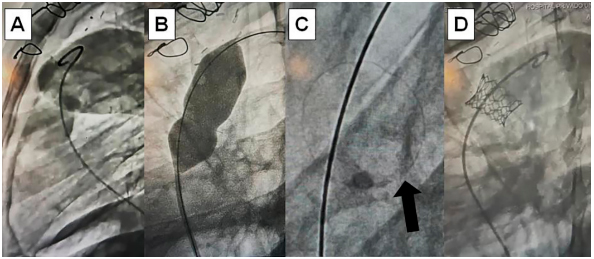
Fig. 2.
(A) Angiography of Epic Supra™ (St Jude Medical, USA) dysfunctional bioprosthesis in tricuspid position in 45 °right anterior oblique projection. (B) Ultra-high pressure non-compliant balloon to produce intentional valve ring fracture. (C) Fractured valve ring. (D) Angiography in the same initial projection: Myval™ balloon-expandable valve (Meril, India) implanted with “valve-in-valve” technique in tricuspid position.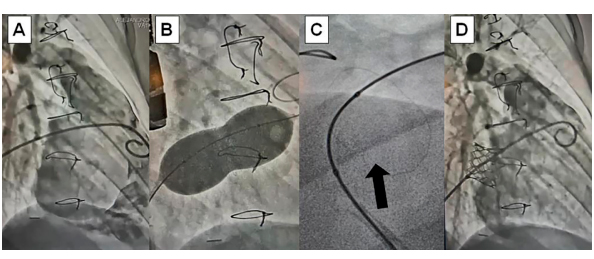
DISCUSSION
Surgical replacement of right-side dysfunctional valves (among them tetralogy of Fallot or Ebstein’s disease) with bioprosthesis implantation is a widely used strategy, based on its ample availability, lack of need for posterior anticoagulation and good results compared with mechanical prostheses. (7,8)
Most bioprostheses have been used off-label for surgical replacement in these positions since their initial development was for aortic valve replacement. These bioprostheses develop progressive impairment, manifested as stenosis and/or incompetence with pathological changes that include calcification, thickening, pannus formation, thrombosis, tears, and inflammation, potentially related to the occurrence of infective endocarditis. (9)
It is generally accepted that BPV remain stable up to 5 years post implantation. However, nearly 80% of patients require a reoperation, or will manifest significant dysfunction at 10 years after bioprosthesis placement. A small pulmonary bioprosthesis and age below 13 years were identified as risk factors for early reintervention. (10) Kwak et al. (11) reported that age under 20 years is associated to greater incidence of dysfunction compared with adult patients, even with larger bioprosthesis diameter. (12)
The TVVR technique is not new, as there is experience in left bioprosthesis dysfunction, mainly when the prevalent dysfunction is regurgitation. (13)
Conversely, in cases of pure valve stenosis, prosthesis dysfunction may coexist with patient-prosthesis mismatch, so it is logical to assume that transcatheter valve implantation might reduce even more the ring size and not solve valve narrowing. However, Allen and Johansen (3,14)
demonstrated that bioprostheses can be fractured using ultra-high pressure non-compliant balloons with diameter at least 1 mm larger than the nominal/ commercial implanted valve diameter, except with Trifecta (Abbott, USA) and Hancock II (Medtronic, USA) bioprostheses. This strategy allows “valvein- valve” implantation, decreasing the problem of patient-prosthesis mismatch. The rupture pressure required varied between 8 and 24 atmospheres and the ring fracture was followed by an immediate drop of pressure in the balloon and frequently an audible “crack”.
Recently reported results using this technique in BPV in tricuspid and pulmonary position, followed by TVVR, demonstrated a patent clinical improvement, a significant increase in the bioprosthesis internal diameter and better hemodynamic outcomes after transcatheter valve implantation. (15,16)
It is probable that to achieve a successful fracture, other variables beyond the bioprosthesis type and size should be considered in the bioprosthesis rupture/remodeling technique, including tissue remodeling inside and around the valve, as well as the geometry of the implant location. Nonetheless, we followed the recommendation about the use of ultra-high pressure non-compliant balloons, with a size at least 1mm larger than the true internal diameter, and up to 3-5 mm larger than the true inner diameter (3,15)
and with this strategy, we were able to intentionally fracture all the rings during the first inflation. The only patient of this cohort with a non-fracturable Hancock II bioprosthesis (Medtronic, USA), presented FC IV, a previous episode of infective endocarditis and small body surface area, which led us to consider that TVVR would not lead to patient- prosthesis-mismatch. It is pertinent to emphasize that the maximum final expansion of a fractured bioprosthesis is not clearly defined, as there are variables inherent to the valve and also to the surrounding tissue that affect the final diameter. (3,17)
It is also discussed whether stent implantation prior to TVVR is necessary to avoid fractured valve recoil, though observations suggesting differential forces contributing to this effect have not been clearly established. (18)
Therefore, we consider that balloon- expandable valve implantation with adequate radial force is enough to mitigate the possibility of subsequent recoil, thus avoiding the implantation of an additional stent. In parallel, it has also been suggested that the intentional ring fracture could be performed after TVVR, although this strategy presents the hypothetical risk that ultra-high-pressure balloons could damage the valve and/or distort the implanted percutaneous valve skeleton. (19)
Another point to highlight is the occurrence of infective endocarditis in patients with bioprostheses and TVVR. According to Cabalka et al. reports (20)
the relative risk is not greater compared with transcatheter valve replacement on a homograft or other types of conduits. However, this association deserves to be studied in depth, especially due to data published in the Society of Thoracic Surgeons registry, in which 12% of adult patients requiring surgical pulmonary valve replacement after percutaneous interventions had a history of infective endocarditis. (21)
In the previously mentioned multicenter study, (20)
the occurrence of infective endocarditis prior to TVVR was not associated with adverse events nor negatively impacted in the results during follow-up.
To conclude, we consider that the collaboration between cardiovascular surgeons and hemodynamic specialists to define the type of bioprosthesis to implant, and the long-term management is essential to achieve the success of this strategy.
Table. 1 Baseline patient characteristics
|
|
Age (years) |
Gender |
Weight (Kg) |
Disease |
Type of prosthetic dysfunction |
Transvalvular gradient |
Functional class |
|
|
#1T |
17 |
Male |
73 |
Ebstein |
Tricuspid stenosis |
11* |
III |
|
|
#2T |
16 |
Female |
36 |
Ebstein |
Tricuspid stenosis/ regurgitation |
11* |
IV |
|
|
#3P |
36 |
Male |
86 |
Idiopathic pulmonary dilation |
Pulmonary stenosis |
76** |
III |
|
|
#4P |
24 |
Female |
48 |
Tetralogy of Fallot |
Pulmonary stenosis |
67** |
III |
|
|
#5P |
13 |
Female |
38 |
Tetralogy of Fallot |
Pulmonary stenosis |
79** |
III |
|
P: Pulmonary; T: Tricuspid; *: Mean tricuspid transvalvular gradient; **: Maximum pulmonary transvalvular gradient
* and ** expressed in mmHg.
Table. 2 Transcatheter procedure and outcomes
|
|
Bioprosthesis |
Number |
High pressure fracture |
High pressure balloon |
Implanted valve |
Number |
Residual gradient |
Follow-up functional class |
Regurgitation |
|
|
#1T |
St Jude Epic |
25 |
Yes |
True Balloon 26 mm (12 atm) |
Myval |
26 |
1.4* |
I |
Moderate |
|
|
#2T |
Hancock |
25 |
No |
No |
Myval |
23 |
6* |
II |
No |
|
|
#3P |
St Jude Epic |
27 |
Yes |
Atlas Gold 26 mm (7 atm) |
Myval |
27.5 |
19** |
I |
No |
|
|
#4P |
St Jude Epic |
21 |
Yes |
Atlas Gold 22 mm (12 atm) |
Melody |
22 |
11** |
I |
No |
|
|
#5P |
St Jude Epic |
23 |
Yes |
True Balloon 24 mm (8 atm) |
Myval |
24.5 |
14** |
I |
No |
|
atm: atmospheres; P: Pulmonary; T: Tricuspid; *: Mean tricuspid transvalvular gradient; **: Maximum pulmonary transvalvular gradient
* and ** expressed in mm Hg.
Limitations
There are limitations in this study. The data were retrospectively collected from a single center with experience in transcatheter valve implantation, so the results presented might not be of general application. In addition, selection bias could have occurred in the incorporation of high-risk patients with dysfunctional bioprostheses. Finally, this is an initial and preliminary report of a novel procedure with valves approved off-label for use in the right ventricular inflow and outflow tract, with evident need of a longerterm follow-up.
CONCLUSIONS
Right-side valve TVVR in patients with congenital heart diseases is an attractive and safe strategy in selected cases, which restores competence and eliminates obstructions in dysfunctional bioprostheses. Intentional BPV fracture is an effective, technically safe and efficient intervention to increase the diameter of a valve to be implanted and is a reasonable alternative to de novo surgical valve replacement. A larger number of cases and more prolonged follow-up will be necessary to confirm these initial findings.
https://creativecommons.org/licenses/by-nc-sa/4.0/

Conflicts of interest
None declared. (See authors' conflict of interests forms on the web).
Financing
None.
©Revista Argentina de Cardiología
REFERENCES
- Burri M, Vogt MO, Hörer J, Cleuziou J, Kasnar-Samprec J, Kühn A, et al. Durability of bioprostheses for the tricuspid valve in patients with congenital heart disease. Eur J Cardiothorac Surg 2016;50:988- 93. https://doi.org/10.1093/ejcts/ezw094
- Lee C, Park C, Lee C, Kwak J, Kim S, Shim W, et al. Durability of bioprosthetic valves in the pulmonary position: Long-term follow-up of 181 implants in patients with congenital heart disease. J Thorac Cardiovasc Surg 2011;142:351-8. https://doi.org/10.1016/j.jtcvs.2010.12.020
- Allen K, Chhatriwalla A, Cohen D, Saxon J, Aggarwal S, Hart A, et al. Bioprosthetic valve fracture to facilitate transcatheter valvein-valve implantation. Ann Thorac Surg 2017;104:1501-8. https://doi.org/10.1016/j.athoracsur.2017.04.007
- Blauwet L, Danielson G, Burkhart H, Dearani J, Malouf J, Connolly H, et al. Comprehensive echocardiographic assessment of the hemodynamic parameters of 285 tricuspid valve bioprostheses early after implantation. J Am Soc Echocardiogr 2010;23:1045-59. https://doi.org/10.1016/j.echo.2010.08.007
- Allen K, Chhatriwalla A, Saxon J, Huded C, Sathananthan J, Nguyen T, et al. Bioprosthetic valve fracture: a practical guide. Ann Cardiothorac Surg 2021;10:564-70. https://doi.org/10.21037/acs-2021-tviv-25
- C, Miller D, Turina M, Kouchoukos N, Blackstone E, Grunkemeier G, et al. Guidelines for reporting mortality and morbidity after cardiac valve interventions. Ann Thorac Surg 2008;85:1490-5. https://doi.org/10.1016/j.athoracsur.2007.12.082
- Nomoto R, Sleeper L, Borisuk M, Bergerson L, Pigula FA, Emani S et al. Outcome and performance of bioprosthetic pulmonary valve replacement in patients with congenital heart disease. J Thorac Cardiovasc Surg 2016;152:1333-42. https://doi.org/10.1016/j.jtcvs.2016.06.064
- Liu P, Qiao WH, Sun FQ, Ruan XL, Al Shirbini M, Hu D, et al. Should a Mechanical or Biological Prosthesis Be Used for a Tricuspid Valve Replacement? A Meta-Analysis. J Card Surg 2016;31:294-302. https://doi.org/10.1111/jocs.12730
- Saleeb S, Newburger J, Geva T, Baird C, Gauvreau K, Padera R, et al. Accelerated degeneration of a bovine pericardial bioprosthetic aortic valve in children and young adults. Circulation 2014;130:51–60. https://doi.org/10.1161/CIRCULATIONAHA.114.009835
- McKenzie ED, Khan MS, Dietzman TW, Guzman-Pruneda FA, Samayoa AX, Liou A, et al. Surgical pulmonary valve replacement: a benchmark for outcomes comparisons. J Thorac Cardiovasc Surg 2014;148:1450-3. https://doi.org/10.1016/j.jtcvs.2014.02.060
- Kwak J, Lee C, Lee M, Lee C, Jang S, Lee S, et al. Does implantation of larger bioprosthetic pulmonary valves in young patients guarantee durability in adults? Durability analysis of stented bioprosthetic valves in the pulmonary position in patients with tetralogy of Fallot. Eur J Cardiothorac Surg 2016;49:1207-12. https://doi.org/10.1093/ejcts/ezv298
- Enezate T, Omran J, Bhatt DL. Percutaneous Versus Surgical Pulmonic Valve Implantation for Right Ventricular Outflow Tract Dysfunction. Cardiovasc Revasc Med 2019;20:553-8. https://doi.org/10.1016/j.carrev.2018.08.020
- Buchholz C, Mayr A, Purbojo A, Glöckler M, Toka O, Cesnjevar RA, et al. Performance of stented biological valves for right ventricular outflow tract reconstruction. Interact Cardiovasc Thorac Surg 2016;23:933-9. https://doi.org/10.1093/icvts/ivw264
- Johansen P, Engholt H, Tang M, Nybo RF, Rasmussen P, NielsenKudsk J. Fracturing mechanics before valve-in-valve therapy of small aortic bio- prosthetic heart valves. EuroIntervention. 2017;13:e1026– e1031. https://doi.org/10.4244/EIJ-D-17-00245
- . Shahanavaz S, Asnes J, Grohmann J, Qureshi, A, Rome J, Tanase D, et al. Intentional Fracture of Bioprosthetic Valve Frames in Patients Undergoing Valve-in-Valve Transcatheter Pulmonary Valve Replacement. Circ Cardiovasc Interv. 2018;11:e006453. https://doi.org/10.1161/CIRCINTERVENTIONS.118.006453
- McElhinney D, Cabalka A, Aboulhosn J, Eicken A, Boudjemline Y, Schubert S, et al. Transcatheter tricuspid valve-in-valve implantation for the treatment of dysfunctional surgical bioprosthetic valves. An International, multicenter registry study. Circulation 2016;133:1582-93. https://doi.org/10.1161/CIRCULATIONAHA.115.019353
- Tanase D, Georgiev S. Concept of fracturing a small-sized bioprothesis for further valve in valve implantation. Catheter Cardiovasc Interv 2022;99:E47. https://doi.org/10.1002/ccd.29126
- Wilson WM, Benson LN, Osten MD, Shah A, Horlick EM. Transcatheter Pulmonary Valve Replacement With the Edwards Sapien System: The Toronto Experience. JACC Cardiovasc Interv 2015;8:1819-27. https://doi.org/10.1016/j.jcin.2015.08.016
- Ziccardi MR, Groves EM. Bioprosthetic Valve Fracture for Valvein-Valve Transcatheter Aortic Valve Replacement: Rationale, Patient Selection, Technique, and Outcomes. Interv Cardiol Clin 2019;8:373- 82. https://doi.org/10.1016/j.iccl.2019.05.004
- Cabalka A, Asnes J, Balzer D, Cheatham J, Gillespie M, Jones T et al. Transcatheter pulmonary valve replacement using the Melody valve for treatment of dysfunctional surgical bioprostheses: A multicenter study. J Thorac Cardiovasc Surg 2018;155:1712-24. https://doi.org/10.1016/j.jtcvs.2017.10.143
- Khanna A, Hill K, Pasquali S, Wallace A, Masoudi F, Jacobs M et al. Benchmark outcomes for pulmonary valve replacement using the Society of Thoracic Surgeons databases. Ann Thorac Surg 2015;100:138-45. https://doi.org/10.1016/j.athoracsur.2015.03.025