Cristhian E. Scatularo1 MTSAC , Luciano Battioni2 MTSAC , Analía Guazzone1 , Guillermina Esperón3 , Luciana Corsico3 , Pablo Alcantara Costas4 , Hugo O. Grancelli1 MTSAC
1 Department of Cardiology, Sanatorio de la Trinidad Palermo, Buenos Aires, Argentina
2 Council on Heart Failure and Pulmonary Hypertension “Dr. Raúl Oliveri”,Argentine Society of Cardiology, Argentina
3 Department of Cardiology, Sanatorio Sagrado Corazón, Buenos Aires, Argentina
4 Department of Cardiology, Sanatorio de la Trinidad Quilmes, Buenos Aires, Argentina
Address for reprints: Cristhian E. Scatularo. E-mail: emmanuelscatularo@gmail.com - La Pampa 2940, CP 1428, CABA, Argentina
Rev Argent Cardiol 2023;91:379-388. http://dx.doi.org/10.7775/rac.v91.i6.20707
SEE RELATED ARTICLE : Rev Argent Car diol 2023:91:374-378. http://dx.doi.org/10.7775/rac.v91.i6.20705
ABSTRACT
Background: In patients with acute decompensated heart failure (ADHF), early evaluation of diuretic efficiency (DE) could predict diuretic response and clinical outcome.
Objectives: The aim of our study was to evaluate the association of DE with diuretic resistance (DR) in-hospital cardiovascular mortality, and readmission or cardiovascular mortality at 60 days in ADHF.
Methods: We conducted a multicenter and prospective study of patients hospitalized for ADHF. All patients received 40 mg of furosemide within two hours of admission and 20 mg every 8 hours in the first 24 hours. Subsequent adjustment of diuretic dose was left to the discretion of the investigator as determined by a pre-established protocol. Diuretic efficiency was defined as the ratio of net fluid balance and cumulative amount of furosemide within the first 24 hours. Diuretic resistance was defined as requirement of furosemide infusion ≥240 mg/day during the first 72 hours. The clinical and biochemical variables were evaluated. The primary outcome was a composite of in-hospital cardiovascular mortality, and cardiovascular mortality or readmissions for ADHF at 60 days.
Results: The cohort was made up of 157 patients; median age was 74 years and 56 % were men. Diuretic efficiency was –15 mL/ mg (interquartile range, IQR, –20 to –11). Diuretic resistance was evident in 13 % of patients, 8 % required sequential diuretic blockade, and 4 % required renal replacement therapy. Worsening renal function occurred in 22 % of patients. Cardiovascular mortality during hospitalization and at 60 days was 5.7 % and 6 %, respectively. Readmission rate for ADHF at 60 days was 12 %. Worse DE value was associated DR (p = 0.013), while patients in DE quartiles above -11 mL/mg were highly unlikely to develop DR (AUC 0.73, negative predictive value, NPV, 92.5 %). Worse DE value was associated with the CEP (p = 0.025), higher in-hospital cardiovascular mortality (p = 0.003), persistent congestion at 48 hours (p = 0.007), higher cumulative dose of furosemide at 72 hours (p = 0.001) worsening ADHF during hospitalization (p = 0.004).
Conclusion: Low initial DE was associated with DR, persistent congestion, and higher in-hospital cardiovascular mortality in ADHF and constitutes a useful parameter to detect those patients who could benefit from early intensive diuretic treatment.
Key words: Heart failure - Furosemide - Diuresis - Mortality
RESUMEN
Introducción: En pacientes con insuficiencia cardíaca aguda descompensada (ICAD) la eficiencia diurética (ED) evaluada en forma precoz podría predecir la respuesta a diuréticos y la evolución clínica.
Objetivos: Nuestro objetivo fue evaluar la asociación de la ED con la resistencia a diuréticos (RD), la mortalidad cardiovascular intrahospitalaria, y la mortalidad cardiovascular y las reinternaciones a 60 días en la ICAD.
Material y métodos: Estudio prospectivo y multicéntrico que incluyó pacientes internados por ICAD. Recibieron 40 mg de furosemida dentro de las 2 horas del ingreso y 20 mg cada 8 horas en las primeras 24 horas. El escalamiento diurético posterior quedó a criterio del investigador según un protocolo preestablecido. Se definió la ED como balance hídrico/dosis de furosemida en las primeras 24 horas y la RD como el requerimiento de infusión de furosemida ≥240 mg/día en las primeras 72 horas. Se evaluaron variables clínicas y bioquímicas, y el punto final combinado (PFC) de mortalidad cardiovascular intrahospitalaria, y mortalidad cardiovascular y reinternaciones por ICAD a 60 días.
Resultados: Se incluyeron 157 pacientes, mediana de edad de 74 años, 56 % hombres. La ED fue –15 mL/mg (rango intercuartílico, RIC, –20 a –11). Se evidenció la RD en el 13 % de los pacientes, el 8 % requirió bloqueo tubular y el 4 % terapia de reemplazo renal. El 22 % desarrolló empeoramiento de la función renal. La mortalidad cardiovascular intrahospitalaria fue del 5,7 % y en el seguimiento a 60 días, del 6 %. Las reinternaciones por ICAD a 60 días fueron del 12 %. Una peor ED se asoció al desarrollo de RD (p = 0,013) y los pacientes con ED superior a –11 mL/mg tuvieron mayor probabilidad de no desarrollar RD (área bajo la curva, AUC, 0,73; valor predictivo negativo, VPN, 92,5 %). Una peor ED se asoció al PFC (p = 0,025), mayor mortalidad cardiovascular intrahospitalaria (p = 0,003), persistencia de congestión a 48 horas (p = 0,007), mayor dosis de furosemida a 72 horas (p = 0,001) y empeoramiento de la ICAD en la internación (p = 0,004).
Conclusión: La ED inicial baja se asoció a la RD, la dificultad en la descongestión y una mayor mortalidad cardiovascular intrahospitalaria en ICAD. Es un parámetro útil para detectar pacientes que podrían beneficiarse de un tratamiento diurético intensivo precoz.
Palabras clave: Insuficiencia cardíaca - Furosemida - Diuresis - Mortalidad
Received: 11/03/2023
Accepted: 11/25/2023
INTRODUCTION
Acute decompensated heart failure (ADHF) is usually defined as the new onset or worsening of symptoms or signs of congestion or systemic hypoperfusion, requiring early diagnosis and timely treatment. (1,2)
Intravenous loop diuretics are the cornerstone to achieve proper decongestion and clinical stability in this condition. Effective response to these drugs is necessary for in-hospital progress and during the vulnerable phase. (3-5)
Identifying patients with diuretic resistance (DR) is clinically relevant because it is associated with higher mortality and readmission risk. However, there is currently no standard definition of DR during hospitalization for ADHF (6-10)
Patients with DR may benefit from alternative decongestive treatments in addition to intravenous furosemide, making early detection crucial. (11-17)
The urinary output achieved, and the absolute dose of diuretics have been classically considered to be variables related to the response to decongestive treatment, DR, and prognosis, (6-10)
However, these individual factors could be inadequate surrogates of the therapeutic effectiveness since there are multiple associated confounding factors, such as the degree of congestion, disease severity and the subjective nature of the treating physician. (6-10)
Diuretic efficiency (DE), considered as the urinary output or weight loss achieved divided by the amount of intravenous diuretics administered, is a parameter that has shown a clear association with diuretic response and the clinical course in patients with ADHF. (18-20)
Therefore, measuring DE, defined as the ratio of the net fluid balance during the first 24 hours and the dose of furosemide given, could be an objective parameter to help identify patients with DR who are likely to have a worse clinical outcome. This can facilitate the implementation of more effective decongestion strategies.
The aim of this study was to evaluate the usefulness of DE within the initial 24 hours in ADHF patients as a predictor of DR, in-hospital outcome, and readmission rate due to decompensation or cardiovascular mortality at 2 month-follow up, considered the vulnerable phase.
METHODS
Study design and objective
We conducted a prospective, multicenter, and open study including consecutive patients > 18 years admitted to the coronary care unit (CCU) due to ADHF. Initial DE was defined as the ratio of net fluid balance and cumulative amount of intravenous furosemide within the first 24 hours. The initial DE of the patients included was calculated, and we also evaluated the clinical, biochemical, and echocardiographic variables on admission and at 72 hours, and the occurrence of clinical events during hospitalization and at 60 days. The variables evaluated are defined in the Appendix.
Population
- Inclusion criteria: patients > 18 years hospitalized in the CCU due to ADHF,
- Exclusion criteria: creatinine (Cr) levels ≥ 2.5 mg/dL or on dialysis on admission, systolic blood pressure < 90 mm Hg, cardiogenic or septic shock, requirement of pressor or inotropic agents, mechanical ventilation (MV) or pregnancy. Those patients who did not sign the informed consent form or who were enrolled in other clinical trials with pharmacological intervention were also excluded.
Protocol of diuretic treatment
The strategy of diuretic treatment during the first 72 hours of hospitalization in the CCU was determined by a pre-established protocol (Figure 1). After bladder evacuation, an initial intravenous bolus of 40 mg of furosemide was given to patients within two hours of admission to the emergency department. In the first 24 hours after admission, all patients received intravenous boluses of 20 mg of furosemide every 8 hours. After the first day of hospitalization, the daily furosemide dose was adjusted (increased or decreased) every 24 hours, with evaluation of the extent of clinical congestion or daily diuresis (fluid balance threshold of -2000 mL/day), according to the pre-established protocol and to the discretion of the treating physician. According to the protocol, up-titration of furosemide was performed by increasing the intravenous infusion. Down-titration of furosemide in the protocol allowed for shifting from intravenous to oral administration upon achieving an adequate response. The diuretic protocol was discontinued 72 hours after enrollment or earlier in case of requirement of doses of furosemide ≥240 mg/day, tubular diuretic blockade (TDB), hypertonic saline (HS), renal replacement therapy (RRT), inotropic drugs, pressor agents, MV , or in the event of a significant clinical complication as determined by the investigator.
Fig. 1.Strategy of diuretic dose adjustment during the first 72 hours of hospitalization as determined per protocol according to the evaluation of the treating physician.
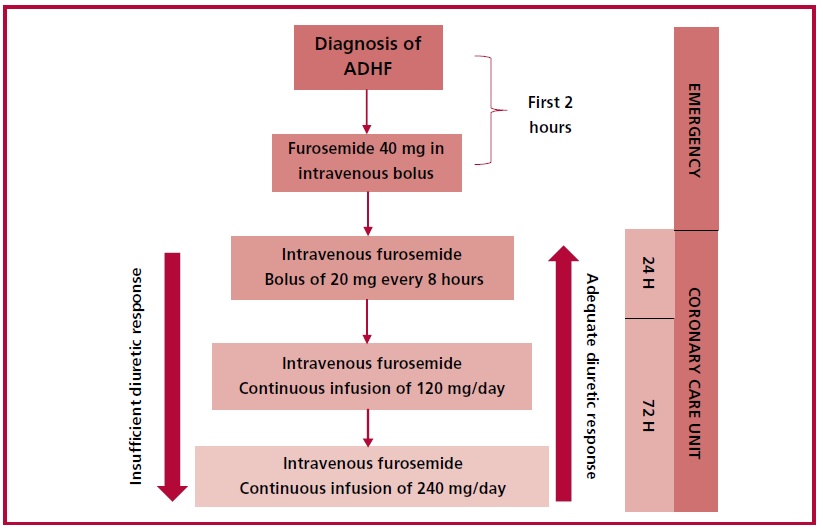
Abbreviations: ADHF (acute decompensated heart failure)
Outcome measures
- Diuretic resistance (DR): requirement of furosemide ≥240 mg/day during the first 72 hours of hospitalization.
- Composite end point: in-hospital mortality, readmissions for ADHF at 60 days or cardiovascular mortality at 60 days.
- Secondary end points:
- Cardiovascular mortality during hospitalization
- Cardiovascular mortality at 60 days.
- Readmissions for ADHF at 60 days.
- Persistent clinical congestion at 48 hours after inclusion.
- Cumulative furosemide dose required at 72 hours after inclusion.
- Weight change at 72 hours after inclusion.
- Worsening renal function (WRF) at 72 hours after inclusion.
- Change in N-terminal pro B-type natriuretic peptide (NT-proBNP) levels at 72 hours after inclusion.
- Change in E/e' ratio and pulmonary artery systolic pressure (PASP) at 72 hours after inclusion.
- Requirement of TDB with > 1 diuretic, HS or RRT during hospitalization.
- Worsening ADHF during hospitalization
- Requirement of inotropic drugs or MV during hospitalization.
- Length of CCU stay
Statistical analysis
Continuous variables were expressed as mean and standard deviation (SD), or median and interquartile range (IQR), according to their distribution. Categorical variables were expressed as absolute and relative frequencies. Continuous variables were compared using the Student's t test or the Mann-Whitney U test, according to their distribution. Proportions were analyzed using the chi-square test or the Fisher's exact test, as applicable according to the frequency of the values expected. The association of the initial DE with the end points was analyzed, with adjustment for predetermined variables: age, chronic kidney disease (CKD), hypoalbuminemia, ambulatory use of furosemide, left ventricular ejection fraction (LVEF), and sepsis. In addition, the distribution of the endpoints was evaluated according to the calculated quartiles of the initial DE. A ROC curve was constructed to evaluate the ability of DE to predict DR, and the sensitivity and specificity of the quartile with the worst DE value (25th percentile) were evaluated. The alpha error was set at 5% to establish statistical significance. All the statistical calculations were performed using SPSS 21.0 software package.
Data collection, ethical aspects, and sources of funding
The clinical and biochemical variables and the relevant images were collected. Patient recruitment started once the protocol was approved by the Committee on Ethics of the Argentine Society of Cardiology. All the subjects gave their informed consent to be included in the study. Data were recorded in a printed form customized for the study, and then incorporated into a single electronic database by the principal investigator. Data confidentiality was maintained in compliance with the Personal Data Protection and Patient Rights Act, ensuring that the identification of each individual participant is not possible. None of the authors have disclosed any conflicts of interest, and this study was not funded.
Ethical considerations
The protocol was evaluated and approved by the Committee on Ethics of the Argentine Society of Cardiology through the PRIISA.BA platform of the Ministry of Health of the City of Buenos Aires. Since this survey was anonymous and self-administered, participants were not required to give informed consent. The survey was conducted following national and international ethical standards for research on human subjects, as the Declaration of Helsinki revised in 2013, National Ministry of Health resolution 1480/2011, law N° 3301 of the city of Buenos Aires, and ANMAT regulation 6677/10 and amendments 4008 and 4009. Data privacy among respondents was protected through anonymity in the electronic survey.
RESULTS
A total of 157 patients were included; median age was 74 years (IQR 62-83), and 56 % were men. Diabetes was present in 36 %, atrial fibrillation in 43 % (n = 68) and CKD in 20 % (n = 31). The etiologies were ischemic heart disease in 27 % (n = 43), valvular heart disease in 22 % (n = 35) and hypertension in 15 % (n = 24). LVEF was reduced in 43 % (n = 67), mildly reduced in 12 % (n = 19), and preserved in 45 % (n = 71). The remaining baseline characteristics of the population are described in Table 1.
The initial DE was -15 mL/mg (RIC -20 to -11) and there were no differences in the DE across the entire spectrum of LVEF (p=0.8) or between the different clinical types of ADHF (p=0.3). Diuretic resistance was evident in 13 % (n= 20) of the cohort, 8 % (n= 13) required TDB, 1 patient required HS and 4 % (n= 6) required RRT. Worsening ADHF occurred in 13 % (n = 20) and WRF in 22 % (n = 35), while 10 % (n = 16) required pressor or inotropic drugs and 4 % (n = 6) required MV during hospitalization. Length of stay in the CCU was 5 days (IQR 4-8). Inhospital cardiovascular mortality was 5.7 % (n = 9) and cardiovascular mortality during the 60-day follow- up was 6.4 % (n = 10). Twelve percent (n = 19) of the patients were readmitted for ADHF at 60 days.
Worse DE predicted the development of DR (OR 1.072; 95 % CI 1.015-1.130; p= 0.013), and was associated with a trend towards higher use of SDB (p = 0.07) and RRT (p = 0.06) adjusted for predetermined variables (age, CKD, hypoalbuminemia, ambulatory use of furosemide, LVEF and sepsis) (Figure 2). The distribution of the end points by DE quartiles is described in Table 2. The ROC curve analysis indicated that patients with DE quartiles above -11 mL/mg are highly unlikely to develop DR, with an AUC of 0.73 (95% CI 0.59-0.87, p=0.001), sensitivity of 60 %, specificity of 80 %, negative predictive value of 92.5 % and positive predictive value of 30 % (Figure 3).
Worse DE value was associated with the composite endpoint, mainly due to higher in-hospital cardiovascular mortality (OR 1.20; 95 % CI 1.06-1.36; p=0.003), with no significant differences in the rate of cardiovascular mortality or readmissions for ADHF at 60-day follow-up.
Worse DE was associated with persistent congestion at 48 hours (OR 1.10; 95 % CI 1.03-1.18; p = 0.007), higher total cumulative dose of furosemide administered at 72 hours (p = 0.001), lower weight loss at 72 hours (p = 0.0001) and worsening of ADHF during hospitalization (OR 1.11; 95 % CI 1.03-2; p = 0.006). Similarly, worse DE was associated with a trend toward a lesser percentage decrease in NTproBNP at 72 hours after inclusion (p = 0.09), with no differences in the reduction of the E/e' ratio and PASP. Patients with the worst DE value presented greater requirement of pressor agents (OR 1.18; 95 % CI 1.04-1.33; p = 0.008) and greater but not significant requirement of inotropic agents (OR 1.05; 95 % CI 0.98-1.13; p = 0.12) during hospitalization, with no differences in the rate of WRF, MV and length of stay in the CCU (Figure 2).
Patients in the lowest quartile of DE value (less than -11 ml/mg) were at a higher risk of developing DR and had a worse prognosis during hospitalization. Furthermore, decongestion was less effective, and a more complex diuretic strategy was required for these patients compared to those in higher DE value quartiles (Table 2).
Table 1. Baseline characteristics of the population.
|
Variable |
|
|
Age
(years), median (IQR) |
74 (62 - 83) |
|
Male sex, n (%) |
88 (56 %) |
|
Hypertension, n (%) |
121 (77 %) |
|
Diabetes, n (%) |
57 (36 %) |
|
Previous atrial fibrillation, n
(%) |
68 (43 %) |
|
Chronic kidney
failure, n (%) |
31 (20 %) |
|
Etiology
of heart failure, n (%) Ischemic heart disease Valvular heart disease Hypertension
Others |
43 (27 %) 35 (22 %) 24 (15 %) 55 (35 %) |
|
Functional
class (NYHA), n (%) I II III
IV |
53 (34 %) 84 (54 %) 16 (10 %) 4 (2 %) |
|
Previous
heart failure treatment, n (%) Beta blockers ACEI/ARB ARNI Aldosterone antagonists iSGLT-2 Oral furosemide Previous dose of furosemide (mg), median
(IQR) |
104 (66 %) 85 (54 %) 11 (7 %) 49 (31 %) 4 (2.5 %) 59 (38 %) 40 (20-40) |
|
Clinical
presentation of heart failure, n (%) Pulmonary congestion Acute pulmonary edema Right ventricular-predominant heart
failure |
92 (58 %) 28 (18 %) 37 (24 %) |
|
NT-proBNP
on admission (pg/mL), median (IQR) |
3939 (2200-6300) |
|
Ejection
fraction (%), median (IQR) Preserved (LVEF ≥50 %), n (%) Mildly reduced (LVEF between 41 % and
49 %), n (%) Reduced (LVEF ≤40 %), n (%) |
48 (30-59) 71 (45 %) 19 (12 %) 67 (43 %) |
Abbreviations: ARB: Angiotensin II receptor blocker; ACEI: angiotensin-converting enzyme inhibitor; ARNI: angiotensin receptor neprilysin inhibitor; iSGLT-2: sodium-glucose co-transporter 2 inhibitor; IQR: interquartile range; LVEF: left ventricular ejection fraction; NYHA: New York Heart Association
DISCUSSION
In this study, measuring DE within the initial 24 hours proved effective in assessing diuretic responsiveness to furosemide in ADHF. It also helped identify patients with DR who will exhibit reduced clinical decongestion and will require a higher dose of furosemide in the first 72 hours. Additionally, the initial DE predicted the development of events during the entire hospitalization, mainly worsening ADHF, use of vasoactive drugs, and in-hospital mortality.
Discrimination of events by quartiles of DE revealed that a DE value better than -11 mL/mg was associated with a low probability of developing DR. Moreover, patients in the quartile with worse values suffered from more complications and required more intense diuretic therapy. The calculation of DE within the initial 24 hours of hospitalization can prove valuable in daily practice to identify patients who are at greater risk of developing DR and may benefit from a more aggressive diuretic treatment that could improve their clinical outcome. This potential clinical usefulness should be evaluated through future randomized studies.
While there is evidence to suggest that urinary output is adequate for evaluating the response to diuretic therapy, it should be noted that it depends on the dosage of furosemide administered, fluid intake, and the level of congestion. It has been reported that the overall amount of furosemide given to patients is a predictor of their clinical outcome during hospitalization. (6-10)
However, this may be influenced by the bias resulting from the severity of ADHF and the criteria of the treating physician. Therefore, relying solely on either urinary output or cumulative diuretic dose would not completely measure the patient's inherent reaction to diuretic treatment. Currently, measuring natriuresis soon after furosemide administration has emerged as a useful parameter for evaluating diuretic response. Although this approach facilitates dynamic optimization of treatment, serial measurement is required, and the method may not be available in all the centers. (21-28)
In our cohort, we excluded more severely ill patients, such as those with Cr ≥ 2.5 mg %, cardiogenic or septic shock, and those requiring MV. These patients have multiple factors that can interfere with the diuretic response and the clinical course, necessitating higher diuretic doses. Thus, our study indicates that low initial DE is associated with adverse events even in ADHF patients with milder clinical presentations. Despite the association of CKD, hypoalbuminemia, and previous use of oral furosemide with DR, (6-10)
it is important to note that the initial DE remains an independent predictor of both DR and in-hospital mortality, even after adjusting for these variables.
Previous experiences have shown that DE is associated with an increased risk of readmission for ADHF and mortality within 60 days of discharge from hospitalization for ADHF. This is a critical period when the risk of clinical events is elevated. (18-20)
However, in the current cohort, this association could not be established, likely due to the low rate of events during the vulnerable stage. Notably, the exclusion of patients with the highest clinical severity by protocol may have contributed to this finding.
In this analysis, we found no correlation between the initial DE and the occurrence of WRF, as indicated by an increase in serum Cr levels or a reduction in the estimated glomerular filtration rate. This finding may be because WRF included patients with persistent congestion despite increasing diuretic doses and poor prognosis (true WRF or acute renal injury), alongside those with clinical improvement during diuretic treatment and a favorable course (pseudo WRF). (29-35)
However, the diuretic response (DE) is not exclusively determined by renal function. It also relies on left and right ventricular function, volemia, and several other factors that affect the pharmacokinetics and pharmacodynamics of diuretics in ADHF. (1-5)
Our study demonstrated that patients with better DE had a greater reduction of NT-proBNP within the initial 72 hours of hospitalization. This finding is consistent with multiple analyses indicating that NT-proBNP levels decrease during decongestive treatment, and this is associated with a better prognosis during hospitalization and the vulnerable stage. (1,2,36,37)
However, to date, there is no evidence to support therapeutic guidance through serial measuring of this biomarker. (1,2,36,37)
There was no correlation found in this cohort between DE and improvement in the dynamic measurement of the E/e' ratio, PASP, and inferior vena cava diameter through echocardiography despite the evidence supporting the efficacy of this method in identifying residual congestion and optimizing diuretic treatment. (38-41)
There are some limitations to this study. We could not measure intra-abdominal pressure or central venous pressure, which are variables related to the development of DR. (6-10)
The presence of ultrasound lung comets was not examined during the periodic evaluation of congestion, despite being a proven useful tool in the diagnosis and therapeutic management of congestion. (42-43)
At present, we do not haved longer-term follow-up, in which there could be an association of initial DE with clinical events as demonstrated in previous experiences. (18-20)
Table 2. Distribution of end points by initial diuretic efficiency quartiles.
|
|
DE Quartile 1 (> -20 mL/mg) N = 46 |
DE Quartile 2 (-20 to -15.1
mL/mg) N = 39 |
DE Quartile 3 (-15 to -11 mL/mg) N = 35 |
DE Quartile 4 (< -11 mL/mg) N = 37 |
|
CEP, n (%) |
8 (17.4 %) |
7 (18.4 %) |
6 (17 %) |
14 (38 %)* |
|
In-hospital CV mortality, n (%) |
0 (0 %) |
3 (7.7 %) |
0 (0 %) |
6 (16.2 %)* |
|
CV mortality at 60 days, n (%) |
5 (10.9 %) |
1 (2.9 %) |
1 (2.9 %) |
3 (9.7 %) |
|
Readmissions for ADHF at 60 days, n (%) |
5 (10.9 %) |
3 (7.9 %) |
5 (14.3 %) |
6 (16.2 %) |
|
Diuretic resistance (furosemide ≥240 mg/day), n (%) |
2 (4.3 %) |
2 (5.1 %) |
5 (14.3 %) |
11 (29.7 %)* |
|
Persistent congestion at 48 h, n (%) |
3 (7 %) |
3 (7 %) |
4 (11 %) |
13 (35 %)* |
|
Cumulative furosemide dose at 72 hours (mg), median (IQR) |
176 (60-1120) |
206 (60-1740) |
275 (60-1740) |
524 (60-3000)* |
|
Weight loss at 72 h (kg) |
–5 (–7 to –3) |
–4 (–5 to –2) |
–3 (–5 to –2) |
–3 ( –4 yo –2)* |
|
Sequential diuretic blockade, n (%) |
2 (4.3 %) |
3 (7.7 %) |
1 (3 %) |
7 (19 %)* |
|
Hypertonic saline, n (%) |
0 (0 %) |
0 (0 %) |
0 (0 %) |
1 (2.7 %) |
|
Renal replacement therapy, n (%) |
1 (2.2 %) |
0 (0 %) |
1 (3 %) |
4 (11 %)* |
|
Worsening ADHF, n (%) |
2 (4.3 %) |
5 (12.8 %) |
3 (8.6 %) |
10 (27 %)* |
|
Percent NT-proBNP reduction, median (IQR) |
56 (73-36) |
53 (62-31) |
49 (70-40) |
41 (61-7)* |
|
Length of stay in the CCU (days), median (IQR) |
6.3 (2-21) |
7.3 (2-24) |
6 (3-23) |
10.6 (3-86)* |
Abbreviations: ADHF: acute decompensated heart failure; CCU: coronary care unit; CV: cardiovascular; DE: diuretic efficiency; CEP: composite end point (in-hospital CV mortality, CV mortality and readmissions at 60 days); IQR: interquartile range. Quartile 1 is the quartile with the best DE and quartile 4 is the quartile with the worst DE * p value < 0.05 between quartile 4 and the rest of the cohort
Fig. 2.Graphical summary: diuretic protocol used and results of the study.
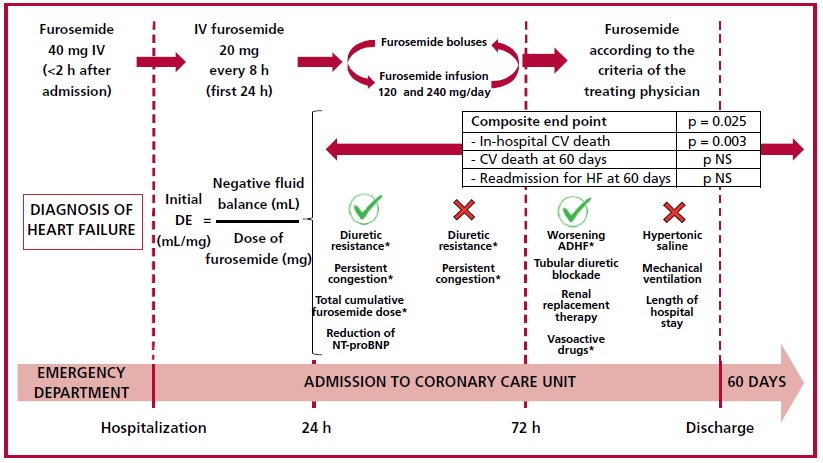
Abbreviations: ADHF: acute decompensated heart failure; CV: cardiovascular; DE: diuretic efficiency; IV: intravenous; NS: non-significant; PASP: pulmonary artery systolic pressure *p value < 0.05
Fig. 3.Discrimination ability of diuretic efficiency to predict the development of diuretic resistance according to the analysis of the ROC curve.
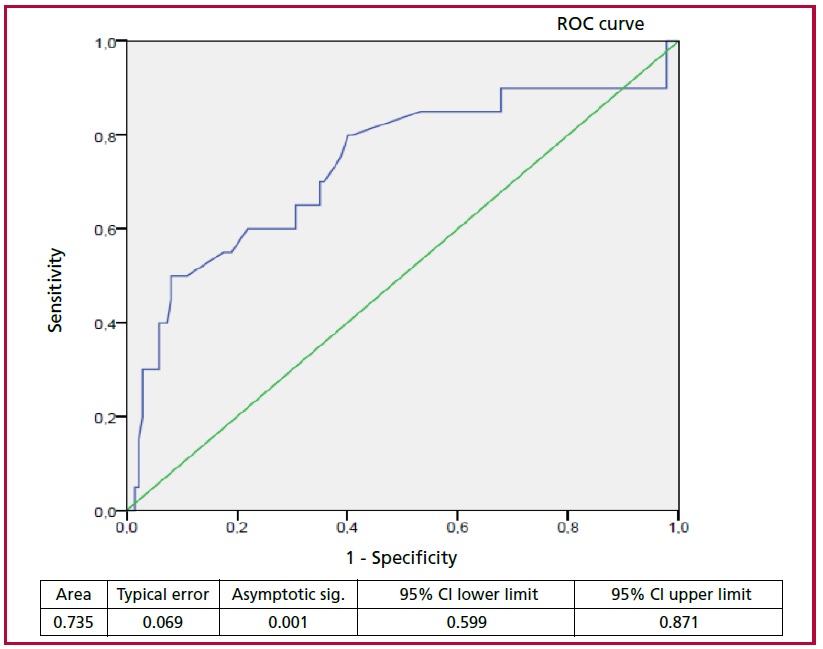
Abbreviations: CI: confidence interval; ROC: receiver operating characteristic; ROC curve (sensitivity vs. 1-specificity)
CONCLUSION
In patients hospitalized for ADHF, initial DE was associated with the development of DR, persistent congestion, higher cumulative dose of furosemide and higher in-hospital cardiovascular mortality. Diuretic efficiency is a useful parameter to detect those patients who could benefit from early intensive diuretic treatment and thus achieve better clinical outcome.
https://creativecommons.org/licenses/by-nc-sa/4.0/

Conflicts of interest
None declared. (See authors' conflict of interests forms on the web).
Source of funding
None.
©Revista Argentina de Cardiología
REFERENCES
- McDonagh TA, Metra M, Adamo M, Gardner RS, Baumbach A, Böhm M, et al. ESC Scientific Document Group. 2021 ESC Guidelines for the diagnosis and treatment of acute and chronic heart failure. Eur Heart J. 2021;42:3599-726. https://doi.org/10.1093/eurheartj/ehab368
- Heidenreich PA, Bozkurt B, Aguilar D, Allen LA, Byun JJ, Colvin MM, et al. 2022 AHA/ACC/HFSA Guideline for the Management of Heart Failure: A Report of the American College of Cardiology/ American Heart Association Joint Committee on Clinical Practice Guidelines. J Am Coll Cardiol. 2022;79:e263-e421. https://doi.org/10.1016/j.jacc.2021.12.012
- Mebazaa A, Yilmaz MB, Levy P, Ponikowski P, Peacock WF, Laribi S, et al. Recommendations on pre-hospital and early hospital management of acute heart failure: a consensus paper from the Heart Failure Association of the European Society of Cardiology, the European Society of Emergency Medicine and the Society of Academic Emergency Medicine--short version. Eur Heart J. 2015;36:1958-66. https://doi.org/10.1093/eurheartj/ehv066
- Felker GM, Ellison DH, Mullens W, Cox ZL, Testani JM. Diuretic Therapy for Patients With Heart Failure: JACC State-of-the-Art Review. J Am Coll Cardiol. 2020;75:1178-95. https://doi.org/10.1016/j.jacc.2019.12.059
- Mullens W, Damman K, Harjola VP, Mebazaa A, Brunner-La Rocca HP, Martens P, et al. The use of diuretics in heart failure with congestion - a position statement from the Heart Failure Association of the European Society of Cardiology. Eur J Heart Fail. 2019;21:137-55. https://doi.org/10.1002/ejhf.1369
- Yilmaz MB, Gayat E, Salem R, Lassus J, Nikolaou M, Laribi S, et al. Impact of diuretic dosing on mortality in acute heart failure using a propensity-matched analysis. Eur J Heart Fail. 2011;13:1244-52. https://doi.org/10.1093/eurjhf/hfr121
- Peacock WF, Costanzo MR, De Marco T, Lopatin M, Wynne J, Mills RM, et al. ADHERE Scientific Advisory Committee and Investigators. Impact of intravenous loop diuretics on outcomes of patients hospitalized with acute decompensated heart failure: insights from the ADHERE registry. Cardiology. 2009;113:12-9. https://doi.org/10.1159/000164149
- Hasselblad V, Gattis Stough W, Shah MR, Lokhnygina Y, O'Connor CM, Califf RM, et al. Relation between dose of loop diuretics and outcomes in a heart failure population: results of the ESCAPE trial. Eur J Heart Fail. 2007;9:1064-9. https://doi.org/10.1016/j.ejheart.2007.07.011
- Valente MA, Voors AA, Damman K, Van Veldhuisen DJ, Massie BM, O'Connor CM, et al. Diuretic response in acute heart failure: clinical characteristics and prognostic significance. Eur Heart J. 2014;35:1284-93. https://doi.org/10.1093/eurheartj/ehu065
- Neuberg GW, Miller AB, O'Connor CM, Belkin RN, Carson PE, Cropp AB, et al. PRAISE Investigators. Prospective Randomized Amlodipine Survival Evaluation. Diuretic resistance predicts mortality in patients with advanced heart failure. Am Heart J. 2002;144:31-8. https://doi.org/10.1067/mhj.2002.123144
- Damman K, Beusekamp JC, Boorsma EM, Swart HP, Smilde TDJ, Elvan A, et al. Randomized, double-blind, placebo-controlled, multicentre pilot study on the effects of empagliflozin on clinical outcomes in patients with acute decompensated heart failure (EMPARESPONSE- AHF). Eur J Heart Fail. 2020;22:713-22. https://doi.org/10.1002/ejhf.1713
- Schulze PC, Bogoviku J, Westphal J, Aftanski P, Haertel F, Grund S, et al. Effects of Early Empagliflozin Initiation on Diuresis and Kidney Function in Patients With Acute Decompensated Heart Failure (EMPAG-HF). Circulation. 2022;146:289-98. https://doi.org/10.1161/CIRCULATIONAHA.122.059038
- Yeoh SE, Osmanska J, Petrie MC, Brooksbank KJM, Clark AL, Docherty KF, et al. Dapagliflozin vs. metolazone in heart failure resistant to loop diuretics. Eur Heart J. 2023 Aug 14;44(31):2966-2977. https://doi.org/10.1093/eurheartj/ehad341
- Trullàs JC, Morales-Rull JL, Casado J, Carrera-Izquierdo M, Sánchez-Marteles M, Conde-Martel A, et al. CLOROTIC trial investigators. Combining loop with thiazide diuretics for decompensated heart failure: the CLOROTIC trial. Eur Heart J. 2023;44:411-21. https://doi.org/10.1093/eurheartj/ehac689
- Mullens W, Dauw J, Martens P, Verbrugge FH, Nijst P, Meekers E, et al. ADVOR Study Group. Acetazolamide in Acute Decompensated Heart Failure with Volume Overload. N Engl J Med. 2022;387:1185- 95. https://doi.org/10.1056/NEJMoa2203094
- Griffin M, Soufer A, Goljo E, Colna M, Rao VS, Jeon S, et al. Real World Use of Hypertonic Saline in Refractory Acute Decompensated Heart Failure: A U.S. Center's Experience. JACC Heart Fail. 2020;8:199-208. https://doi.org/10.1016/j.jchf.2019.10.012
- Wang MJ, Zheng YM, Jin HX. Ultrafiltration for patients with acute decompensated heart failure: A systematic review and meta-analysis. Medicine (Baltimore). 2021;100:e28029. https://doi.org/10.1097/MD.0000000000028029
- Testani JM, Brisco MA, Turner JM, Spatz ES, Bellumkonda L, Parikh CR, et al. Loop diuretic efficiency: a metric of diuretic responsiveness with prognostic importance in acute decompensated heart failure. Circ Heart Fail. 2014;7:261-70. https://doi.org/10.1161/CIRCHEARTFAILURE.113.000895
- Palazzuoli A, Testani JM, Ruocco G, Pellegrini M, Ronco C, Nuti R. Different diuretic dose and response in acute decompensated heart failure: Clinical characteristics and prognostic significance. Int J Cardiol. 2016;224:213-9. https://doi.org/10.1016/j.ijcard.2016.09.005
- Kiernan MS, Stevens SR, Tang WHW, Butler J, Anstrom KJ, Birati EY, et al. NHLBI Heart Failure Clinical Trials Network Investigators. Determinants of Diuretic Responsiveness and Associated Outcomes During Acute Heart Failure Hospitalization: An Analysis From the NHLBI Heart Failure Network Clinical Trials. J Card Fail. 2018;24:428-38. https://doi.org/10.1016/j.cardfail.2018.02.002
- Hodson DZ, Griffin M, Mahoney D, Raghavendra P, Ahmad T, Turner J, et al. Natriuretic Response Is Highly Variable and Associated With 6-Month Survival: Insights From the ROSE-AHF Trial. JACC Heart Fail. 2019;7:383-91. https://doi.org/10.1016/j.jchf.2019.01.007
- Biegus J, Zymliński R, Sokolski M, Todd J, Cotter G, Metra M, et al. Serial assessment of spot urine sodium predicts effectiveness of decongestion and outcome in patients with acute heart failure. Eur J Heart Fail. 2019;21:624-33. https://doi.org/10.1002/ejhf.1428
- Honda S, Nagai T, Nishimura K, Nakai M, Honda Y, Nakano H, et al. NaDEF investigators. Long-term prognostic significance of urinary sodium concentration in patients with acute heart failure. Int J Cardiol. 2018;254:189-94. https://doi.org/10.1016/j.ijcard.2017.08.053
- Brinkley DM Jr, Burpee LJ, Chaudhry SP, Smallwood JA, Lindenfeld J, Lakdawala NK, et al. Spot Urine Sodium as Triage for Effective Diuretic Infusion in an Ambulatory Heart Failure Unit. J Card Fail. 2018;24:349-54. https://doi.org/10.1016/j.cardfail.2018.01.009
- Collins SP, Jenkins CA, Baughman A, Miller KF, Storrow AB, Han JH, et al. Early urine electrolyte patterns in patients with acute heart failure. ESC Heart Fail. 2019;6:80-8. https://doi.org/10.1002/ehf2.12368
- Cobo-Marcos M, Zegri-Reiriz I, Remior-Perez P, Garcia-Gomez S, Garcia-Rodriguez D, Dominguez-Rodriguez F, et al. Usefulness of natriuresis to predict in-hospital diuretic resistance. Am J Cardiovasc Dis. 2020;10:350-5.
- Luk A, Groarke JD, Desai AS, Mahmood SS, Gopal DM, Joyce E, et al. First spot urine sodium after initial diuretic identifies patients at high risk for adverse outcome after heart failure hospitalization. Am Heart J. 2018;203:95-100. https://doi.org/10.1016/j.ahj.2018.01.013
- Singh D, Shrestha K, Testani JM, Verbrugge FH, Dupont M, Mullens W, et al. Insufficient natriuretic response to continuous intravenous furosemide is associated with poor long-term outcomes in acute decompensated heart failure. J Card Fail. 2014;20:392-9. https://doi.org/10.1016/j.cardfail.2014.03.006
- Mullens W, Damman K, Testani JM, Martens P, Mueller C, Lassus J, et al. Evaluation of kidney function throughout the heart failure trajectory - a position statement from the Heart Failure Association of the European Society of Cardiology. Eur J Heart Fail. 2020;22:584- 603. https://doi.org/10.1002/ejhf.1697
- Llauger L, Jacob J, Miró Ò. Renal function and acute heart failure outcome. Med Clin (Barc). 2018;151:281-90. English, Spanish. https://doi.org/10.1016/j.medcli.2018.05.010
- Damman K, Valente MA, Voors AA, O'Connor CM, van Veldhuisen DJ, Hillege HL. Renal impairment, worsening renal function, and outcome in patients with heart failure: an updated meta-analysis. Eur Heart J. 2014;35:455-69. https://doi.org/10.1093/eurheartj/eht386
- Ahmad T, Jackson K, Rao VS, Tang WHW, Brisco-Bacik MA, Chen HH, et al. Worsening Renal Function in Patients With Acute Heart Failure Undergoing Aggressive Diuresis Is Not Associated With Tubular Injury. Circulation. 2018;137:2016-28. https://doi.org/10.1161/CIRCULATIONAHA.117.030112
- McCallum W, Tighiouart H, Testani JM, Griffin M, Konstam MA, Udelson JE, et al. Acute Kidney Function Declines in the Context of Decongestion in Acute Decompensated Heart Failure. JACC Heart Fail. 2020;8:537-47. https://doi.org/10.1016/j.jchf.2020.03.009
- Fudim M, Loungani R, Doerfler SM, Coles A, Greene SJ, Cooper LB, et al. Worsening renal function during decongestion among patients hospitalized for heart failure: Findings from the Evaluation Study of Congestive Heart Failure and Pulmonary Artery Catheterization Effectiveness (ESCAPE) trial. Am Heart J. 2018;204:163-73. https://doi.org/10.1016/j.ahj.2018.07.019
- Berra G, Garin N, Stirnemann J, Jannot AS, Martin PY, Perrier A, et al. Outcome in acute heart failure: prognostic value of acutekidney injury and worsening renal function. J Card Fail. 2015;21:382- 90. https://doi.org/10.1016/j.cardfail.2014.12.015
- Januzzi JL Jr, Maisel AS, Silver M, Xue Y, DeFilippi C. Natriuretic peptide testing for predicting adverse events following heart failure hospitalization. Congest Heart Fail. 2012;18 Suppl 1:S9-S13. https://doi.org/10.1111/j.1751-7133.2012.00306.x
- Bhardwaj A, Januzzi JL Jr. Natriuretic peptide-guided management of acutely destabilized heart failure: rationale and treatment algorithm. Crit Pathw Cardiol. 2009;8:146-50. https://doi.org/10.1097/HPC.0b013e3181c4a0c6
- Price S, Platz E, Cullen L, Tavazzi G, Christ M, Cowie MR, et al. Expert consensus document: Echocardiography and lung ultrasonography for the assessment and management of acute heart failure. Nat Rev Cardiol. 2017 Jul;14(7):427-40
- Öhman J, Harjola VP, Karjalainen P, Lassus J. Focused echocardiography and lung ultrasound protocol for guiding treatment in acute heart failure. ESC Heart Fail. 2018;5:120-28. https://doi.org/10.1002/ehf2.12208
- Arvig MD, Laursen CB, Jacobsen N, Gæde PH, Lassen AT. Monitoring patients with acute dyspnea with serial point-of-care ultrasound of the inferior vena cava (IVC) and the lungs (LUS): a systematic review. J Ultrasound. 2022;25:547-61. https://doi.org/10.1007/s40477-021-00622-7
- Öhman J, Harjola VP, Karjalainen P, Lassus J. Assessment of early treatment response by rapid cardiothoracic ultrasound in acute heart failure: Cardiac filling pressures, pulmonary congestion and mortality. Eur Heart J Acute Cardiovasc Care. 2018;7:311-20. https://doi.org/10.1177/2048872617708974
- Gargani L, Pang PS, Frassi F, Miglioranza MH, Dini FL, Landi P, et al. Persistent pulmonary congestion before discharge predicts rehospitalization in heart failure: a lung ultrasound study. Cardiovasc Ultrasound. 2015;13:40. https://doi.org/10.1186/s12947-015-0033-4
- Pang PS, Russell FM, Ehrman R, Ferre R, Gargani L, Levy PD, et al. Lung Ultrasound-Guided Emergency Department Management of Acute Heart Failure (BLUSHED-AHF): A Randomized Controlled Pilot Trial. JACC Heart Fail. 2021;9:638-48. https://doi.org/10.1016/j.jchf.2021.05.008
- Rhodes A, Evans LE, Alhazzani W, Levy MM, Antonelli M, Ferrer R, et al. Surviving Sepsis Campaign: International Guidelines for Management of Sepsis and Septic Shock: 2016. Crit Care Med. 2017;45:486-552. https://doi.org/10.1097/CCM.0000000000002255