Julián Feder1 , Ana Lía Gamarra2 , Diego Conde1 MTSAC , Alan Sigal1 , Juan Furmento1 , Pablo Lamelas1 , Ian Chapman1, Mariano Vrancic 1, Marcelo Trivi 1, Juan Pablo Costabel1 MTSAC
1Ischemic Heart Disease Program of Instituto Cardiovascular de Buenos Aires.
2Division of Cardiology, Hospital Italiano de Buenos Aires
Address for correspondence: Juan Pablo Costabel - Instituto Cardiovascular de Buenos Aires, Argentina - Blanco de Encalada 1543 - 1428, Buenos Aires, Argentina - E-mail: jpcostabel@icba.com.ar
Rev Argent Cardiol 2023;91:349-356. http://dx.doi.org/10.7775/rac.v91.i5.20670
ABSTRACT
The decision on the best revascularization strategy for patients with multivessel disease has become a complex task as coronary angioplasty has improved its results. In the following review, we set out to evaluate the variables that, in our experience, define the benefit of one technique over the other, understanding that in this way the treating physician's decision will become simpler and more objective. On the other hand, and celebrating the healthy prominence given to patients, we believe that this evaluation allows solid arguments to help them in decision making.
Key words: Percutaneous Coronary Angioplasty - Coronary Artery Bypass Graft - Multiple Vessels Disease
RESUMEN
La decisión sobre la mejor estrategia de revascularización para los pacientes con enfermedad de múltiples vasos se ha tornado una tarea compleja a medida que la angioplastia coronaria ha mejorado sus resultados. En la siguiente revisión nos propusimos evaluar las variables que en nuestra experiencia definen el beneficio de una técnica sobre la otra, entendiendo que de esta manera la decisión del médico tratante se hace más sencilla y objetiva. Por otro lado, y festejando el saludable protagonismo que se le da al paciente, creemos que esta evaluación permite ofrecer argumentos sólidos para ayudarlo en la toma de la decisión.
Palabras clave: Cirugía de revascularización miocárdica - Angioplastia coronaria - Enfermedad coronaria de múltiples vasos
Received: 08/01/23
Accepted: 09/12/23
INTRODUCTION
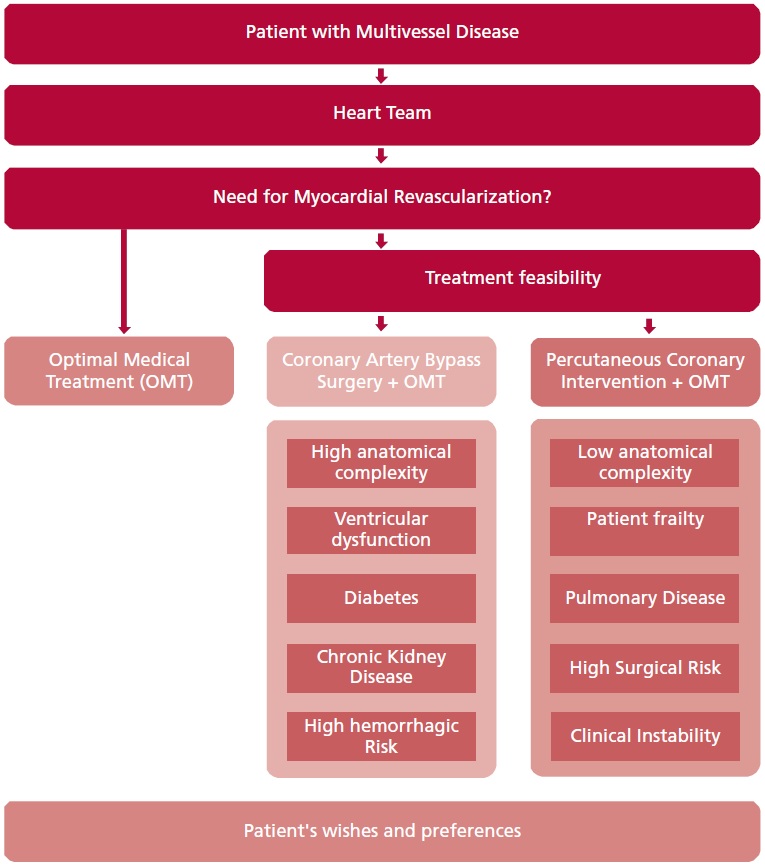
Patients with multivessel coronary disease represent a frequent challenge for the cardiological medical team at the time of indicating the need for revascularization and the way to perform it. With the purpose of providing evidence for decision making, a review was carried out of the key points considered when defining the conduct in daily practice. The following bibliographic analysis focuses in patients who according to the treating team require revascularization (Figure 1).
Comparison between angioplasty and surgical revascularization
Several studies comparing coronary artery bypass graft surgery (CABG) with percutaneous coronary intervention (PCI) in patients with multivessel lesions (1-9) show some similarities, as a stable or stabilized clinical condition for random patient assignment. In addition, there are differences regarding the technology employed to carry out PCI, such as type of stent and use of intracoronary technology [intravascular ultrasound (IVUS), optical coherence tomography (OCT) or measurement of fractional flow reserve (FFR)], as well as those related with the surgical technique, with or without extracorporeal circulation, number of bridges, arterial conduits and surgical experience. However, in general terms, short- and mid-term follow-up have shown a higher rate of events with PCI, mainly at the expense of need for new revascularization and acute myocardial infarction (AMI), but with minimal differences in cardiovascular mortality
How does coronary anatomical complexity influence in the decision?
One of the first factors to consider at the time of decision making in the revascularization strategy of acute or chronic ischemic heart disease are the characteristics of the coronary anatomy, which should be evaluated both by an expert in angioplasty as by a surgeon to decide the feasibility of revascularization. Specialists should define which lesions will be treated and which will be impossible to resolve.
To quantify the complexity, the SYNTAX score was developed with 11 analyzable variables derived from the coronary angiography. These characteristics generate the score, a continuous variable that was analyzed in the core study as low ( < 22), intermediate (23-32) and high (> 32) category. (10)
In the original study, PCI revascularization did not reach the non-inferiority objective when compared with surgical revascularization. However, when the results at 12 months were evaluated by subgroup analyses, differences disappeared in patients with low and intermediate score, with a statistically similar incidence of the composite event (all-cause death, stroke, AMI or new revascularization (13.6% vs. 14.7%, p=0.47 for the low score; and 16.7% vs. 12%, p=0.10 for the intermediate score). (11)
In the case of the high SYNTAX score, this difference not only persisted, but a greater separation between the curves was observed (23.4% vs. 10.9%, p < 0.001). When the trend for events was analyzed along time, the differences were preserved in the 3-year follow-up, and the curves tended to separate in favor of the surgical strategy at 5 years. (6)
Therefore, the difference in the rate of major adverse cardiovascular and cerebrovascular events (MACCE) started to be significant in patients with intermediate SYNTAX score, with 110 events (36.0%) in the PCI arm vs. and 72 (25.8%) in the CABG arm (HR 1.50; 95% CI 1.11-2.01, p=0.008) at the expense not only of more revascularizations but also of higher AMI rate. When the rate of events was evaluated in patients with high SYNTAX score, this difference increased, with the addition that a significant difference in MACCE [PCI 126 (44.0%) vs. CABG 80 (26.8%), HR 1.89; 95% CI 1.43–2.50, p < 0.0001] and specifically in cardiovascular death [PCI 38 (13.6%) vs. CABG 14 (4.9%), HR 2.99; 95% CI 1.62–5.52, p= 0.0002] was observed.
Tables 1 and 2 summarize the results of the comparative studies with follow-up at 1 and 5 years.
A sub-study derived from the SYNTAX study at 10 years (SYNTAXes) provides data of a more prolonged follow-up with a low rate of abandonment, as the study ended with 93% of the population in the CABG group and 95% in the PCI group. (12)
In this study, the primary endpoint was total mortality at 10 years, with evident absence of statistically significant global difference. When subgroups were evaluated, it could be seen that in patients with left main coronary artery disease the difference between both arms continued to be non-significant, while in 3-vessel disease it became statistically significant in favor of surgery: PCI 151 (28%) vs. CABG: 113 (21%), HR 1.41; 95% CI 1.10- 1.80. (12,13)
In conclusion, it seems that in patients with more complex coronary anatomy, PCI does not achieve the same mid-and long-term results as in those with low or median complexity. The SYNTAX score is a better predictor of PCI than of CABG outcomes and provides a good tool to assess the complexity and predict the results of the former.
How does the possibility of achieving complete revascularization influence in the decision?
The concept of complete revascularization was tested in several interventional works, in which it was seen that the lower the number of unresolved residual anatomical or functional lesions, the better the mid- and long-term evolution of patients. The residual SYNTAX score is a good way of rating this incomplete revascularization, and studies have demonstrated that when the residual value is ≥8, the rate of events increases significantly (14-16)
Complete revascularization was associated with a reduction of ischemic events and better quality of life, so it is an important factor at the time of deciding the technique to implement and the objective to achieve, both for hemodynamic specialists as well as surgeons.
In most studies, CABG has achieved greater rate of complete revascularization and this is one of the potential reasons for its superiority. (17,18)
What is the influence of ventricular function?
In patients with severe ventricular dysfunction, the importance of complete medical treatment is well known, with a great body of evidence for multiple pharmacological groups. When therapeutic strategies in patients with ischemic-necrotic etiology are evaluated, it is intuitive to consider both surgical and percutaneous revascularization; however, in the evidence currently available, no studies have been found comparing in the same conditions CABG vs. PCI in patients with ischemic-necrotic disease, exclusively with severe ventricular dysfunction.
The STICH study compared surgical revascularization versus optimal medical therapy (OMT) in patients with left ventricular ejection fraction (LVEF) ≤35%. (19)
No differences were found in terms of 5-year total mortality, but a statistically significant reduction of cardiovascular hospitalization and mortality rates was achieved: CABG 58% vs. OMT 68% (HR 0.74; 95% CI 0.64-0.85, p < 0.001). In the 10-year follow-up (STICHES study, published in 2016) a reduction of total mortality was evidenced with surgical treatment compared with medical therapy: 58.9% vs. 66% (HR 0.84; 95% CI 0.73-0.97, p=0.02). (20)
These findings seem to demonstrate that in the long-term follow-up, the rate of perioperative mortality loses importance with respect to posterior events.
In the case of percutaneous revascularization, the REVIVED-BCIS2 trial including 700 patients with LVEF ≤35% of ischemic-necrotic etiology, with viability of at least 4 segments in territories of feasible revascularization, compared the PCI strategy plus OMT (N=347) vs. OMT alone (n=353). (21)
The primary endpoint at 24 months was a composite of total mortality and hospitalizations for heart failure. The incidence of events was similar: 37.2% with PCI vs. 38% in the OMT group (HR 0.99; 95% CI 0.78-1.27, p=0.96). Neither were there differences observed in ventricular function measured by transthoracic Doppler echocardiography at 6 and 12 months nor in quality of life measured by KCCQ and EQ-5D-5L scales.
Therefore, when speaking of patients with ischemic- necrotic ventricular dysfunction, no direct conclusions can be drawn on the superiority of CABG over PCI due to the gap in the evidence. Nevertheless, retrospective cohort studies are available, as the one published by Sun et al. in 2020, evaluating 12 113 patients in Ontario, Canada with severely impaired LVEF of ischemic-necrotic origin, undergoing surgical or percutaneous revascularization and followed up for 5 years. (22)
After performing a propensity score to match baseline variables, and with 2397 patients in each strategy, surgical revascularization superiority was found, with PCI associated to HR 1.6 (95% CI 1.4-1.7, p < 0.001) for total mortality; HR 1.4 (95% CI 1.1-1.6, p < 0.001) for cardiovascular mortality and HR 2.0 (95% CI 1.9-2.2, p < 0.001) for MACCE. The subgroup analyses evidenced that the difference in total mortality disappeared in those with complete revascularization.
Table 1: Events at 12 months.
|
Variable |
Study
|
Percutaneous coronary
intervention |
Coronary
artery bypass
grafting |
|
Mortality |
FAME 3 (1) |
1.6% (12/757) |
0.9% (6/743) |
|
SYNTAX (11) |
4.3% (39/903) |
3.3% (30/897) |
|
|
FREEDOM (3) |
3.3% (32/953) |
4% (38/947) |
|
|
CARDia (4) |
3.2% (8/254) |
3.2% (8/248) |
|
|
VA CARDS (5) |
7.9% (8/101) |
5.15% (5/97) |
|
|
AMI |
FAME 3 |
5.2% (39/757) |
3.5% (26/743) |
|
SYNTAX |
4.76% (43/903) |
3.12% (28/897) |
|
|
FREEDOM |
5.77 % (55/953) |
3.16% (30/947) |
|
|
SYNTAX II |
4.7% (12/254) |
4.4% (11/248) |
|
|
VA CARDS |
4.95% (5/101) |
13.4% (13/97) |
|
|
Stroke |
SYNTAX |
0.5% (5/903) |
2.11% (19/897) |
|
FREEDOM |
0.83% (8/953) |
1.79% (17/947) |
|
|
CARDia |
0.4% (1/254) |
2.8% (7/248) |
|
|
VA CARDS |
1% (1/100) |
1% (1/97) |
|
|
FAME 3 |
0.9% (8/757) |
1.1% (8/743) |
|
|
Revascularization of the treated vessel |
SYNTAX |
13% (120/903) |
5.5% (50/897) |
|
FREEDOM |
12% (117/953) |
4.4% (42/947) |
|
|
VA CARDS |
11% (12/101) |
11.4% (11/97) |
|
|
CARDia |
11.8% (30/254) |
2% (5/248) |
|
|
FAME 3 |
5.9% (44/754) |
3.9% (28/743) |
|
|
Stent or graft occlusion |
SYNTAX |
3.3% (28/848) |
3.4% (27/784) |
|
FAME 3 |
0.8% (6/754) |
NA |
AMI: Acute myocardial infarction; NA: not available.
Table 2: Events at 5 years.
|
Variable |
Study |
Percutaneous coronary intervention |
Coronary artery bypass grafting |
|
|
SYNTAX (6) |
13.9% (129/909) |
11.4% (94/897) |
|
FREEDOM (7) |
11.2% (114/953) |
8.75% (83/947) |
|
|
CARDia (8) |
14.5% (37/254) |
12.9% (32/248) |
|
|
SYNTAX II (9) |
8.1% (36 /254) |
NA |
|
|
AMI |
SYNTAX |
9.7% (83/903) |
3.8% (33/897) |
|
FREEDOM |
10.2% (98/953) |
4.9% (48/947) |
|
|
CARDia |
14.7% (36/254) |
7.25% (18/248) |
|
|
SYNTAX II |
2.7% (12/454) |
NA |
|
|
Stroke |
SYNTAX |
2.4% 20/903 |
3.7% (31/897) |
|
FREEDOM |
2.1% (20/953) |
3.9% (37/947) |
|
|
CARDia |
3.14% (8/254) |
4.4% (11/248) |
|
|
SYNTAX II |
2.3% (10/454) |
NA |
|
|
Revascularization of the treated vessel |
SYNTAX |
25.9% |
13.7% |
|
SYNTAX II |
13.8% (60/454) |
NA |
|
|
Stent or graft occlusion |
SYNTAX |
5.2% (47/903) |
3.56% (32/897) |
|
SYNTAX II |
1.4% (6/454) |
NA |
AMI: Acute myocardial infarction; NA: not available.
What is the influence of the coronary syndrome affecting our patient?
Studies comparing CABG versus PCI were performed in chronic coronary syndrome or stabilized acute coronary syndrome (ACS) patients. An analysis of the ACUITY study using propensity score, and including patients with multivessel disease and moderate and high risk ACS, showed that those treated with PCI had a lower rate of periprocedural stroke, AMI, major bleeding and kidney injury, with a similar long-term mortality rate, infarction and major cardiovascular events (MACE). (23)
When the patient presents a condition of hemodynamic instability, refractory angina or refractory arrhythmias, the best solution appears to be fast stabilization, as less complex as possible. In the absence of randomized studies in this scenario, PCI seems the best tool to obtain a satisfactory outcome. (26-28)
In cases in which, from a technical point of view, PCI is not a suitable stabilization tool, intra-aortic balloon pump counterpulsation, balloon PCI, intravenous anticoagulant and antiplatelet treatment, as well as use of negative chronotropic drugs and vasodilators, are tools available to minimize the risk of events until the patients reaches CABG.
How does diabetes influence in the decision?
Diabetes is one of the factors more strongly associated with the genesis of atherosclerotic disease, producing extensive, diffuse and rapidly progressive vascular disease. The FREEDOM trial, published in 2021, was one of the most important studies comparing revascularization strategies in diabetic patients. It enrolled 1900 patients (953 in the PCI group and 947 in the CABG group) with a primary endpoint of MACE (total mortality, AMI, and stroke) and 2- and 5-year follow-up. (7)
A statistically significant difference was observed at 5 years in favor of CABG (PCI 26.6% vs. CABG 18.7%, p=0.005) at the expense of all-cause death (16.3% vs. 10.9%) and AMI (13.9% vs. 6.0 %) with a slight increase in stroke events in the CABG group without statistical significance. A higher rate of revascularization was observed in the PCI group during the first 12 months of follow-up. No changes in the benefits of surgical revascularization were found with or without insulin use, but total mortality was higher in insulin users regardless the strategy employed.
In the 5-year follow-up of the SYNTAX study, a higher rate of events was obtained in the subgroup of diabetic patients; however, a statistically significant difference was only encountered when comparing the rate of new revascularization. (6)
When assessed according to the SYNTAX score, this difference was evident in the group with diabetes even with a low score.
In an observational study published in 2016 based on data from the Swedish SWEDEHEART registry, a significant increase in mortality was observed as glycosylated hemoglobin levels increased. (24)
Diabetes appears to affect more negatively PCI than CABG results, a situation that is magnified the worse the metabolic control or the complexity of the disease.
How does the presence of lung disease influence in the decision?
Chronic obstructive pulmonary disease (COPD) is a common comorbidity in patients with coronary heart disease due to the direct relationship between smoking and these last 2 entities. An increase in the event rate during follow-up in revascularized patients has been reported in multiple prospective studies, showing increased COPD-related complications linked to respiratory assistance, with the consequent prolongation of hospitalizations.
A substudy of the extended SYNTAX study (SYNTAXES) evaluated the 30-day event rate, 3-year MACCE and 10-year total mortality in patients diagnosed with COPD (n=154, 8.7%). A significant increase in 10-year mortality was observed in patients with COPD, regardless of the revascularization strategy (43.1% in COPD vs. 24.9% in non-COPD, p < 0.001). In multivariate analysis, COPD was an independent predictor of mortality in patients with CABG, but not in the PCI group. (25)
We should not consider COPD as a dichotomous variable but as a continuum, as can be seen in an observational study published by Fuster et al. in 2006. This study observed that in-hospital mortality after CABG in patients with COPD was directly related to FEV1, with higher mortality in those with values below the expected 60%, compared with those with higher values. (26)
COPD was regarded as a continuous variable, divided into 4 categories according to severity adjusted by FEV1 levels (normal, with FEV1>80%, mild with FEV1 60-80%, moderate with FEV1 40-59% and severe with values < 40%). The difference in terms of increased mortality started to be significant in the moderate group.
What is the influence of the patient's age?
Age is one of the main determining risk factors in coronary heart disease. When making decisions regarding revascularization strategies, not only the greater anatomical complexity resulting from the higher number of years of exposure should be assessed, but also the comorbidities associated with advanced age. The effects of age at the level of global cardiovascular function are manifested both directly in the myocardium, with changes at the molecular, ionic and biomechanical level (greater rigidity of the cardiac muscle and lower contractile reserve), and at the vascular level, with molecular changes that promote fibrosis and the replacement of smooth muscle fibers due to increased collagen deposition, as well as changes in molecular signaling with lower production of nitric oxide and greater availability of endothelin-1. (27)
All these phenomena, a product of physiological aging added to a longer period of exposure to coronary atherosclerosis, promote an environment with a higher rate of complications and morbidity and mortality. Thus, it is necessary to evaluate the advantages and disadvantages of the therapeutic strategies available in this group of patients.
In a substudy of the SYNTAXES extension registry, a subgroup analysis was performed in the population >70 years of age (n= 575, 31.9%); 290 randomly assigned to PCI and 285 to CABG). No statistically significant differences were observed in total mortality at 10 years in PCI vs. CABG (44.0% vs. 41.5%) as well as in MACCE at 5 years (39.4% vs. 35.1%). (27)
This can be explained because older patients per se have a higher rate of events due to their greater anatomical complexity, coupled with an increased susceptibility to non-cardiovascular complications.
What is the influence of surgical risk and frailty?
The risk of periprocedural events may minimize the mid- and long-term benefits of CABG. There are different scores that can identify this risk, but it is always advisable to evaluate each of the variables that influence the rate of in-hospital complications and the ease of outpatient recovery from the surgical procedure. Frailty is a variable incorporated in the last 5 years. A fragile phenotype represents a decrease in physiological reserve and greater vulnerability. It reflects higher biological age and has therefore become a substantial factor in the evaluation of various special medical situations and integrated into clinical decision making. (28-30)
How does kidney function influence in the decision?
Chronic kidney disease (CKD) is common in patients with coronary heart disease, due to the multiple shared pathophysiological mechanisms involved in the genesis of both diseases. In addition to this, concomitant kidney disease plays a fundamental role when assessing risks and benefits in terms of ischemic and hemorrhagic profiles in the different clinical scenarios we face.
In a subanalysis at 5 years of the SYNTAX database, mortality was significantly higher in patients with CKD compared with patients with normal renal function after PCI (26.7% vs. 10.8%, p < 0.001) and CABG (21.2% vs. 10.6%, p=0.005). (31)
When comparing PCI with CABG, a significant interaction with renal function was found for death (p int=0.017), but not for the composite of death/stroke/AMI (p int=0.070) or MACCE (p int=0.15). In patients with CKD, the rate of MACCE was significantly higher after PCI compared with CABG (42.1% vs. 31.5%, p=0.019), driven by repeat revascularization (21.9% vs. vs. 8.9%, p=0.004) and all-cause death (26.7% vs. 21.2%, p=0.14). In patients with CKD who also had diabetes, PCI vs. CABG was significantly worse in terms of death/stroke/AMI (47.9% vs. 24.4%, p=0.005) and all-cause mortality (40 .9% vs. 17.7%, p=0.004).
These differences could be related to the fact that CKD patients have a higher risk of thrombotic events with PCI as a result of different hemostatic properties, severe atherosclerosis, and limitation in the use of potent antiplatelet agents. The long-term benefits of surgery must be balanced with the risk of worsening renal function, with variables such as contrast volume, use of extracorporeal circulation, and hemodynamic instability. (32-34)
What is the influence of bleeding risk?
A chronically high bleeding risk determines the possibility of patients receiving prolonged potent antiplatelet therapy without complications. Patients undergoing complex angioplasties have a considerable risk of in-stent thrombosis, and therefore benefit from this treatment. When this situation is associated with the presence of atrial fibrillation or another indication for the use of oral anticoagulation, short triple therapy and dual treatments (antiplatelet + anticoagulant) for a reasonable period of time should be considered. (35) Facing a high bleeding risk, which does not guarantee the possibility of using some of these schemes, the risk of continuing with PCI increases significantly and the option of CABG should be evaluated.
How do patient's wishes influence in the decision?
When defining reperfusion strategies in coronary patients with multivessel disease, clinical and anatomical conditions are not the only variables to be considered when deciding a treatment.
Historically, the relationship between doctor and patient was framed within an asymmetry of knowledge with a paternalistic model where the physician decided over the interests and expectations of the patient which was the best therapeutic strategy. Although this method does not present conflicts for the physician, it can generate conflicts for the patient. On multiple occasions, the adverse effects or the outcome do not end up being satisfactory for the patient and result in therapeutic failure.
Since 1982 strategies have been designed to improve this type of conflicts, considering both the expectations of the physician and the patient, which is called “shared decision making.”
The first publications by Charles et al. refer to the phrase “It Takes at Least Two to Tango”, and they set out the main premises of this strategy: 1) Exchange of information: the physician must clearly present the available strategies, expressing their advantages and disadvantages, while the patient must explain individual preferences and values with respect to his/her expectations from the treatment; 2) Deliberation: both participants (physician and patient) openly discuss the available options; 3) Decision: both participants jointly make the final decision on what treatment to perform. (36)
Subsequently, multiple models on how to carry out the shared decision-making strategy were put into practice, with slight differences from the initial premise, although all of them respected these 3 elements.
Therefore, it is defined that in this type of decisions, 3 aspects must always be considered: the scientific evidence of the different therapeutic options, the expectations and values of the patient and the context in which both the patient and the physician develop.
In our specific case on revascularization strategies in patients with multivessel coronary artery disease, various articles have been published using information tools for the patient both before and at the time of consultation, in interactive digital format or on paper, which are called “Decision AIDS”. (37-38
)
When patient preferences and values are discussed in conjunction with medical evidence at the time of choosing revascularization strategies, we can observe that there is a clear difference in the severity weighting of the different adverse events in the therapeutic options offered (CABG vs. PCI). In this way we can clearly see that our opinion regarding the benefits for the patient should never be taken as correct without first sharing it with him/her.
Magliano et al. evaluated the differences in the weight of adverse events in 56 patients hospitalized for coronary artery disease prior to revascularization, and 22 cardiologists specialized in ischemic heart disease. A list of the 14 most frequent adverse events was made and they were weighted from 1 to 14 by patients and physicians. These are listed below: 1) atrial fibrillation; 2) heart failure; 3) incision scar; 4) hospital stay 5) long-term survival; 6) AMI; 7) periprocedural death 8) post-surgical infection; 9) post-operative angina; 10) pseudoaneurysm; 11) kidney failure; 12) reoperation; 13) new PCI; and 14) stroke. It was observed that, in the group of patients, the adverse event weighted as the most important was acute kidney failure, followed by peri-procedural death, stroke and then reoperation and AMI, whereas in the group of cardiologists the weighting was as follows: peri-procedural death, longterm survival, stroke, AMI and heart failure.
CONCLUSIONS
When evaluating the revascularization strategy to choose, we must take into account multiple factors derived from the patient's characteristics, including coronary anatomy, baseline conditions, hemorrhagic risk, comorbidities, preexisting diseases and wishes, with the aim of achieving the most individualized decision possible that provides more satisfactory results.
That is why we have published this review considering, in our opinion, the most influential and relevant factors in the choice of a coronary revascularization strategy. A summary was made of each of them, and we analyzed the most recent evidence in each of the aspects discussed.
https://creativecommons.org/licenses/by-nc-sa/4.0/

Conflicts of interest
None declared.
(See authors' conflict of interests forms on the web/Additional material).
©Revista Argentina de Cardiología
REFERENCES
- Fearon WF, Zimmermann FM, De Bruyne B, Piroth Z, van Straten AHM, Szekely L, et al; FAME 3 Investigators. Fractional Flow Reserve- Guided PCI as Compared with Coronary Bypass Surgery. N Engl J Med. 2022;386:128-37. https://doi.org/10.1056/NEJMoa2112299
- Serruys PW, Morice MC, Kappetein AP, Colombo A, Holmes DR, Mack MJ, et al; SYNTAX Investigators. Percutaneous coronary intervention versus coronary-artery bypass grafting for severe coronary artery disease. N Engl J Med. 2009;360:961-72. https://doi.org/10.1056/NEJMoa0804626
- Farkouh ME, Domanski M, Sleeper LA, Siami FS, Dangas G,Mack M, et al; FREEDOM Trial Investigators. Strategies for multivessel revascularization in patients with diabetes. N Engl J Med. 2012;367:2375-84. https://doi.org/10.1056/NEJMoa1211585
- Kapur A, Hall RJ, Malik IS, Qureshi AC, Butts J, de Belder M, et al. Randomized comparison of percutaneous coronary intervention with coronary artery bypass grafting in diabetic patients. 1-year results of the CARDia (Coronary Artery Revascularization in Diabetes) trial. J Am Coll Cardiol. 2010;55:432-40. https://doi.org/10.1016/j.jacc.2009.10.014
- Kamalesh M, Sharp TG, Tang XC, Shunk K, Ward HB, Walsh J, et al; VA CARDS Investigators. Percutaneous coronary intervention versus coronary bypass surgery in United States veterans with diabetes. J Am Coll Cardiol. 2013;61:808-16. https://doi.org/10.1016/j.jacc.2012.11.044
- Head SJ, Davierwala PM, Serruys PW, Redwood SR, Colombo A, Mack MJ, et al. Coronary artery bypass grafting vs. percutaneous coronary intervention for patients with three-vessel disease: final five-year follow-up of the SYNTAX trial. Eur Heart J. 2014;35:2821- 30. https://doi.org/10.1093/eurheartj/ehu213
- Farkouh ME, Domanski M, Dangas GD, Godoy LC, Mack MJ, Siami FS, et al; FREEDOM Follow-On Study Investigators. Long-Term Survival Following Multivessel Revascularization in Patients With Diabetes: The FREEDOM Follow-On Study. J Am Coll Cardiol. 2019;73:629-38. https://doi.org/10.1016/j.jacc.2018.11.001
- Kapur A, Bartolini D, Finlay MC, Qureshi AC, Flather M, Strange JW, et al. The Bypass Angioplasty Revascularization in Type 1 and Type 2 Diabetes Study: 5-year follow-up of revascularization with percutaneous coronary intervention versus coronary artery bypass grafting in diabetic patients with multivessel disease. J Cardiovasc Med (Hagerstown). 2010 ;11:26-33. https://doi.org/10.2459/JCM.0b013e328330ea32
- Banning AP, Serruys P, De Maria GL, Ryan N, Walsh S, Gonzalo N, et al. Five-year outcomes after state-of-the-art percutaneous coronary revascularization in patients with de novo three-vessel disease: final results of the SYNTAX II study. Eur Heart J. 2022;43:1307-16. https://doi.org/10.1093/eurheartj/ehab703
- Serruys PW, Onuma Y, Garg S, Sarno G, van den Brand M, Kappetein AP, et al. Assessment of the SYNTAX score in the Syntax study. EuroIntervention. 2009;5:50-6. https://doi.org/10.4244/eijv5i1a9
- Serruys PW, Morice MC, Kappetein AP, Colombo A, Holmes DR, Mack MJ, et al. SYNTAX Investigators. Percutaneous coronary intervention versus coronary-artery bypass grafting for severe coronary artery disease. N Engl J Med. 2009;360:961-72. https://doi.org/10.1056/NEJMoa0804626
- Thuijs DJFM, Kappetein AP, Serruys PW, Mohr FW, Morice MC, Mack MJ, et al; SYNTAX Extended Survival Investigators. Percutaneous coronary intervention versus coronary artery bypass grafting in patients with three-vessel or left main coronary artery disease: 10-year follow-up of the multicentre randomised controlled SYNTAX trial. Lancet. 2019;394:1325-34. https://doi.org/10.1016/S0140-6736(19)31997-X
- Mohr FW, Morice MC, Kappetein AP, Feldman TE, Ståhle E, Colombo A, et al. Coronary artery bypass graft surgery versus percutaneous coronary intervention in patients with three-vessel disease and left main coronary disease: 5-year follow-up of the randomised, clinical SYNTAX trial. Lancet. 2013;381:629-38. https://doi.org/10.1016/S0140-6736(13)60141-5
- Melina G, Angeloni E, Refice S, Benegiamo C, Lechiancole A, Matteucci M, et al. Residual SYNTAX score following coronary artery bypass grafting. Eur J Cardiothorac Surg. 2017;51:547-53. https://doi.org/10.1093/ejcts/ezw356
- Li X, Ni S. Could Residual SYNTAX Score Predict the Prognosis of Patients With Cardiogenic Shock? J Am Coll Cardiol. 2021;77:2872. https://doi.org/10.1016/j.jacc.2021.02.069
- Burgess SN, Juergens CP, Mussap CJ, Lo STH, French JK. Cardiogenic Shock, the Residual SYNTAX Score, and Prognosis: Corroborative “Real-World” Data. J Am Coll Cardiol. 20218;77:2871-2. https://doi.org/10.1016/j.jacc.2021.02.068
- Stone GW, Ali ZA, O’Brien SM, Rhodes G, Genereux P, Bangalore S, et al; ISCHEMIA Research Group. Impact of Complete Revascularization in the ISCHEMIA Trial. J Am Coll Cardiol. 2023;82:1175-88. https://doi.org/10.1016/j.jacc.2023.06.015
- Sandoval Y, Brilakis ES, Canoniero M, Yannopoulos D, Garcia S. Complete versus incomplete coronary revascularization of patients with multivessel coronary artery disease. Curr Treat Options Cardiovasc Med. 2015;17:366. https://doi.org/10.1007/s11936-015-0366-1
- Velazquez EJ, Lee KL, Jones RH, Al-Khalidi HR, Hill JA, Panza JA, et al; STICHES Investigators. Coronary-Artery Bypass Surgery in Patients with Ischemic Cardiomyopathy. N Engl J Med. 2016;374:1511-20. https://doi.org/10.1056/NEJMoa1602001
- Petrie MC, Jhund PS, She L, Adlbrecht C, Doenst T, Panza JA, et al; STICH Trial Investigators. Ten-Year Outcomes After Coronary Artery Bypass Grafting According to Age in Patients With Heart Failure and Left Ventricular Systolic Dysfunction: An Analysis of the Extended Follow-Up of the STICH Trial (Surgical Treatment for Ischemic Heart Failure). Circulation. 2016 ;134:1314-24. https://doi.org/10.1161/CIRCULATIONAHA.116.024800
- Perera D, Clayton T, O’Kane PD, Greenwood JP, Weerackody R, Ryan M, et al; REVIVED-BCIS2 Investigators. Percutaneous Revascularization for Ischemic Left Ventricular Dysfunction. N Engl J Med. 2022;387:1351-60. https://doi.org/10.1056/NEJMoa2206606
- Sun LY, Gaudino M, Chen RJ, Bader Eddeen A, Ruel M. Longterm Outcomes in Patients With Severely Reduced Left Ventricular Ejection Fraction Undergoing Percutaneous Coronary Intervention vs Coronary Artery Bypass Grafting. JAMA Cardiol. 2020;5:631-41. https://doi.org/10.1001/jamacardio.2020.0239
- Ben-Gal Y, Moses JW, Mehran R, Lansky AJ, Weisz G, Nikolsky E, et al. Surgical versus percutaneous revascularization for multivessel disease in patients with acute coronary syndromes: analysis from the ACUITY (Acute Catheterization and Urgent Intervention Triage Strategy) trial. JACC Cardiovasc Interv. 2010;3:1059-67. https://doi.org/10.1016/j.jcin.2010.06.017
- Kuhl J, Sartipy U, Eliasson B, Nyström T, Holzmann MJ. Relationship between preoperative hemoglobin A1c levels and long-term mortality after coronary artery bypass grafting in patients with type 2 diabetes mellitus. Int J Cardiol. 2016;202:291-6. https://doi.org/10.1016/j.ijcard.2015.09.008
- Wang R, Tomaniak M, Takahashi K, Gao C, Kawashima H, Hara H, et al. Impact of chronic obstructive pulmonary disease on 10-year mortality after percutaneous coronary intervention and bypass surgery for complex coronary artery disease: insights from the SYNTAX Extended Survival study. Clin Res Cardiol. 2021;110:1083-95. https://doi.org/10.1007/s00392-021-01833-y
- Fuster RG, Argudo JA, Albarova OG, Sos FH, López SC, Codoñer MB, et al. Prognostic value of chronic obstructive pulmonary disease in coronary artery bypass grafting. Eur J Cardiothorac Surg. 2006;29:202-9. https://doi.org/10.1016/j.ejcts.2005.11.015
- Ono M, Serruys PW, Hara H, Kawashima H, Gao C, Wang R, et al; SYNTAX Extended Survival Investigators. 10-Year Follow-Up After Revascularization in Elderly Patients With Complex Coronary Artery Disease. J Am Coll Cardiol. 2021;77:2761-73. https://doi.org/10.1016/j.jacc.2021.04.016
- Tse G, Gong M, Nunez J, Sanchis J, Li G, Ali-Hasan-Al-Saegh S, et al; International Health Informatics Study (IHIS) Network. Frailty and Mortality Outcomes After Percutaneous Coronary Intervention: A Systematic Review and Meta-Analysis. J Am Med Dir Assoc. 2017;18:1097. e1-1097.e10. https://doi.org/10.1016/j.jamda.2017.09.002
- Schulman-Marcus J, Peterson K, Banerjee R, Samy S, Yager N. Coronary Revascularization in High-Risk Stable Patients With Significant Comorbidities: Challenges in Decision-Making. Curr Treat Options Cardiovasc Med. 2019;21:5. https://doi.org/10.1007/s11936-019-0706-7
- Kozlov KL, Bogachev AA. Coronary revascularization in the elderly with stable angina. J Geriatr Cardiol. 2015;12:555-68. https://doi.org/10.11909/j.issn.1671-5411.2015.05.017
- Milojevic M, Head SJ, Mack MJ, Mohr FW, Morice MC, Dawkins KD, et al. The impact of chronic kidney disease on outcomes following percutaneous coronary intervention versus coronary artery bypass grafting in patients with complex coronary artery disease: five-year follow-up of the SYNTAX trial. EuroIntervention. 2018;14:102-11. https://doi.org/10.4244/EIJ-D-17-00620
- Sarnak MJ, Amann K, Bangalore S, Cavalcante JL, Charytan DM, Craig JC, et al; Conference Participants. Chronic Kidney Disease and Coronary Artery Disease: JACC State-of-the-Art Review. J Am Coll Cardiol. 2019;74:1823-38. https://doi.org/10.1016/j.jacc.2019.08.1017
- Costanzo P, Džavík V. Coronary Revascularization in Patients With Advanced Chronic Kidney Disease. Can J Cardiol. 2019;35:1002-14. https://doi.org/10.1016/j.cjca.2019.02.026
- Klein EC, Kapoor R, Lewandowski D, Mason PJ. Revascularization Strategies in Patients with Chronic Kidney Disease and Acute Coronary Syndromes. Curr Cardiol Rep. 2019;21:113. https://doi.org/10.1007/s11886-019-1213-x
- Iijima R, Nakamura M. Optimal Revascularization Strategy for Acute Coronary Syndromes With High Bleeding Risk - It Is Hard to Please All Parties. Circ J. 2021;85:1942-3. https://doi.org/10.1253/circj.CJ-21-0334
- Charles C, Gafni A, Whelan T. Shared decision-making in the medical encounter: what does it mean? (or it takes at least two to tango). Soc Sci Med. 1997;44:681-92. https://doi.org/10.1016/s0277-9536(96)00221-3
- Magliano CADS, Monteiro AL, de Oliveira Rebelo AR, de Aguiar Pereira CC. Patients’ preferences for coronary revascularization: a systematic review. Patient Prefer Adherence. 2018 ;13:29-35. https://doi.org/10.2147/PPA.S188268
- Hughes TM, Merath K, Chen Q, Sun S, Palmer E, Idrees JJ, et al. Association of shared decision-making on patient-reported health outcomes and healthcare utilization. Am J Surg. 2018;216:7-12. https://doi.org/10.1016/j.amjsurg.2018.01.011