Ricardo D. Olano1 , Walter G. Espeche1 , Betty C. Leiva Sisnieguez1 , Patricia M. Carrera Ramos1 , Camilo Martinez1 , Carlos E. Leiva Sisnieguez1 , Ana De Iraola2 , Daniela R. Gomez2 , Julián Minetto1 , Martín R. Salazar
1Cardiometabolic Unit, Hospital San Martín de La Plata. Buenos Aires, Argentina
2Cardiology Division, Hospital San Martín de La Plata. Buenos Aires, Argentina.
Address for correspondence: Ricardo D. Olano - Unidad Cardiometabólica, Hospital San Martín de La Plata. Argentina. 1 y 70 - B1900, La Plata, Provincia de Buenos Aires, Argentina - E-mail: olanodaniel@gmail.com
Rev Argent Cardiol 2023;91:330-335. http://dx.doi.org/10.7775/rac.v91.i5.20675
SEE RELATED ARTICLE: Rev Argent Cardiol 2023:91:307-308. http://dx.doi.org/10.7775/rac.v91.i5.20664
ABSTRACT
Background: Preeclampsia (PE) is the main cause of maternal-fetal morbidity and mortality in our country. Early hemodynamic changes during pregnancy could predict progression to PE. Machine learning (ML) enables the discovery of hidden patterns that could early detect PE development.
Objectives: The aim of this study was to build a classification tree with non-invasive hemodynamic variables for the early prediction of PE occurrence.
Results: Seventeen patients (15.18%) presented PE. A predictive classification tree was generated with arterial compliance index (ACI), cardiac index (CI), cardiac work index (CWI), ejective time ratio (ETR), and Heather index (HI). A total of 93.75% patients were correctly classified (Kappa 0.70, positive predictive value 0.94 and negative predictive value 0.35; accuracy 0.94, and area under the ROC curve 0.93).
Conclusion: ACI, CI, CWI, ETR and HI variables predicted the early development of PE in our sample with excellent discrimination and accuracy, non-invasively, safely and at low cost.
Key words: Machine Learning, Preeclampsia, Non-invasive Hemodynamics, Impedance Cardiography
RESUMEN
Introducción: la preeclampsia (PE) es la principal causa de morbimortalidad materno-fetal en nuestro país. Alteraciones hemodinámicas precoces durante el embarazo podrían predecir la evolución a PE. El machine learning (ML) permite el hallazgo de patrones ocultos que podrían detectar precozmente el desarrollo de PE.
Objetivos: desarrollar un árbol de clasificación con variables de hemodinamia no invasiva para predecir precozmente desarrollo de PE.
Material y métodos: estudio observacional prospectivo con embarazadas de alto riesgo (n=1155) derivadas del servicio de Obstetricia desde enero 2016 a octubre 2022 para el muestreo de entrenamiento por ML con árbol de clasificación j48. Se seleccionaron 112 embarazadas entre semanas 10 a 16, sin tratamiento farmacológico y que completaron el seguimiento con el término de su embarazo con evento final combinado (PE): preeclampsia, eclampsia y síndrome HELLP. Se evaluaron simultáneamente con cardiografía de impedancia y velocidad de onda del pulso y con monitoreo ambulatorio de presión arterial de 24 hs (MAPA).
Resultados: presentaron PE 17 pacientes (15,18%). Se generó un árbol de clasificación predictivo con las siguientes variables: índice de complacencia arterial (ICA), índice cardíaco (IC), índice de trabajo sistólico (ITS), cociente de tiempos eyectivos (CTE), índice de Heather (IH). Se clasificaron correctamente el 93,75%; coeficiente Kappa 0,70, valor predictivo positivo (VPP) 0,94 y negativo (VPN) 0,35. Precisión 0,94, área bajo la curva ROC 0,93.
Conclusión: las variables ICA, IC, ITS, CTE e IH predijeron en nuestra muestra el desarrollo de PE con excelente discriminación y precisión, de forma precoz, no invasiva, segura y con bajo costo.
Palabras clave: Aprendizaje automático, Preeclampsia, Hemodinamia no invasiva, Cardiografía de impedancia
Received: 08/26/2023
Accepted: 09/15/2023
INTRODUCCIÓN
Preeclampsia (PE) is one of the main causes of worldwide perinatal morbidity and mortality, and hypertensive disorders of pregnancy (HDP) are generally responsible for frequent hospitalizations, maternal-fetal morbidity and preterm birth, affecting approximately 10% of all pregnancies. (1)
In addition, PE is associated with numerous disorders in the offspring of mothers with PE, mainly hypertension, diabetes and obesity. (2)
The adverse results of PE can be prevented with the early use of acetyl salicylic acid (ASA) as the most efficient therapeutic strategy. Randomized studies including more than 60 000 pregnant women showed that aspirin administered at an adequate dose (≥150 mg/day) and initiated before 16 weeks of pregnancy could prevent most PE cases, especially those of early initiation and those of maternal and fetal severe disease. However, ASA treatment after this week of gestation has not shown maternal-fetal benefits. Therefore, there is urgent need of identifying women with risk of developing PE who could benefit from early ASA prophylaxis at the beginning of pregnancy. (3)
The hemodynamic adaptations during normal pregnancy allow fulfilling the greater metabolic demands to satisfy fetal requirements (4,5)
In the first and third trimester maternal physiological hemodynamics is similar to that of a non-pregnant woman. (6,7)
However, in the second trimester, the metabolic demand decreases blood pressure (BP) as a consequence of vascular dilation, increasing cardiac output (CO) due to enhanced heart rate and stroke volume, with a decrease in peripheral vascular resistance (PR), generating a hyperdynamic phenotype. (8)
Conversely, if this hemodynamic pattern presents in the first trimester, the risk of developing PE increases. Thus, the detection of inadequate behavior in the hemodynamic patterns at the beginning of pregnancy could prematurely predict PE development to optimize the therapeutic opportunity with ASA.
The gold standard to evaluate CO and PR is thermodilution assessment with a Swan-Ganz catheter (invasive hemodynamics), which is not applicable in “healthy” individuals and even less in pregnant women as routine complementary methods. Echocardiography has been used to identify cardiovascular adaptations during pregnancy. However, measurements require a long time, are demanding from a technical point of view and depend on the investigator. (9)
We conducted a cross-sectional, descriptive, and multicenter study in five outpatient cardiology centers in the Autonomous City of Buenos Aires and Great Buenos Aires, between July and November 2022. The population was made up of consecutive patients between 45 and 80 years (age range that allows calculation of residual risk with the SMART-REACH model) with a history of cardiovascular disease (coronary artery disease, peripheral artery disease or cerebrovascular disease). For the present study, patients who did not meet the LDL-C goal recommended by current guidelines ( < 55 mg/dL) were selected. (8) The medical records of the patients included were reviewed, obtaining information on their history, cardiovascular risk factors, and medication received.
Serum levels of glucose, total cholesterol, high-density lipoprotein cholesterol (HDL-C), triglycerides, C-reactive protein, lipoprotein(a) [Lp(a)] and creatinine were measured according to standard methods. LDL-C was calculated using the Friedewald formula. (9) Glomerular filtration rate was estimated according to the CKD-EPI (chronic kidney disease-epidemiology collaboration) equation. (10)
The 10-year and lifetime risks of recurrent events (MI, stroke, or vascular death) were estimated using the SMARTREACH model. (7)
Subsequently, a simulation model was used to further optimize the lipid-lowering treatment in each patient, estimating the decrease in LDL-C and verifying if the recommended lipid goal was reached or not. Based on the recommendations of the latest Consensus on Cardiovascular Prevention of the Argentine Society of Cardiology for secondary prevention of very high-risk patients who do not reach LDL-C goal, (11) we considered the following order for optimizing lipid-lowering treatment: 1) raise statins to maximum dose, using high-intensity statins (atorvastatin 80 mg/day or rosuvastatin 40 mg/day). The choice between the two regimens was based on previous treatment (raising the dose of the statin the patient was already receiving). If, on the other hand, the patient was receiving another statin, we arbitrarily chose to administer atorvastatin 80 mg/day; 2) add ezetimibe if LDL-C goal was not met despite treatment with statins; 3) add a proprotein convertase subtilisin/kexin type 9 inhibitor (PCSK9i) when despite treatment with statins and ezetimibe, LDL-C threshold was ⩾ 70 mg/dL. The simulation model considered no more than three steps in the process for optimizing lipid-lowering therapy (one strategy at a time). Some patients required a single step to optimize treatment (following the order previously mentioned), while others required two or three steps.
Finally, the 10-year and lifetime residual cardiovascular risk reduction with the corresponding NNT were calculated for each necessary optimizing step to achieve LDL-C goal. These results were automatically calculated by the SMARTREACH model with each treatment change for each patient.
For this reason, non-invasive hemodynamics evaluated by impedance cardiography (ICG) has gained relevance, especially in the evaluation of pregnant women. (10-12)
Classically, the hemodynamic phenotypes of pregnancy have been defined by the behavior of two variables: cardiac index (CI) and PR, (6,7) but the information of circulatory physiology is not limited to only these two hemodynamic variables, and hidden phenotypes could be identified with non-invasive techniques considering other variables which can also be assessed with ICG. (13)
Artificial intelligence (AI) is widely used in the medical field and automatic learning, and has the potential of revolutionizing medical care, both in the general population and pregnant women, (14)
as it could provide earlier and more accurate diagnoses, relieve the physicians’ workload, reduce costs and deliver reference analyses to be interpreted by the different specialists acting during pregnancy. (15,16)
AI with machine learning (ML) could be the key to unblock the potential of ICG and carotid-femoral pulse wave velocity (cfPWV) evaluation in pregnancy, (17)
and would allow the addition of non-invasive hemodynamic markers, beyond the classically described CI and PR in gestational hemodynamics. This would provide relevant additional information to the results at the beginning of pregnancy and could also be used as a predictive tool of associated diseases, generating a clinically relevant PE predictive algorithm to optimize the indication of ASA from the onset. (18)
Therefore, the objectives of the present study were: 1. To generate a model, using AI, of non-invasive hemodynamic phenotypic patterns for the prediction of PE in the first trimester of high-risk pregnant women, and 2. To evaluate hidden patterns beyond PVR and CO, which will consider other hemodynamic variables (flow, resistance, contractility, fluid, systolic function, left ventricular energy efficiency and arterial distensibility) in PE prediction.
METHODS
High-risk pregnant women with 10 to16-week of gestation were consecutively referred from the Obstetrics Division (Hospital San Martín, La Plata) to the Cardiometabolic Diseases Unit for BP monitoring. A pre-established BP evaluation protocol and non-invasive hemodynamics was carried out in women who had completed their pregnancy, from January 2016 to October 2022. The endpoint was the composite of PE, eclampsia or HELLP syndrome. Pregnant women under pharmacological antihypertensive treatment, previous valve diseases, congenital heart diseases, atrial fibrillation and/or dialysis were excluded from the study.
Impedance cardiography and carotid-femoral PWV (Zlogic 1.20b, Aortic 3.10, Exxer, Argentina) were performed and 24-hour ambulatory BP monitoring (ABPM) (Spacelabs 90207, USA) was fitted on the same day. (19) Impedance cardiography was carried out with the patient in dorsal decubitus position and at 3 minutes of adaptation in standing position, acquiring an average of at least 10 consecutive beats, with simultaneous carotidogram channel to solve any ambiguity between point B and point X. (11) The following hemodynamic parameters were measured: of flow: stroke volume index (SVI) (mL/beat/m2), CI (L/min/m2); of afterload: vascular resistance index (VRI) (dyn*s*cm-5), arterial compliance index (ACI) (mL/mmHg/m2); of contractility: velocity index (VI) ((1000*sec)-1), cardiac acceleration index (CAI) ((100*sec2)-1), systolic time ratio (STR) (%), ejective time ratio (ETR) (%), cardiac work index (CWI) (kg*m/m2), Heather index (HI) ohm/s2); of thoracic fluid level: thoracic fluid content (TFC) (Kohms-1); of cardiovascular performance: arterial elastance (Ea) (mmHg/mL), end-systolic ventricular elastance (Ees) (mmHg/mL), ventriculo-arterial coupling (VAC); and of arterial distensibility: cfPWV (m/sec).
The latter was measured immediately after the ICG study with applanation tonometry in the left carotid and femoral arteries, with pedometer-assessed carotid-femoral distance and average of three consecutive recordings with at least 10 beats in each recording. Twenty-four-hour ABPM was carried out defining daytime and nighttime periods according to the patient’s diary. Studies were considered valid with >70 % recordings and at least 1 per hour.
Statistical analysis
Data were collected with relational database management software (Filemaker 12, 0v1, USA), which allowed exporting data of interest to a Microsoft Excel 2010 chart to be analyzed by descriptive statistics with MedCalc™ 22.009 (Belgium). Results were expressed as mean, standard deviation (SD), standard error (SE) and confidence interval (95% CI), or median and 95% CI, according to the D’Agostino-Pearson normality test. Unpaired Student’s t test or unpaired Mann- Whitney test, according to normal or non-normal distribution, were used for comparisons between groups. Qualitative variables were expressed as percentages and the chi-square test or Fisher’s exact test was used to compare groups, as appropriate. Statistical significance was established at p < 0.05. A j48 classification tree (Weka 3.8.6. Waikato University, New Zeland) was used for automatic learning training sampling. The classification instances were evaluated with Cohen’s Kappa index as goodness of fit of the complete model, assuming a value ≥0.7 to consider that the model was appropriate. Mean absolute error was used to indicate the estimated error, in which lower values signify a more correct model. The area under the ROC curve assessed the model’s capacity to detect PE events, and its accuracy was described as percentage of well-classified instances.
Ethical considerations
All participants provided their oral and written informed consent to participate in the study, which was approved by the Research Ethics Committee of Hospital San Martín of La Plata (HSMLP2022/0079)
Sponsor
This work is part of the research protocol: “Machine learning to predict preeclampsia by non-invasive hemodynamics in high-risk pregnancy” which received financial support from the Ministry of Health of the Buenos Aires Province, through the Scholarship Program to Julieta Lanteri, awarded from the Research and Technical Cooperation Direction of the “Floreal Ferrara” Health Government School Provincial Direction. The study was registered in the Joint Health Research Commission (CCIS) (EX-2023-20132598-GDEBACCISMSALGP).
RESULTS
Among a total of 1155 high-risk pregnant women registered with non-invasive vascular studies, 112 were included to model automatic learning after selection and exclusion criteria, 17 with PE (15.18%). Mean age was 30 years, without difference between groups with and without PE. Pregnancy termination was premature, with a median of 35 weeks in pregnant women with PE and 38 in those without PE (p=0.007). In patients with PE, the frequency of early PE was low (n= 3, 17.64%), and late in most cases (n=14, 82.35%). Blood pressure in 24-h ABMP did not present differences between groups in the 24-hour, daytime, nighttime and without diagnosis of hypertension averages (Table 1). The hemodynamic variables did not show significant differences between the two groups, although there was a tendency to greater CWI (p=0.06) and HI (p=0.05), which are variables related to left ventricular contractility, in patients with PE. Also, afterload, evaluated with VRI (p=0.05) and TFC (p=0.02) was higher in those with PE (Table 2). Arterial stiffness assessed by cfPWV showed no differences. The composite endpoint (n=17) generated a supervised algorithm by classification tree where a predictive pattern with 5 non-invasive hemodynamic variables was detected (Figure 1). The main node was ACI and secondary nodes were CI, CWI, ETR and HI. It was seen that ETR and HI, which had differences between PE and non-PE, were selected by the automatic learning algorithm. However, TFC was excluded from the algorithm because it did not add predictive capacity and to optimize modeling dimensionality. From a total of 112 instances with training model, 93.75% were correctly classified. The model was adequate with 0.70 Kappa, low mean absolute value of 0.23, high positive predictive value (PPV) of 0.94, and low negative predictive value (NPV) of 0.35. The capacity of the model to detect PE predictive hemodynamic variables was high, with an area under the ROC curve of 0.93.
Table 1. Age, weeks of pregnancy and average daytime, nighttime and 24-h ABPM by presence or not of preeclampsia (PE) events
|
Variable |
PE (n=17) |
Non-PE (n=95) |
|
||
|
Median |
95% CI |
Median |
95% CI |
p |
|
|
Age |
30 |
25.01-36.98 |
31 |
29.00-33.00 |
0.928 |
|
Weeks
of pregnancy |
35 |
34.00-37.00 |
38 |
37.00-38.00 |
0.007 |
|
SBP
24-h ABPM |
120.50 |
113.58-131.10 |
118.50 |
116.00-119.45 |
0.329 |
|
DBP
24-h ABPM |
73.00 |
69.79-77.52 |
69.00 |
68.00-71.45 |
0.072 |
|
SBP
ABPM daytime |
124.00 |
116.00-131.62 |
122.00 |
120.00-125.00 |
0.363 |
|
DBP
ABPM daytime |
77.00 |
73.58-81.31 |
74.00 |
72.54-75.00 |
0.068 |
|
SBP
ABPM nighttime |
108.00 |
104.00-121.93 |
109.00 |
108.00-111.00 |
0.494 |
|
DBP
ABPM nighttime |
65.00 |
59.79-70.00 |
61.00 |
59.00-63.00 |
0.144 |
ABPM: ambulatory blood pressure monitoring; DBP: diastolic blood pressure; PE: preeclampsia SBP: systolic blood pressure.
Table 2. Differences between hemodynamic variables in patients with and without PE. Automatic learning variables are represented in bold characters
|
Variable |
PE (n=17) |
Non-PE (n=95) |
|
||||||
|
Median |
95% CI |
Median |
95% CI |
p |
|||||
|
TFC |
44.91 |
41.43-49.87 |
41.46 |
39.78-43.49 |
0.026 |
||||
|
ETR |
36.28 |
35.71-39.25 |
35.99 |
35.08-36.67 |
0.210 |
||||
|
STR |
32.15 |
26.07-36.03 |
32.54 |
31.25-33.71 |
0.748 |
||||
|
CAI |
291.37 |
196.82-382.56 |
314.48 |
279.68-351.57 |
0.529 |
||||
|
CI |
4.71 |
4.25-5.01 |
4.66 |
4.39-4.97 |
0.951 |
||||
|
ACI |
1.31 |
1.11-1.40 |
1.47 |
1.35-1.54 |
0.100 |
||||
|
SVI |
62.51 |
46.56-71.98 |
62.48 |
59.71-69.38 |
0.641 |
||||
|
CWI |
6.65 |
5.07-7.02 |
5.55 |
5.27-5.91 |
0.065 |
||||
|
VRI |
1684.63 |
1542.56-1912.82 |
1507.83 |
1412.24-1586.64 |
0.054 |
||||
|
Ea |
1.04 |
0.91-1.27 |
0.96 |
0.92-1.06 |
0.204 |
||||
|
Ees |
1.34 |
1.17-1.56 |
1.25 |
1.21-1.32 |
0.169 |
||||
|
VAC |
0.75 |
0.66-0.89 |
0.74 |
0.71-0.78 |
0.932 |
||||
|
|
Mean |
SD |
SE |
95% CI |
Mean |
SD |
SE |
95% CI |
p |
|
HI |
18.12 |
4.11 |
0.99 |
16.00-20.23 |
20.96 |
5.69 |
0.58 |
19.80-22.12 |
0.052 |
|
VI |
117.12 |
23.43 |
5.68 |
105.07-129.17 |
123.96 |
25.92 |
2.65 |
118.68-129.24 |
0.312 |
|
PWVcf |
6.42 |
1.12 |
0.28 |
5.82-7.02 |
6.09 |
1.03 |
0.10 |
5.87-6.31 |
0.250 |
ACI: Arterial compliance index; CAI: Cardiac acceleration index; CI: Cardiac index; Ea: Arterial elastance; Ees: Ventricular end-systolic elastance; ETR: Ejective time ratio; HI: Heather index; PE: preeclampsia; cfPWV: carotid-femoral pulse wave velocity; STR: systolic time ratio; SVI: Stroke volume index; SWI: Cardiac work index; TFC: Thoracic fluid content; VAC: Ventriculo-arterial coupling; VI: Velocity index; VRI: Vascular resistance index.
DISCUSIÓN
Non-invasive hemodynamic pregnancy evaluation has already been performed with reliable results. (20)
However, most published studies have been made in the second and third trimester of pregnancy. We observed that CI evidenced no differences, but VRI was higher between weeks 10 and 16 in patients who later developed PE (p=0.05). Different from previous works describing hyperdynamics in the first trimester as PE predictor, (21,22)
this behavior was not observed in the present work, though the inadequate hemodynamic trend in PE patients with 10 to 15- week gestational age should be pointed out. A similar interpretation was made regarding arterial stiffness, as cfPWV showed no difference because it increases in PE. (17-23)
Conversely, contractility variables (CWI, with p=0.06, and HI with p=0.05) showed a statistical trend to increase in patients who developed PE, and were coherent with their classification tree selection by the ML algorithm, evidencing that, different from the classically assessed CI and VRI, occult patterns could be determined with other variables, (13)
which were detected at an early gestational age.
AI is widely used in the medical field, and ML is increasingly employed in health care, prediction, and diagnosis as method to establish priorities. (24)
Previous reports have used AI with ML with non-invasive hemodynamics. (25)
The most frequently used algorithms were of Deep Learning with neuronal networks and classification trees for the detection of hidden patterns in great volume of data. In the present work, we chose a classification tree, as it is easier to reason, different from neuronal networks where synaptic weights shape a more complex not emulative interpretation for posterior database programming to be used in the hospital reality. The identification of patterns helps to predict and make decisions for diagnosis and treatment planning. (26)
Regarding limitations, as the referral of pregnant women is more frequent since week 20, the selected sample size was not enough to analyze the incidence of PE with a logistic regression model. However, the advantage of ML is shown, as it was able to detect a hemodynamic pattern associated with PE. This suggests that AI worked supplementing traditional statistics, having the potential of revolutionizing medical care during pregnancy, by providing more accurate diagnoses, achieving better therapeutic opportunities, reducing costs of hospital medical care and improving the interpretation between specialists. (27)
Another limitation is that an internal algorithm validation could not be made at the moment of the analysis. However, recruitment of pregnant women continues with the possibility of achieving this goal. In our study, AI could detect a pattern with five noninvasive hemodynamic variables that were not the classical ones (CI and VRI), and which were able to predict the development of PE in early pregnancy stages. It can be observed that the initiation node was ACI and the secondary nodes corresponded mostly to contractility (HI, ETR, and CWI). This suggests that the afterload versus contractility relationship is altered prematurely between weeks 10 and 16, and still does not impact in arterial and ventricular elastances, does not affect VAC and maintains an adequate cardiovascular performance (VAC=0.75 in PE patients, Table 2). (28-31)
The identification of pregnant women with hemodynamic behavior that characterizes PE between weeks 10 and 16, where the therapeutic opportunity is optimal for ASA indication, could derive more benefit from programming the hemodynamic pattern generated by ML in the databases or electronic clinical histories that automatically and timely alert on the indication of ASA with implications on maternal risk and her offspring in the future development.
Fig1 Automatic learning algorithm with non-invasive hemodynamics in high-risk pregnant women with 10 to 16-weeks of gestation. Information gain from hemodynamic variables with PE in red rectangle. Abbreviations as in Table 2.
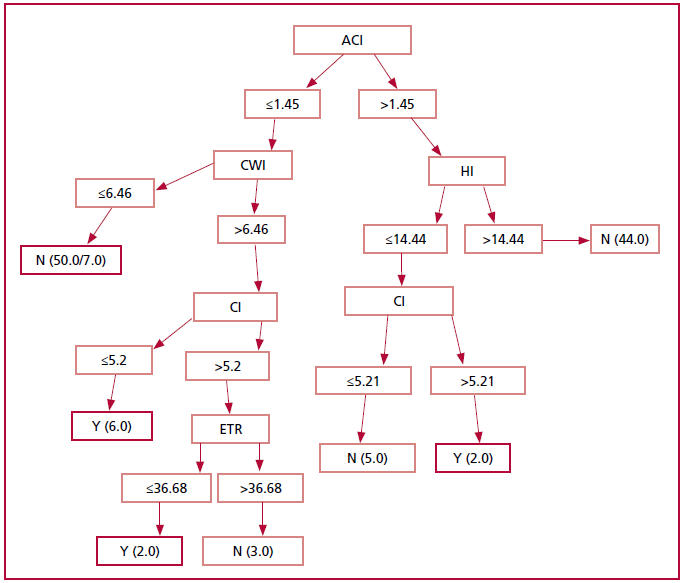
ACI: Arterial compliance index; CI: Cardiac index; CWI: Cardiac work index; ETR: Ejective time ratio; HI: Heather index; N: No; Y: Yes.
CONCLUSION
Using ML, ACI, CI, CWI., ETR and HI predicted in our sample an early pattern of risk for PE development between weeks 10 to 16 of pregnancy, with excellent discrimination and accuracy, non-invasively, safely and at low cost. The early non-invasive assessment of hemodynamic variables would be a useful tool to optimize the therapeutic opportunity of ASA indication in high-risk pregnancy.
https://creativecommons.org/licenses/by-nc-sa/4.0/

Conflicts of interest
None declared. (See authors' conflict of interests forms on the web/Additionalmaterial).
©Rev Argent Cardiol
REFERENCES
- Garovic VD, Dechend R, Easterling T, Karumanchi SA, McMurtry Baird S, Magee LA et al. American Heart Association Council on Hypertension; Council on the Kidney in Cardiovascular Disease, Kidney in Heart Disease Science Committee; Council on Arteriosclerosis, Thrombosis and Vascular Biology; Council on Lifestyle and Cardiometabolic Health; Council on Peripheral Vascular Disease; and Stroke Council. Hypertension in Pregnancy: Diagnosis, Blood Pressure Goals, and Pharmacotherapy: A Scientific Statement From the American Heart Association. Hypertension. 2022;79:e21-e41. https://doi.org/10.1161/HYP.0000000000000208
- Cottrell J, Cummings K, Jude D, Chaffin D. The effect of impedance cardiography directed antihypertensive therapy on fetal growth restriction rates and perinatal mortality in women with chronic hypertension. Pregnancy Hypertens. 2022;28:123-7. https://doi.org/10.1016/j.preghy.2022.03.006
- Chaiyasit N, Sahota DS, Ma R, Choolani M, Wataganara T, Sim WS, et al. Prospective Evaluation of International Prediction of Pregnancy Complications Collaborative Network Models for Prediction of Preeclampsia: Role of Serum sFlt-1 at 11-13 Weeks’ Gestation. Hypertension. 2022;79:314-22. https://doi.org/10.1161/HYPERTENSIONAHA.121.18021
- Hunter S, Robson SC. Adaptation of the maternal heart in pregnancy. Br Heart J. 1992;68:540-3. https://doi.org/10.1136/hrt.68.12.540
- Sanghavi M, Rutherford JD. Cardiovascular physiology of pregnancy. Circulation. 2014;130(12):1003-8. https://doi.org/10.1161/CIRCULATIONAHA.114.009029
- Mahajan S, Gu J, Lu Y, Khera R, Spatz ES, Zhang M, et al. Hemodynamic Phenotypes of Hypertension Based on Cardiac Output and Systemic Vascular Resistance. Am J Med. 2020;133:e127-39. https://doi.org/10.1016/j.amjmed.2019.08.042
- Meah VL, Cockcroft JR, Backx K, Shave R, Stöhr EJ. Cardiac output and related haemodynamics during pregnancy: a series of meta-analyses. Heart. 2016;102:518-26. https://doi.org/10.1136/heartjnl-2015-308476
- Buddeberg BS, Sharma R, O’Driscoll JM, Kaelin Agten A, Khalil A, Thilaganathan B. Cardiac maladaptation in term pregnancies with preeclampsia. Pregnancy Hypertens. 2018;13:198-203. https://doi.org/10.1016/j.preghy.2018.06.015
- Orabona R, Vizzardi E, Sciatti E, Bonadei I, Valcamonico A, Metra M, et al. Insights into cardiac alterations after pre-eclampsia: an echocardiographic study. Ultrasound Obstet Gynecol. 2017;49:124-33. https://doi.org/10.1002/uog.15983
- Staelens A, Tomsin K, Grieten L, Oben J, Mesens T, Spaanderman M, et al. Non-invasive assessment of gestational hemodynamics: benefits and limitations of impedance cardiography versus other techniques. Expert Rev Med Devices. 2013;10:765-79. https://doi.org/10.1586/17434440.2013.853466
- Sherwood A, Allen MT, Fahrenberg J, Kelsey RM, Lovallo WR, van Doornen LJ. Methodological guidelines for impedance cardiography. Psychophysiology. 1990;27:1-23. https://doi.org/10.1111/j.1469-8986.1990.tb02171.x
- Mansouri S, Alhadidi T, Chabchoub S, Salah RB. Impedance cardiography: recent applications and developments. Biomedical Research. 2018;29:3542-52. https://doi.org/10.4066/BIOMEDICALRESEARCH.29-17-3479
- Medina-Lezama J, Narvaez-Guerra O, Herrera-Enriquez K, Morey- Vargas OL, Bolaños-Salazar JF, Abugattas JP, et al. Hemodynamic Patterns Identified by Impedance Cardiography Predict Mortality in the General Population: The PREVENCION Study. J Am Heart Assoc. 2018;7:e009259. https://doi.org/10.1161/JAHA.118.009259
- Mennickent D, Rodríguez A, Opazo MC, Riedel CA, Castro E, Eriz-Salinas A, et al. Machine learning applied in maternal and fetal health: a narrative review focused on pregnancy diseases and complications. Front Endocrinol (Lausanne). 2023;14:1130139. https://doi.org/10.3389/fendo.2023.1130139
- Hedley PL, Hagen CM, Wilstrup C, Christiansen M. The use of artificial intelligence and machine learning methods in early pregnancy pre-eclampsia screening: A systematic review protocol. PLoS One. 2023;18:e0272465. https://doi.org/10.1371/journal.pone.0272465
- Bertini A, Salas R, Chabert S, Sobrevia L, Pardo F. Using Machine Learning to Predict Complications in Pregnancy: A Systematic Review. Front Bioeng Biotechnol. 2022;9:780389. https://doi.org/10.3389/fbioe.2021.780389
- Majul C, Paez O, De María M, Prieto N, Caballer C, Catallini A, y cols. Velocidad de onda de pulso y onda de reflexión precoz en el embarazo con riesgo de preeclampsia. Rev Arg de Cardiol. 2000;68:353-8.
- Phumsiripaiboon P, Suksai M, Suntharasaj T, Geater A. Screening for pre-eclampsia: Performance of National Institute for Health and Care Excellence guidelines versus American College of Obstetricians and Gynecologists recommendations. J Obstet Gynaecol Res. 2020;46:2323-31. https://doi.org/10.1111/jog.14425
- Gijón-Conde T, Gorostidi M, Banegas JR, de la Sierra A, Segura J, Vinyoles E, et al. Documento de la Sociedad Española de Hipertensión-Liga Española para la Lucha contra la Hipertensión Arterial (SEH-LELHA) sobre monitorización ambulatoria de la presión arterial (MAPA) 2019 [Position statement on ambulatory blood pressure monitoring (ABPM) by the Spanish Society of Hypertension (2019)]. Hipertens Riesgo Vasc. 2019;36:199-212. Spanish. https://doi.org/10.1016/j.hipert.2019.05.002
- Tomsin K, Mesens T, Molenberghs G, Gyselaers W. Impedance cardiography in uncomplicated pregnancy and pre-eclampsia: a reliability study. J Obstet Gynaecol. 2012;32:630-4. https://doi.org/10.3109/01443615.2012.673036
- Bujold E. Optimal Screening for Preeclampsia in the First Trimester of Pregnancy. Hypertension. 2022;79:323-24. https://doi.org/10.1161/HYPERTENSIONAHA.121.18421
- Perry H, Lehmann H, Mantovani E, Thilaganathan B, Khalil A. Correlation between central and uterine hemodynamics in hypertensive disorders of pregnancy. Ultrasound Obstet Gynecol. 2019;54:58- 63. https://doi.org/10.1002/uog.19197
- Hausvater A, Giannone T, Sandoval YH, Doonan RJ, Antonopoulos CN, Matsoukis IL, et al. The association between preeclampsia and arterial stiffness. J Hypertens. 2012;30:17-33. https://doi.org/10.1097/HJH.0b013e32834e4b0f
- Koshimizu H, Kojima R, Okuno Y. Future possibilities for artificial intelligence in the practical management of hypertension. Hypertens Res. 2020;43:1327-37. https://doi.org/10.1038/s41440-020-0498-x
- Naidu SMM, Pandey PC, Bagal UR, Hardas SP. Beat-to-beat estimation of stroke volume using impedance cardiography and artificial neural network. Med Biol Eng Comput. 2018;56:1077-89. https://doi.org/10.1007/s11517-017-1752-5.
- Sufriyana H, Husnayain A, Chen YL, Kuo CY, Singh O, Yeh TY, et al. Comparison of Multivariable Logistic Regression and Other Machine Learning Algorithms for Prognostic Prediction Studies in Pregnancy Care: Systematic Review and Meta-Analysis. JMIR Med Inform. 2020;8:e16503. https://doi.org/10.2196/16503
- Ramakrishnan R, Rao S, He JR. Perinatal health predictors using artificial intelligence: A review. Womens Health (Lond). 2021;17:17455065211046132. https://doi.org/10.1177/17455065211046132
- Ikonomidis I, Aboyans V, Blacher J, Brodmann M, Brutsaert DL, Chirinos JA, et al. The role of ventricular-arterial coupling in cardiac disease and heart failure: assessment, clinical implications and therapeutic interventions. A consensus document of the European Society of Cardiology Working Group on Aorta & Peripheral Vascular Diseases, European Association of Cardiovascular Imaging, and Heart Failure Association. Eur J Heart Fail. 2019;21:402-24. https://doi.org/10.1002/ejhf.1436
- Kass DA. Ventricular arterial stiffening: integrating the pathophysiology. Hypertension. 2005;46:185-93. https://doi.org/10.1161/01.HYP.0000168053.34306.d4
- Chirinos JA. Ventricular-arterial coupling: Invasive and noninvasive assessment. Artery Res. 2013;7:10.1016/j.artres.2012.12.002. https://doi.org/10.1016/j.artres.2012.12.002.
- Olano RD, Espeche WG, Salazar MR, Forcada P, Chirinos JA, de Iraola A, et al. Evaluation of ventricular-arterial coupling by impedance cardiography in healthy volunteers. Physiol Meas. 2019;40:115002. https://doi.org/10.1088/1361-6579/ab5172