Alberto Lorenzatti1
1 Instituto Médico DAMIC / Fundación Rusculleda de Investigación en Medicina. Córdoba, Argentina
Epidemiologic and genetic studies suggest that low- density lipoprotein cholesterol (LDL-C) is directly causal in atherosclerotic cardiovascular disease, (1) while the results of many randomized clinical trials have clearly demonstrated that lowering LDL-C levels is associated with a proportional reduction in the risk of cardiovascular (CV) events. (2)
In modern society, individuals have LDL-C levels well above 25 mg/dL, what is considered physiological. (3) Similarly, individuals with genetically determined low LDL-C levels are healthy and have a very low incidence of CV events. (4) These observations, together with the results of the most recent clinical trials with proprotein convertase subtilisin/kexin type 9 inhibitors (PCSK9i), have revealed significant reductions in LDL-C levels beyond classical recommendations. Consequently, achieving very low LDL-C levels may safely confer additional clinical benefits. (5,6) In 2019, the “ESC/EAS Guidelines for the management of dyslipidaemias: lipid modification to reduce cardiovascular risk”, (7) set new LDL-C targets ( < 55mg/dL + 50% reduction from baseline) for individuals at very high cardiovascular risk, based on the very strong evidence emerging form the most recent studies with nonstatin medications, on top of statins. These LDL-C control targets have been widely adopted by local and international recommendations.
On the other hand, the concept of residual cardiovascular risk (RCVR) emerged some years ago when, despite the excellent results obtained with the use of statins in cardiovascular prevention, a significant number of subjects showed progression of their atherosclerotic disease and clinical events. Thus, residual risk began to be understood as the risk that persists beyond the benefit provided by statin treatment.
The concept of residual risk expanded rapidly to other factors or markers considered as determinants of risk despite appropriate control of LDL cholesterol. 1 Instituto Médico DAMIC / Fundación Rusculleda de Investigación en Medicina. Córdoba, Argentina In this way, the following concepts have emerged in the broad spectrum of residual risk: residual inflammatory risk, residual thrombotic risk, residual lipoprotein (a) risk, and residual triglyceride risk and residual diabetes risk (residual metabolic risk). (8,9)
In general, residual risk means the risk of CV events that remains even after receiving treatment or taking measures to reduce that risk. Additionally, other factors like genetics, lack of adherence to treatment or lifestyle suggestions, and even ineffective medications can contribute to residual risk.
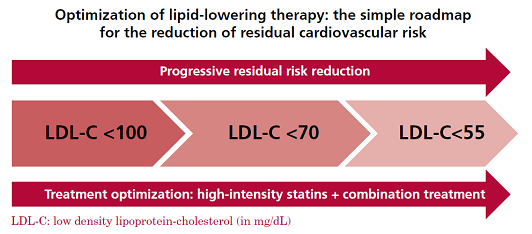
In this issue of the Argentine Journal of Cardiology, Masson et al. publish a very interesting study about “The impact of optimizing lipid-lowering therapy on residual cardiovascular disease risk” using the SMART-REACH model that predicts the risk of recurrent CV events.(10) They evaluated 10-year and lifetime residual risks in a secondary prevention population with LDL-C levels above the recommended goal (> 55 mg/dL) and determined the impact of optimizing lipid-lowering therapies in terms of RCVR reduction.
 The authors included 187 patients (mean age, 67.9 ± 9.3 years; 72.7% men). The calculated 10-year and lifetime residual risks, approximately 37% and 60%, respectively. The use of high-intensity statins (58.9%) was similar to other secondary prevention cohorts, (11) although the administration of ezetimibe in the study by Masson et al. was much higher (40.6%), probably because many cardiologists have started using this therapy. As expected, very few people were prescribed a PCSK9 inhibitor
The authors included 187 patients (mean age, 67.9 ± 9.3 years; 72.7% men). The calculated 10-year and lifetime residual risks, approximately 37% and 60%, respectively. The use of high-intensity statins (58.9%) was similar to other secondary prevention cohorts, (11) although the administration of ezetimibe in the study by Masson et al. was much higher (40.6%), probably because many cardiologists have started using this therapy. As expected, very few people were prescribed a PCSK9 inhibitor
Nearly 40% of patients reached the targets after optimizing treatment with statins (1st step). An additional 40% reached the goals in a 2nd step by adding ezetimibe or a PCSK9i to the model. Finally, all those who could be included in a 3rd step reached the goals in the simulation model.
The study highlights the urgent necessity to control LDL-C effectively with the tools available in clinical practice. Similarly, the 10-year and lifetime residual risks decreased steadily after the 1st, 2nd a 3rd steps in treatment optimization. The combination of triple lipid-lowering drugs reduced the 10-year residual risk by 12.8% and the lifetime residual risk by 16.6%, and the number necessary to treat (NNT) to prevent one event was only 8.2 and 6.1, respectively. Finally, 54% and 63% of the actions implemented to optimize treatment showed a 10-year or lifetime NNT < 30 to prevent an event, respectively, which categorically underscores the benefits of optimizing treatment for LDL-C control in these patients
Data from the recently presented INTERASPIRE study (ESC Congress, Amsterdam, August 2023) indicate that just 20% of patients at very high CV risk in our nation reach the LDL-C target of < 55 mg/dL, demonstrating the need for improved management of LDL-C and reduction of RCVR through simple lipidlowering treatment optimization.
Conflicts of interest
None declared.
(See authors’ conflict of interests forms on the web).
https://creativecommons.org/licenses/by-nc-sa/4.0/

Rev Argent Cardiol 2023;91:305-306. http://dx.doi.org/10.7775/rac.v91.i5.20663
SEE RELATED ARTICLE: Rev Argent Cardiol 2023;91:309-316. http://dx.doi.org/10.7775/rac.v91.i5.20666
©Revista Argentina de Cardiología