Coronary Artery Disease in Post-COVID Multisystem Inflammatory Syndrome Case Report
Multisystem inflammatory syndrome in children (MIS-C) associated with coronavirus disease 2019 is defined by the presence of persistent fever, inflammation, and organ dysfunction, with evidence of past or recent SARS-CoV-2 infection and exclusion of any other microbial cause. It involves overlapping with other inflammatory diseases (Kawasaki disease and toxic shock syndrome). (1)
The Argentine Society of Pediatrics divides the clinical occurrence of this syndrome into two phenotypes: (a) complete or incomplete Kawasaki-like disease (KD), and (b) unspecific occurrence, in children with shock and/or fever and gastrointestinal, respiratory, or neurological symptoms who fail to meet the criteria for KD. (2)
Cardiovascular involvement incidence has been high in several international reports. Shock occurred in 43%, myocardial compromise with elevated enzymes in 31% to 50%, and echo-assessed left ventricular dysfunction in 42%. Coronary artery abnormalities were experienced in 18.5% of cases. (1,2,3)
The standard of care is support therapy while specific therapy consists of immunomodulator agents. For both subtypes, intravenous immunoglobulin (IV Ig) is the first-line treatment. Second-line treatment is systemic corticosteroids; for patients with the KD phenotype aged under 12 months or with coronary artery disease, the first and second lines are co-administered. The third line is biological agents. (1,5,6)
Our goal is to introduce a clinical case of coronary artery dilation due to post-COVID-19 multisystem inflammatory syndrome from Hospital del Niño Jesús, in the Argentine province of Tucumán.
A previously healthy 1-year-old patient, with 8.6-kg weight and 77-cm length, whose uncle (mother’s brother) was COVID-19 positive 14 days earlier, presented with congested upper airways and one-week bilateral conjunctival injection that did not respond to symptom therapy. There was also fever, urticariform rash over the trunk, vomiting and diarrhea, with poor oral tolerance. The patient was taken to the Emergency Room and hospitalized as a result of moderate dehydration.
Laboratory tests were requested; systemic inflammatory reaction was observed, and C reactive protein (CRP) and immunoglobulin (IgG and IgM) were negative for SARS-CoV-2. The echo-Doppler showed 5-mm pericardial effusion with no coronary artery disease and with good ventricular function. The patient also experienced cheilitis, raspberry tongue, left submandibular adenomegaly, bilateral palpebral edema, lower and upper limb edema with no Godet’s sign. Rash was extensive to the face and abdomen, and thus considered to be Kawasaki-like syndrome. Gamma globulin plus methylprednisolone, and acetylsalicylic acid (ASA) were administered at anti-inflammatory doses with good response. After 72 hours with no fever following the end of treatment, the patient was discharged under corticosteroids and ASA treatment.
Within 24 hours from discharge, the patient had fever, poor general condition, raspberry tongue, bilateral palpebral edema, and conjunctivitis. The patient was hospitalized. Laboratory tests showed inflammation and positive SARS-CoV 2 IgG, and a repeat echo-Doppler (Figure 1) showed aneurysmal dilation in both coronary arteries with preserved ventricular function (Table 1 outlines coronary values with their corresponding Z scores upon successive echocardiography monitoring). The condition was reassessed as MIS-C. The patient received immune-mediated treatment and platelet aggregation inhibitors with gamma globulin (2 g/kg/dose), methylprednisolone (10 mg/kg/dose, 3 pulses), ASA 5 mg/kg/day. The patient showed mild clinical and analytical improvement. Heart monitoring after 5 days revealed similar characteristics. The patient was classified as treatment-resistant, and infliximab was prescribed (5 mg/kg). He showed good progression, with significant clinical and cardiovascular improvement, and was discharged after 16 days. Cardiologic and rheumatologic workup plus platelet aggregation inhibitor and anti-inflammatory therapy were scheduled.
In conclusion, MIS-C occurred as a complex inflammatory condition related to past SARS-CoV-2 infection. The patient developed significant coronary artery disease with no ventricular dysfunction, requiring three-line stepwise immune-mediated treatment and showing good response during hospitalization and follow-up after discharge.
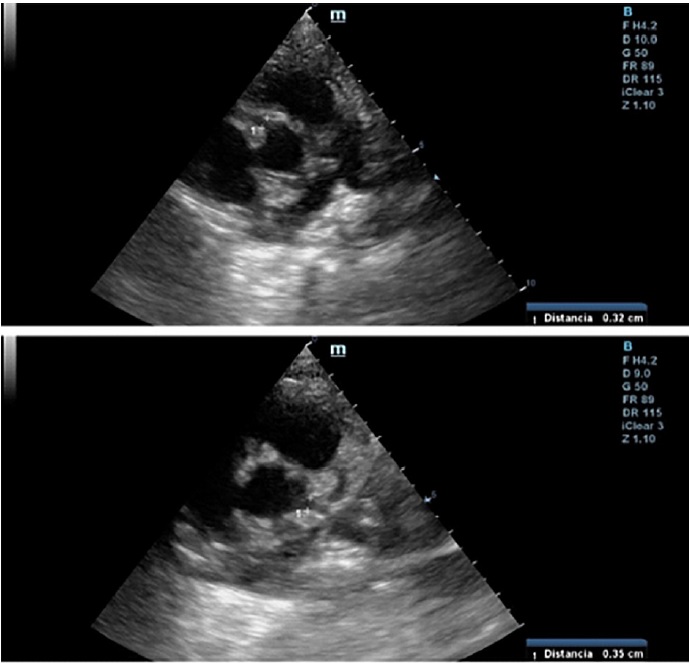
Fig. 1 2D echo-Doppler (vessel short axis): Right coronary artery (above) and left coronary artery (below) are shown.
Pediatric Cardiology and Cardiovascular Surgery Division. Hospital Privado Universitario de Córdoba. Instituto Universitario de Ciencias Biomédicas de Córdoba. Tel: 543514688220. E-mail: Ebanille@hotmail.com
Table 1
|
Coronary
arteries diameter (Z-score)
|
Day 1 |
Day 6 |
Day 11 Proximal 2.4 mm (+2.6) |
Day 21 |
Day 35 |
Day 119 |
|
Right coronary artery |
1.6 mm (+0.3) |
3.5 mm (+5.8) |
Distal 3.5 mm (+7.2) |
2.2 mm (+2.7) |
2 mm (+1.5) |
1.9 mm (+1.2) |
|
Left coronary artery |
1.8 mm (+0.27) |
3.5 mm (+5.2) |
Proximal 3.5 mm (+5.2) Distal 3.9 mm (+8) |
2.4 mm (+1.9) |
1.8 mm (+0.27) |
2.4 mm (+2) |
Emilia Croigverg, Patricia Baselga, Susana Chicco Campos, Valeria Hasbani, Eliana Santillán
Conflicts of interest
None declared. (See authors' conflict of interests forms on the web)
Ethical considerations
Not applicable.
REFERENCES
- Sociedad Argentina de Pediatría. Consenso sobre el tratamiento del síndrome inflamatorio multisistémico asociado a COVID-19. Arch Argent Pediatr 2021;119:S198-S211. https://doi.org/10.5546/aap.2021.s198
- Taffarel P, Jorro Barón F, Rodríguez AP, Widmer J, Meregalli C. Síndrome inflamatorio multisistémico en niños, relacionado con COVID-19: actualización a propósito de la presentación de 2 pacientes críticos. Arch Argent Pediatr 2021;119: e26-e35. https://doi.org/10.5546/aap.2021.e26
- Sociedad Argentina de Pediatría y Sociedad Argentina de Cardiología. Enfermedad de Kawasaki: consenso interdisciplinario e intersociedades (guía práctica clínica). Arch Argent Pediatr. 2016.
- Asociación Española de Pediatría. Documento español de consenso sobre diagnóstico, estabilización y tratamiento del síndrome inflamatorio multisistémico pediátrico vinculado a SARS-CoV-2 (SIM-PedS). https://doi.org/10.1016/j.anpedi.2020.09.005
- Ministerio de Salud de la Nación. “Manejo inicial del síndrome inflamatorio multisistémico en niños y adolescentes relacionados temporalmente al COVID-19 (SIM-C)” Recomendaciones 31 de agosto de 2020. Disponible en: https://bancos.salud.gob.ar/sites/default/files/2020-09/covid19-manejo-inicial-del-sindrome-inflamatorio-multisistemico-en-ninios-y-adolescentes-relacionados-temporalmente-alcvod19-sim-c.pdf
- American College of Rheumatology Clinical Guidance for Multisystem Inflammatory Syndrome in Children Associated with SARSCoV- 2 and Hyperinflammation in Pediatric COVID-19: Version 3. Arthritis Rheumatol. 2022. https://doi.org/10.1002/art.42062
Rev Argent Cardiol 2023;91:357-358.
http://dx.doi.org/10.7775/rac.v91.i5.20673
Iatrogenic Coronary Stenosis and Ventricular Aneurysm after Surgical Treatment as a Cause of Non-atherosclerotic Ischemic-Necrotic Cardiomyopathy
Thoracic trauma is responsible for 25% of trauma deaths. Approximately 15 to 30% of patients with penetrating cardiothoracic trauma require surgical treatment. Albeit infrequent, one of its complications is iatrogenic coronary stenosis due to surgical suture. Iatrogenic coronary stenosis is an unusual complication involving a high rate of morbidity and mortality. It can be observed after surgical treatment, as valve replacement surgery or penetrating cardiothoracic trauma repair.
Clinical presentation depends on the affected coronary artery, the degree, site and extent of injury, as well as the preoperative myocardial reserve. During the procedure, acute myocardial infarction, ventricular arrhythmia or cardiogenic shock can occur, leading to urgent coronary artery bypass. However, unstable angina, congestive heart failure, ventricular arrhythmias and sudden death are the classical presentations after several months of the procedure.
We present the case of a 30-year-old male patient with history of non-atherosclerotic dilated ischemicnecrotic cardiomyopathy, with reduced left ventricular ejection fraction (LVEF) (35%) and ventricular aneurysm (Figure 1), as a result of extrinsic iatrogenic lesion following anterior descending artery suture due to gunshot wound and urgent surgery. He subsequently underwent various hospitalizations for recurrent sustained monomorphic ventricular tachycardia (SMVT) (Figure 2a), leading to implantable cardioverter defibrillator (ICD) placement and substrate ablation, with favorable outcome.
A biannual ICD reading has been performed, with no detection of new arrhythmic events up to the present (two years after the ablation).
Sustained monomorphic ventricular tachycardia is an important cause of sudden death and morbidity in patients with coronary artery heart disease. It originates in the scar tissue and can develop in the subacute phase or even years after an ischemic event. The extent of myocardial necrosis and the degree of left ventricular dysfunction are arrhythmia risk predictors, higher in patients with reduced LVEF and extensive ischemia. (1)
The pathophysiology involves a reentrant mechanism associated to an ischemic event, with already installed or developing scar in more than 95% of cases.
Conversely, that originating from fibrosis with intertwined myocardial bundles, creates the substrate for a fixed and or functional conduction blockade, with interrupted and slow conduction that leads to an arrhythmia-triggering mechanism.
Ventricular aneurysms are observed in the left ventricle in 95% of cases. They are defined by the presence of a scar generating chamber deformity, with non-contractile dilation restricted to the ventricle. It can be associated with severe congestive heart failure, low cardiac output and even cardiogenic shock, systemic embolisms and relapsing ventricular tachycardia (VT) or a combination of the former
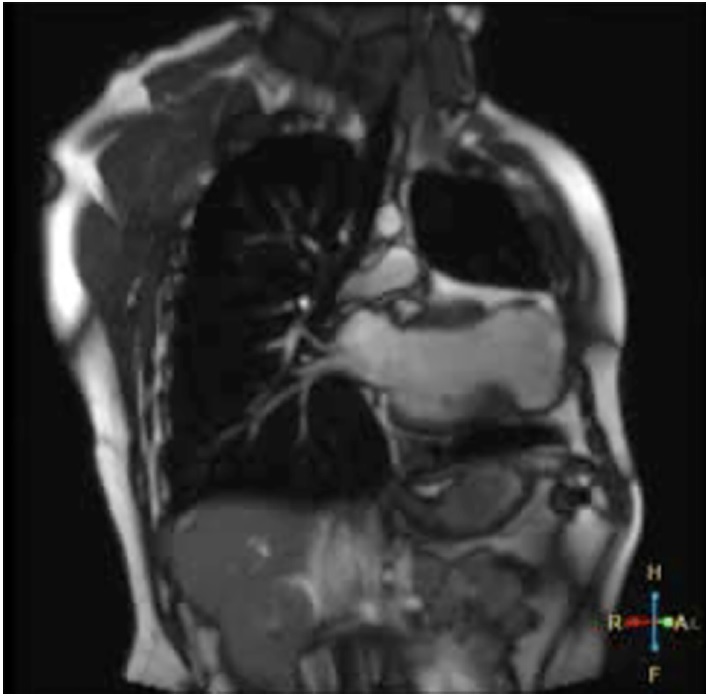
Fig. 1 T2 sequence magnetic resonance coronal section showing left ventricular dilation, presence of apical aneurysmatic sac, myocardial thinning and low-intensity signal.
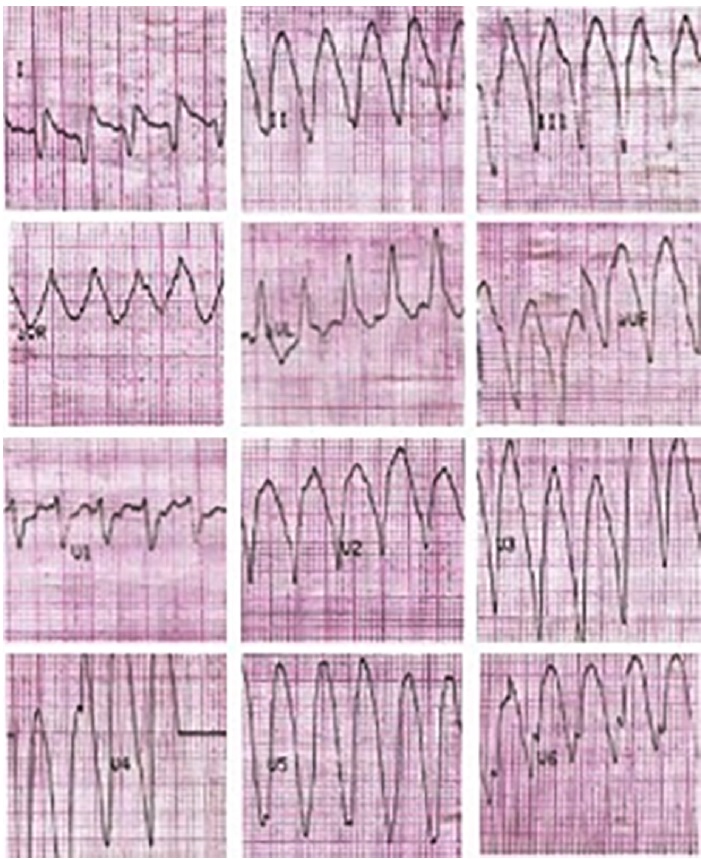
Fig. 2a Monomorphic ventricular tachycardia with wide QRS complex, regular, at 190 bpm, with apical origin compatible with ventricular aneurysm.
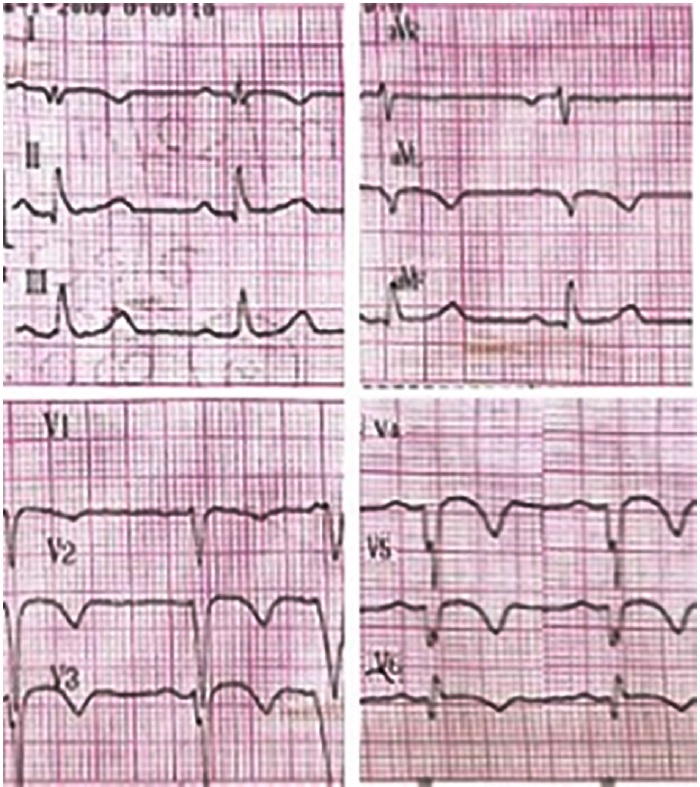
Fig. 2b Electrocardiogram after electrical cardioversion showing sinus rhythm at 65 bpm, with presence of negative Q and T waves in the anterolateral wall, compatible with necrosis sequel and repolarization disorders.
The first therapeutic option when an iatrogenic ventricular aneurysm is detected is revascularization, which can be achieved by percutaneous coronary intervention (PCI) or aorto-coronary bypass, depending on the time at which the damage is detected. If it is detected during surgery and/or the vessel is completely obstructed by a suture, a bypass is chosen; on the other hand, if the detection is after closure of the sternotomy, PCI is the best option to restore flow and avoid a new sternotomy. Sometimes a hybrid procedure can be chosen. It is also important to note that the long-term results of any of these interventions are unknown. (2)Currently, we have different therapeutic alternatives for the secondary prevention of VT. Surgical resection of ventricular aneurysm to eliminate ventricular tachycardia is out of use due to the high mortality rate in the procedure.
At present, ICD has become the treatment of choice in subjects with ventricular arrhythmia associated with coronary artery disease. It is a I A recommendation in patients with LVEF ≤35% in NYHA functional class (FC) II-III or in FC I with LVEF ≤30% in primary prevention, and in cardiac arrest survivors due to VT or ventricular fibrillation (VF) not attributable to reversible or transient causes in secondary prevention. Reported studies based on the use of this device in secondary prevention evidenced a significant reduction in the mortality rate, as a result of sudden cardiac death reduction and not to a decrease in the number of arrhythmic events. (3)
In these cases, combined therapy with pharmacological treatment and/or ablation is additionally recommended, ablation being selected for those with SMVT, due to a lower rate of recurrence. (4)
The approach used for VT ablation depends on several factors, as inducibility, sustainability and clinical tolerance to VT. Moreover, the combination of computed tomography (CT) and nuclear magnetic resonance (MRI) anatomical images of the myocardial scar, among others, contribute to the detection of the arrhythmogenic site. (5)
In cases of no inducible or poorly tolerated VT caused by hemodynamic instability or systolic and diastolic ventricular dysfunction, substrate ablation can be performed using 3D navigation systems. With this technique, the radiofrequency energy is administered in regions with scar tissue that topographically correlate with the region of origin of the tachycardia reentrant circuit, as suggested by the intracavitary electrogram where abnormal potentials are recorded. The areas of extremely low voltage are designed as dense scar. Radiofrequency lesions can then be generated in parallel to the scar border zone (where the amplitude of the bipolar electrogram, by pacemaker technique, is generally between 0.5 mV and 1.0 mV).
When a VT similar to that presented by the patient can be induced, the ablation is performed in the zone in which the arrhythmia was generated, and once this has been accomplished, the success is confirmed if it cannot be induced again. If, however, as in this case, it cannot be reproduced, the ablation is carried out mediated by the anatomical substrate and it is not possible to assess its effectiveness. (6)
In general, the ablation is immediately successful, and abolishes one or more scar related SMVT in 75- 95% of cases. Nevertheless, the recurrence of VT with the same morphology, or more often with a different morphology, can occur in 12-50% of patients, needing a reintervention. Therefore, a successful VT ablation does not eliminate the need for ICD or oral antiarrhythmic treatment. Due to the high SMVT morbidity and mortality and recurrence in patients with ischemic heart disease, an effective treatment should be chosen, which in most cases consists of a combined therapy. Implantable cardioverter defibrillator placement is chosen to decrease the incidence of sudden death, and the use of substrate ablation and/or oral antiarrhythmic agents to avoid recurrence.
Ana Inés María Vinuesa, Giuliana Tresenza, Guillermo LiniadoMTSAC
Conflicts of interest
None declared. (See authors' conflict of interests forms on the web)
Ethical considerations
Not applicable.
REFERENCES
- Guandalini GS, Liang JJ, Marchlinski FE. Ventricular Tachycardia Ablation: Past, Present, and Future Perspectives. JACC Clin Electrophysiol. 2019;5:1363-83. https://doi.org/10.1016/j.jacep.2019.09.015
- Comanici M, Raja SG. Iatrogenic Coronary Artery Injuries during Non-cardiac Artery Adult Cardiac Surgery. AME Med J 2023;8-12
- Al-Khatib SM, Stevenson WG, Ackerman MJ, Bryant WJ, Callans DJ, Curtis AB, et al. 2017 AHA/ACC/HRS Guideline for Management of Patients With Ventricular Arrhythmias and the Prevention of Sudden Cardiac Death: A Report of the American College of Cardiology/ American Heart Association Task Force on Clinical Practice Guidelines and the Heart Rhythm Society. J Am Coll Cardiol. 2018;72:e91- e220. https://doi.org/10.1016/j.jacc.2017.10.054
- Liu G, Xu X, Yi Q, Lv T. The efficacy of catheter ablation versus ICD for prevention of ventricular tachycardia in patients with ischemic heart disease: a systematic review and meta-analysis. J Interv Card Electrophysiol. 2021;61:435-43. https://doi.org/10.1007/s10840-020-00848-1
- Cuculich PS, Schill MR, Kashani R, Mutic S, Lang A, Cooper D, et al. Noninvasive Cardiac Radiation for Ablation of Ventricular Tachycardia. N Engl J Med. 2017;377:2325-36. https://doi.org/10.1056/NEJMoa1613773.
- Berruezo A, Fernández-Armenta J, Andreu D, Penela D, Herczku C, Evertz R, et al. Scar dechanneling: new method for scar-related left ventricular tachycardia substrate ablation. Circ Arrhythm Electrophysiol. 2015;8:326-36. https://doi.org/10.1161/CIRCEP.114.002386 a 65 lpm, con presencia de ondas Q y T negativas en cara anterolateral, compatible con secuela de necrosis y trastornos en la repolarización.
Rev Argent Cardiol 2023;91:358-360.
http://dx.doi.org/10.7775/rac.91.i5.20674
Deep Vein Thrombosis by Unusual May-Thurner Syndrome Variant. Percutaneous Mechanical Thrombectomy Resolution Without Use of Thrombolytic Agents
We present a case of a 63-year-old patient admitted to the emergency room (ER) for complaints of dysphagia. He reported a left-sided neck distension which later affected his capacity to eat appropriately. These symptoms appeared after a pacemaker implantation for second-degree atrioventricular block, two weeks prior. There were no symptoms of heart failure, syncope, pre-syncope, palpitations, dizziness, constitutional symptoms, or fever. The physical examination showed a cervical asymmetry, with a distended left side, associated with local warmth, and discrete asymmetry between the right and left arms. The rest of the physical examination was unremarkable. The patient has a history of schizophrenia, depression, and dyslipidemia. Following an invasive procedure, the authors suspected an infection related to the procedure as the point of origin.
The patient underwent a computed tomography (CT) scan that reported thrombosis of the left internal jugular vein, with occlusion of the left brachiocephalic and subclavian veins (shown in Fig. 1). There was no evidence of collections/abscesses. A brief transthoracic echocardiography was performed in the ER, which rose the suspicion of thrombus/endocarditis. The subsequent transesophageal echocardiogram documented a thrombus adherent to the pacemaker leads. There was an equivocal image of vegetation. Blood cultures, blood panel with C-reactive protein (CRP) and procalcitonin (PCT) were collected. The patient was also scheduled for a repeat CT for evaluation of pulmonary embolism.
Anticoagulation was immediately started with low molecular weight heparin (LMWH). Blood cultures and PCT were normal. There was no evidence of pulmonary embolism on the new CT scan.
After treatment with LMWH for a week, there was a complete resolution of the thrombus (shown in Fig. 2). The patient was also seen in the immunohemotherapy clinic, and thrombophilia was discarded. After a 12-month follow-up, the patient remains asymptomatic and generally well.
Reported permanent pacemaker complications are mostly related to the risk of infection and thrombosis and embolic events. Other rarer complications also described are beyond the scope of this scientific letter. (1)
Serious thrombotic events related to pacemaker implantation have been described, with an incidence from 0.6% to 3.5%. These serious events include heart failure presentation and pulmonary thromboembolism. (2)
Nevertheless, clinically asymptomatic thrombus appears to be much more frequent, with an incidence of up to 35-45% in the same cohorts. (3)
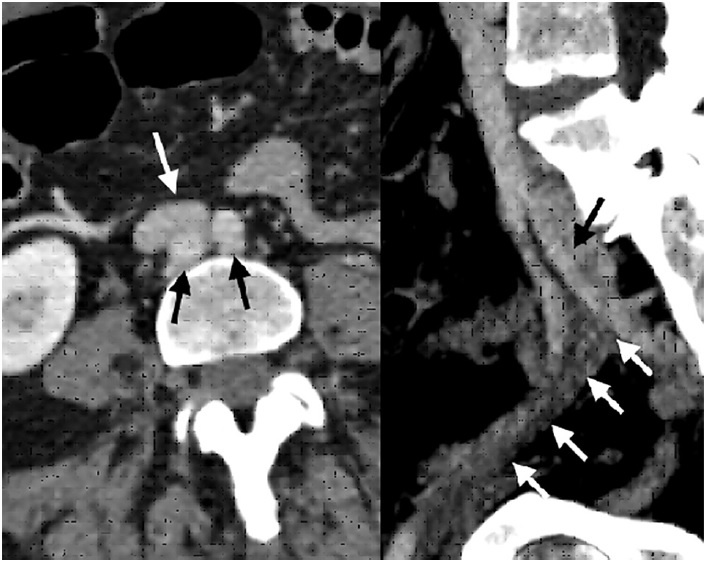
Fig. 1 Left. Computed tomography angiography axial section evidencing aortic bifurcation (black arrows), behind the iliocaval junction (white arrow). Right. Sagittal section shows compression by the left hypogastric artery (black arrow) causing external iliac vein thrombosis (white arrows).
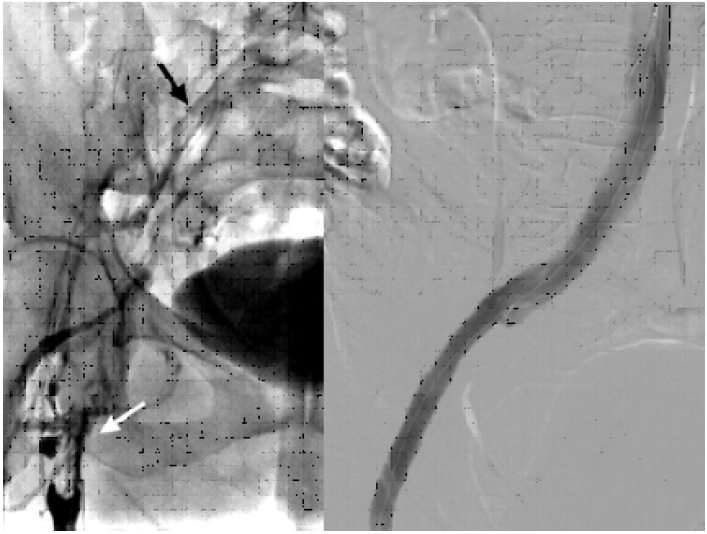
Fig. 2 Left. Initial phlebography showing thrombosis from the proximal femoral vein (white arrow), which rechannels with low flow at the common iliac vein level (black arrow). Right. Final phlebography after stent placement showing absolute permeability of the iliac axis
Miguel Ferrer, Sebastián Romero, Fernando Álvarez, Ricardo Parra, Horacio Romero
Division of Vascular Surgery and Division of Hemodynamics. Hospital Privado Regional del Sur. San Carlos de Bariloche, Argentina
Conflicts of interest
None declared.
(See authors’ conflicts of interest forms on the website).
REFERENCES
- Mako K, Puskas A. May-Thurner syndrome - Are we aware enough? Vasa 2019;48:381-8. https://doi.org/10.1024/0301-1526/a000775
- Steinberg JB, Jacocks MA. May-Thurner Syndrome: a previously unreported variant. Ann Vasc Surg 1993;7:577-81. https://doi.org/10.1007/BF02000154
- Sharafi S, Farsad K. Variant May-Thurner syndrome: Compression of the left common iliac vein by the ipsilateral internal iliac artery. Radio Case Rep 2018;13:419-423. https://doi.org/10.1016/j.radcr.2018.01.001
- Kakkos SK, Gohel M, Baekgaard N, Bauersachs R, Bellmunt- Montoya S, Black SA, et al., European Society for Vascular Surgery (ESVS) 2021 Clinical Practice Guidelines on the Management of Venous Thrombosis. European Journal of Vascular and Endovascular Surgery. https://doi.org/10.1016/j.ejvs.2020.09.023
- Haig Y, Enden T, Sandset PM, CaVenT Study Group. Postthrombotic syndrome after catheter-directed thrombolysis for deep vein thrombosis (CaVenT): 5-year follow-up results of an open-label, randomized controlled trial. Lancet Haematol 2016,3:e64-71. https://doi.org/10.1016/S2352-3026(15)00248-3
- Loffroy R, Falvo N, Chevallier O. Single-Session Percutaneous Mechanical Thrombectomy Using the Aspirex ®S Device Plus Stenting for Acute Iliofemoral Deep Vein Thrombosis: Safety, Efficacy, and Mid-Term Outcomes. Diagnostics (Basel) 2020;10:544. https://doi.org/10.3390/diagnostics10080544
Rev Argent Cardiol 2023;91:360-362.
http://dx.doi.org/10.7775/rac.v91.i5.20671
https://creativecommons.org/licenses/by-nc-sa/4.0/
