Acute heart failure (AHF) is one of the leading causes of hospitalization worldwide and is associated with high post- discharge morbidity and mortality. (1) Despite therapeutic advances, persistent signs of congestion at discharge are common and remain the most important and modifiable predictor of early rehospitalization, adverse events, and mortality. (2,3) This congestion, often subclinical, is frequently unnoticed on traditional physical examination, underscoring the need for more sensitive and reproducible assessment tools.
Traditional assessment methods used to determine discharge timing, such as changes in weight or physical examination findings, have proven insufficient to ensure adequate decongestion, as reflected by the high rates of rehospitalization within 30 days after discharge. (4) In clinical practice, the perception of "clinical stability" does not always equate to true decongestion, reinforcing the need to incorporate objective and reproducible measurements.
Historically, clinical assessment has been the cornerstone of therapeutic decision- making. However, multiple studies have demonstrated its limited sensitivity and prognostic accuracy. Subclinical congestion at hospital discharge may be present in up to 40% of patients, even among those considered clinically "compensated." (5) This dissociation between clinical findings and actual congestion led to the recognition of the concept of subclinical congestion and called into question the reliability of physical examination as the sole criterion for defining hospital discharge.
Achieving adequate decongestion is the central therapeutic goal during hospitalization for acute heart failure. Beyond symptomatic relief, achieving euvolemia provides the physiological basis necessary to tolerate and optimize prognostic-modifying therapies. However, defining and quantifying congestion remains challenging, as it may present as intravascular congestion, tissue congestion, or both, each with distinct diagnostic and therapeutic profile. (6)
In light of this limitation, complementary assessment tools have been developed. Biomarkers such as NT-proBNP (N-terminal pro-B-type natriuretic peptide), while useful for diagnosis and prognosis, have limitations as therapeutic guidelines during the acute phase. Clinical trials such as GUIDE-IT and PRIMA II demonstrated that a treatment strategy guided exclusively by NT-proBNP levels did not reduce mortality or rehospitalizations. (7,8) Other biomarkers—such as CA-125 (cancer antigen 125), sST2 (soluble suppression of tumorigenicity 2), and bio-ADM (biologically active adrenomedullin)—have been proposed as dynamic markers of volume overload, particularly in the outpatient setting; however, their clinical application remains limited. (9,10)
In recent years, the focus has shifted toward noninvasive tools. Lung ultrasound (LUS) enables the detection of B-lines indicative of pulmonary congestion with greater sensitivity than chest radiography. (11) Complementarily, inferior vena cava (IVC) ultrasound and Doppler assessment of hepatic, portal, and renal veins using the Venous Excess Ultrasound (VExUS) protocol allow objective evaluation of systemic intravascular congestion. (12-14) More recently, remote dielectric sensing (ReDS) has shown promising results for guiding decongestion management in hospitalized patients in a clinical trial. (15) These accessible and reproducible methods have demonstrated prognostic value across multiple cohorts.
In this context, our group conducted the CAVAL US-AHF (CAVA and Lung Ultrasound- guided Therapy in Acute Heart Failure) study, (16,17) a randomized, single-center clinical trial conducted in Argentina that enrolled patients hospitalized for AHF. All patients underwent IVC and LUS assessments according to protocol at admission, during hospitalization, and at discharge. They were performed by trained physicians who were not involved in therapeutic decision-making. The images were analyzed and reported to the treating physicians by an external core lab blinded to the treatment assignment and patient identity. Congestion was classified into three categories (A, B, or C) according to ultrasound findings previously reported in the literature to be associated with an increased risk of hospitalization or mortality after discharge. Patients were randomly assigned and stratified by age and left ventricular ejection fraction into two groups: (a) a control group receiving standard care based on clinical, symptoms, and laboratory findings, and (b) an intervention group (CAVAL US) receiving standard care plus protocol-guided therapeutic adjustment based on IVC + LUS findings. The intervention algorithm included clinical assessment, the number of B-lines (assessed in 8 zones), and IVC parameters (diameter and collapsibility). Physicians in the intervention group had access to the ultrasound findings, whereas those in the control group remained blinded.
Prior to hospital discharge, a systematic checklist was implemented to ensure adherence to guideline-directed medical therapy, vaccination, patient education, and follow-up planning. All patients received structured counseling, including educational material and a written drug therapy plan. In both groups, outpatient follow- up visits were scheduled 7-10 days after discharge and at least monthly thereafter.
The primary endpoint of subclinical congestion at discharge (>5 B lines and/or dilated IVC) was observed in 13.3% of the CAVAL US group versus 66.6% in the control group (p< 0.001) , representing an 80% relative reduction. Similarly, the composite endpoint at 90 days (readmission, unplanned visit, or death) was significantly reduced (13.3% vs. 36.7% p = 0.038 - This intervention was safe and not associated with a longer hospital stay.
Recently, the Heart Failure Association (HFA) of the European Society of Cardiology (ESC) proposed a multiparametric algorithm to assess decongestion at discharge, which includes three criteria: < 5 B-lines on US, IVC diameter <21 mm with collapsibility >50% and >30% reduction in NT-proBNP compared to admission. (3) These criteria were established based on recent prognostic studies and expert consensus. On this basis, we conducted a substudy recently published in the European Journal of Heart Failure. (18). According to the HFA- ESC criteria, patients were classified into two groups: optimal decongestion (IVC diameter <21 mm and collapsibility >50%, <5 B-lines on 8-zone LUS, and >30% reduction in NT-proBNP) and suboptimal decongestion (failure to meet any of the three criteria). The primary endpoint was a composite of HF rehospitalization, urgent visit due to worsening symptoms, or death at 90 days after discharge. Forty- five percent met all three optimal decongestion criteria. None experienced events during the 90- day follow-up, whereas 42.4% of patients with suboptimal decongestion reached the primary endpoint (p<0.001). No significant differences were observed between groups in furosemide dose at discharge, left ventricular ejection fraction, or use of guideline- directed medical therapy, suggesting that residual congestion may persist regardless of clinical assessment or treatment intensity. This analysis provides prospective evidence supporting the prognostic value of the HFA-ESC multiparametric algorithm and reinforces the applicability of an objective and guided decongestion strategy in routine clinical practice.
The incorporation of complementary tools does not represent unnecessary sophistication but rather an evolution toward more precise and reproducible medicine. Subclinical congestion is common, and its persistence after discharge remains one of the main determinants of adverse events. In this context, the integration of inferior vena cava and lung ultrasound with accessible biomarkers offers an objective, feasible, and high- impact clinical strategy, particularly in resource- limited health care systems.
Effective decongestion is not achieved with diuretics alone. Sodium retention reflects neurohormonal and inflammatory activation, which requires early and comprehensive initiation of guideline- directed prognosis- modifying therapy-angiotensin receptor- neprilysin inhibitors (ARNIs), beta-blockers, mineralocorticoid receptor antagonists (MRAs), and sodium-glucose linked transporter 2 (SGLT2) inhibitors. (19) The STRONG- HF trial demonstrated that intensive up- titration of ARNIs, beta- blockers, and MRAs within the first weeks after discharge was not only safe but also significantly reduced mortality and hospitalizations. (20) In a subanalysis of this trial, intensive titration of neurohormonal blockade was associated with more efficient and sustained decongestion at 90 days, together with a significant reduction in the risk of the primary endpoint. (21)
Guided decongestion therapy in acute heart failure must be precise, comprehensive, and reproducible. The integration of multiple objective tools combined with clinical criteria offers diagnostic synergy to achieve this goal. As illustrated in Figure 1, each of these pillars provides complementary information for a multiparametric assessment of congestion. Table 1 presents a clinical decalogue that summarizes the principles of this emerging practice integrating both evidence and clinical experience.
Fig. 1.
Assessment of Decongestion
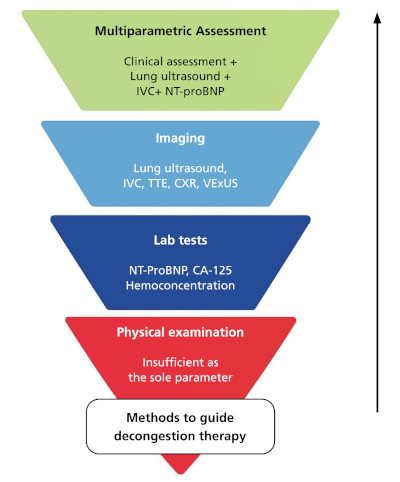
63TOWARDS OPTIMAL DECONGESTIVE THERAPY / Lucrecia M. Burgos et alCA-125: cancer antigen 125; CXR: chest X-ray; IVC: inferior vena cava;NT-proBNP:N-terminal pro-B-type natriuretic peptide; TTE, transthoracic echo-cardiography
Table 1.
Decalogue of Congestion in Acute Heart Failure
| 1. Congestion is the key predictor of post-discharge events. |
| 2. Subclinical congestion is common and associated with an increased risk of adverse events. |
| 3. Physical examination alone is insufficient. |
| 4. Congestion assessment should be multiparametric. |
| 5. The HFA-ESC algorithm provides an objective framework. |
| 6. Diuretics are necessary but not sufficient. |
| 7. Guideline-directed medical therapy (GDMT) is an essential component of decongestion. |
| 8. Decongestion is not equivalent to simply "removing water." |
| 9. The vulnerable phase represents a critical window of opportunity. |
| 10. Validating decongestion strategies in our setting is both feasible and necessary. |
Achieving complete decongestion before hospital discharge is a decisive step toward reducing rehospitalizations and improving clinical outcomes. Residual congestion at discharge remains one of the strongest predictors of mortality and adverse events; therefore, its detection should be incorporated into the standard of care. However, the optimal assessment of decongestion and the appropriate strategies to guide treatment are not yet fully defined or standardized.
Recent literature indicates that no single parameter is sufficient. Therefore, a multiparametric assessment combining clinical, ultrasound, and laboratory tools - all of which are available, reproducible, and relatively low cost- is recommended. In health care systems such as those in Latin America, where pressure for early discharge is common and resources are often limited, the integration of lung ultrasound, inferior vena cava assessment, and biomarkers represents an objective, safe, and feasible strategy capable of improving decision-making and clinical outcomes.
In this context, the CAVAL US-AHF study and its substudy demonstrate that simple and effective evidence-based care models can be implemented to redefine how we assess congestion and determine the discharge timing. The true paradigm shift lies in incorporating this comprehensive approach into the standard of care. Moving from "treating decompensation" to "ensuring complete decongestion" before discharge represents a transition toward more precise, reproducible, and outcome- oriented medicine.
Conflicts of interest
None declared. (See authors' conflict of interests forms on the web/Additional material).
