Walter Masson1,2, MTSAC, Leandro Barbagelata2 MTSAC, Gerardo Masson 1,3 MTSAC, Santiago Lynch3 MTSAC, Melina Huerin1,4, Daniel Siniawski1,2 MTSAC
1Council on Epidemiology and Cardiovascular Prevention, Argentine Society of Cardiology, Buenos Aires, Argentina.
2Department of Cardiology, Hospital Italiano de Buenos Aires, Argentina.
3Department of Cardiology, Instituto Cardiovascular San Isidro, Sanatorio Las Lomas, San Isidro, Buenos Aires, Argentina.
4Department of Cardiology, Instituto Cardiovascular Lezica, San Isidro, Buenos Aires, Argentina.
Address for correspondence: Walter Masson. E-mail: walter.masson@hospitalitaliano.org.ar. Dirección postal: Perón 4190. Ciudad Autónoma de Buenos Aires. Argentina (Código postal: C1199ABB). E-mail: walter.masson@hospitalitaliano.org.ar.
Rev Argent Cardiol 2023;91:309-316 http://dx.doi.org/10.7775/rac.v91.i5.20666
SEE RELATED ARTICLE: Rev Argent Cardiol 2023;91:305-306. http://dx.doi.org/10.7775/rac.v91.i5.20663
ABSTRACT
Background: The SMART-REACH model predicts the risk or recurrent cardiovascular events.
Objectives: The objectives of this study were: a) to evaluate the residual cardiovascular risk in a secondary prevention population with LDL-C levels above the recommended goal, using a simulation model; and b) to determine the impact of optimizing lipid-lowering therapies in terms of residual cardiovascular risk reduction.
Methods: We conducted a cross-sectional, descriptive and multicenter study. Patient with a history of cardiovascular disease and a LDL-C ≥55 mg/dL were consecutively included. The 10-year and lifetime risk of recurrent events (myocardial infarction, stroke, or vascular death) were estimated using the SMART-REACH model. By means of a simulation, lipid-lowering LDL-C reduction, checking if lipid-lowering goal was achieved and calculating the reduction in cardiovascular risk and the corresponding number needed to treat (NNT).
Results: The cohort was made up of 187 patients; mean age was 67.9 ± 9.3 years and 72.7% were men. The calculated 10-year and lifetime residual risks were 37.1 ± 14.7% and 60.3 ± 10.7%, respectively. Overall, treatment was optimized with a single pharmacological strategy with statins, ezetimibe or PCSK9 inhibitor in 38.5%, 11.5% and 5.5% of the population, respectively. Optimization based on two treatments was performed in 27.5% (statins + ezetimibe), 7.7% (statins + PCSK9 inhibitor) and 1.1% (ezetimibe + PCSK9 inhibitor) of the cases. In 15 patients, treatment was optimized when the three drugs (statins + ezetimibe + PCSK9 inhibitor) were considered. Overall, 53.9% and 62.9% of the actions implemented to optimize treatment showed a 10-year or lifetime NNT < 30 to prevent an event, respectively.
Conclusion: In this study, patients with a history of cardiovascular disease who do not reach LDL-C goal showed significant residual cardiovascular risk. The simulation model showed a significant margin for optimizing treatment, with a marked reduction in residual cardiovascular risk.
Key words: Residual Cardiovascular Risk - Cholesterol LDL - Hydroxymethylglutaryl-CoA Reductase Inhibitors Ezetimibe - PCSK9 Inhibitors
RESUMEN
Introducción: el modelo SMART-REACH predice el riesgo de eventos cardiovasculares recurrentes.
Objetivos: los objetivos de este estudio fueron: a) evaluar el riesgo residual en una población en prevención secundaria y niveles de colesterol asociado a lipoproteínas de baja densidad (C-LDL) fuera de meta; b) mediante un modelo de simulación, determinar el impacto de optimizar las terapias hipolipemiantes en términos de reducción del riesgo residual.
Material y métodos: estudio transversal, descriptivo y multicéntrico. Se incluyeron consecutivamente pacientes con antecedentes cardiovasculares y un C-LDL mayor o igual que 55 mg/dL. El riesgo de eventos recurrentes (infarto agudo de miocardio, accidente cerebrovascular o muerte vascular) a 10 años y a lo largo de la vida se estimó utilizando el modelo SMART-REACH. Mediante una simulación, se optimizó el tratamiento hipolipemiante de cada paciente (utilizando estatinas, ezetimibe o inhibidores de proproteína convertasa subtilisina kexina tipo 9 [iPCSK9]), se estimó el descenso del C-LDL, se verificó el alcance del objetivo lipídico y se calculó la reducción del riesgo cardiovascular y el número necesario a tratar (NNT) correspondiente.
Resultados: se incluyeron 187 pacientes (edad media 67,9 ± 9,3 años, 72,7% hombres). Los riesgos residuales calculados a 10 años y a lo largo de la vida fueron 37,1 ± 14,7% y 60,3 ± 10,7%, respectivamente. Globalmente, se pudo optimizar una sola estrategia farmacológica con estatinas, ezetimibe o un iPCSK9 en el 38,5%, el 11,5% y el 5,5% de la población, respectivamente. La optimización basada en dos tratamientos se realizó en el 27,5% (estatinas + ezetimibe), el 7,7% (estatinas + iPCSK9) y el 1,1% (ezetimibe + iPCSK9) de los casos. En 15 pacientes se optimizó el tratamiento considerando los tres fármacos. El 53,9% y el 62,9% de las acciones para optimizar el tratamiento mostraron un NNT menor que 30 para evitar un evento a 10 años o a lo largo de la vida, respectivamente.
Conclusión: en este estudio, los pacientes con antecedentes cardiovasculares que no alcanzan la meta de C-LDL mostraron un riesgo residual considerable. La simulación mostró un importante margen para optimizar el tratamiento, con un impacto notable en el riesgo residual.
Palabras clave: Riesgo cardiovascular residual - Colesterol LDL - Inhibidores de la hidroximetilglutaril-CoA reductasa - Ezetimibe - Inhibidores de la PCSK9
Received: 28/08/2023
Accepted: 03/09/2023
INTRODUCCIÓN
Cardiovascular disease is the leading cause of death worldwide. (1)
In the field of secondary cardiovascular prevention, current therapies have demonstrated a remarkable ability to reduce cardiovascular risk in our patients. However, despite these measures, cardiovascular events still occur, even among patients receiving optimal pharmacological treatment. As a result, the concept of "residual cardiovascular risk" is commonly used to describe the persistent risk of recurrent events despite adequate management of traditional cardiovascular risk factors, as low-density lipoprotein cholesterol (LDL-C), blood pressure, or blood glucose levels. (2)
The prognosis of patients with a history of cardiovascular disease is highly variable. Traditional risk factors are independent predictors of recurrent events and mortality.(3)
Furthermore, the possibility of estimating residual cardiovascular risk using scores has been explored several years ago. (4,5)
However, their utility is limited in contemporary populations who are on statins and other cardioprotective therapies as the standard of care. (6)
Recently, a model has been developed for the prediction of recurrent cardiovascular events based on the Reduction of Atherothrombosis for Continued Health (REACH) cohort study. (7)
This model, called SMARTREACH, can estimate the 10-year and lifetime risk of recurrent cardiovascular events [acute myocardial infarction (AMI), stroke, or vascular death]. These estimations are based on clinical characteristics of patients that are readily available in a usual clinical setting. In addition, this model allows quantifying the impact of different pharmacological treatments on residual cardiovascular risk, estimating risk reduction, and calculating the number needed to treat (NNT) to avoid an event.
The objectives of this study were: a) to evaluate the residual cardiovascular risk in a population with a history of cardiovascular disease and LDL-C levels above the recommended goals, using the SMART-REACH model; and b) to determine the potential impact of optimizing lipid-lowering therapies in terms of residual cardiovascular risk reduction using a simulation model.
MATERIAL Y MÉTODOS
We conducted a cross-sectional, descriptive, and multicenter study in five outpatient cardiology centers in the Autonomous City of Buenos Aires and Great Buenos Aires, between July and November 2022. The population was made up of consecutive patients between 45 and 80 years (age range that allows calculation of residual risk with the SMART-REACH model) with a history of cardiovascular disease (coronary artery disease, peripheral artery disease or cerebrovascular disease). For the present study, patients who did not meet the LDL-C goal recommended by current guidelines ( < 55 mg/dL) were selected. (8) The medical records of the patients included were reviewed, obtaining information on their history, cardiovascular risk factors, and medication received.
Serum levels of glucose, total cholesterol, high-density lipoprotein cholesterol (HDL-C), triglycerides, C-reactive protein, lipoprotein(a) [Lp(a)] and creatinine were measured according to standard methods. LDL-C was calculated using the Friedewald formula. (9) Glomerular filtration rate was estimated according to the CKD-EPI (chronic kidney disease-epidemiology collaboration) equation. (10)
The 10-year and lifetime risks of recurrent events (MI, stroke, or vascular death) were estimated using the SMARTREACH model. (7)
Subsequently, a simulation model was used to further optimize the lipid-lowering treatment in each patient, estimating the decrease in LDL-C and verifying if the recommended lipid goal was reached or not. Based on the recommendations of the latest Consensus on Cardiovascular Prevention of the Argentine Society of Cardiology for secondary prevention of very high-risk patients who do not reach LDL-C goal, (11) we considered the following order for optimizing lipid-lowering treatment: 1) raise statins to maximum dose, using high-intensity statins (atorvastatin 80 mg/day or rosuvastatin 40 mg/day). The choice between the two regimens was based on previous treatment (raising the dose of the statin the patient was already receiving). If, on the other hand, the patient was receiving another statin, we arbitrarily chose to administer atorvastatin 80 mg/day; 2) add ezetimibe if LDL-C goal was not met despite treatment with statins; 3) add a proprotein convertase subtilisin/kexin type 9 inhibitor (PCSK9i) when despite treatment with statins and ezetimibe, LDL-C threshold was ⩾ 70 mg/dL. The simulation model considered no more than three steps in the process for optimizing lipid-lowering therapy (one strategy at a time). Some patients required a single step to optimize treatment (following the order previously mentioned), while others required two or three steps.
Finally, the 10-year and lifetime residual cardiovascular risk reduction with the corresponding NNT were calculated for each necessary optimizing step to achieve LDL-C goal. These results were automatically calculated by the SMARTREACH model with each treatment change for each patient.
Statistical analysis
Continuous variables were expressed as mean ± standard deviation (SD), or median and interquartile range (IQR) 25- 75, as applicable, and categorical variables as frequencies and percentages. Continuous data with normal distribution were compared between two groups using the Student's t test; the Wilcoxon-Mann-Whitney was used to compare data with non-normal distribution. A p value < 0.05 was considered statistically significant. All the statistical calculations were performed using STATA 13.1 software package.
Ethical considerations
The study was conducted following the recommendations regarding medical research of the Declaration of Helsinki, the Guidelines for Good Clinical Practice and the rules of the local Committee on Ethics. The study protocol was approved by the institutional review board.
RESULTS
A total of 187 patients were included; median age was 67.9 ± 9.3 years and 72.7% were men. The prevalence of type 2 diabetes in the population was 27.3% and 72.2% had hypertension; blood pressure was not well controlled in 17.1% and 10.7% were current smokers. Mean LDL-C level was 81.4 ± 23.8 mg/dL and median triglyceride level was 115 mg/dL (IQR 88-153). The baseline characteristics of the population are described in Table 1.
In total, 95.2% of the population was taking statins (high-intensity statins: 58.9%; moderate/low intensity- statins: 41.1%). The type and dose of statins used are shown in Table 2.
Regarding non-statin therapy, 40.6% of the subjects received ezetimibe (as monotherapy in only one patient, the rest received combination therapy with ezetimibe and statins), while two patients were treated with a PCSK9i.
On average, the calculated 10-year and lifetime residual risks were 37.1 ± 14.7% and 60.3 ± 10.7%, respectively.
The simulation showed that only 5 patients (2.7%) did not meet criteria to consider optimizing treatment. In all the cases, these were patients who had already been receiving high-intensity statins on maximum doses in combination with ezetimibe and had LDL-C value below the threshold for indicating a PCSK9i ( < 70 mg/dL). The remaining 97.3% of patients met the criteria for the first step for optimizing lipid-lowering treatment. In 81.9%, 12.6% and 5.5% of these cases it was possible to optimize statin treatment (switching to higher-intensity statin or raising to the maximum dose), adding ezetimibe, or administering an PCSK9i, respectively. However, only 37.9% of patients could achieve the LDL-C goal. A second step for optimizing treatment could be done in 81 patients. In this opportunity, ezetimibe or a PCSK9i was added in 80.2% and 19.8% of cases, respectively. Nevertheless, 35.8% of patients would not reach the LDL-C goal. Finally, a third step for optimizing treatment was achieved in 15 patients (all of them received a PCSK9i). In this occasion, LDL-C goal would be achieved in all the cases.
Considering all the steps for optimizing treatment altogether, monotherapy with statins, ezetimibe or PCSK9i could optimize treatment in 38.5% (n = 70), 11.5% (n = 21) and 5.5% (n = 10) of the population, respectively. The use of two drugs optimized treatment in 27.5% (statins + ezetimibe, 50 patients), 7.7% (statins + PCSK9i, 14 patients) and 1.1% (ezetimibe + PCSK9i, 2 patients) of the cases. In 15 patients (8.2%) treatment was optimized by considering three drugs (statins + ezetimibe + PCSK9i).
The simulation model showed that, after applying all possible strategies to optimize treatment, 72.7% (136 patients) would achieve the lipid-lowering goal. The remaining 27.3% consisted of 5 patients with no possibility of optimizing treatment and 46 patients with LDL-C value between the goal ( < 55 mg/dL) and the threshold value (70 mg/dL) eligible to indicate a PCSK9i. The stepwise sequence for optimizing treatment and the proportion of patients who achieved LDL-C goals can be seen in Figure 1.
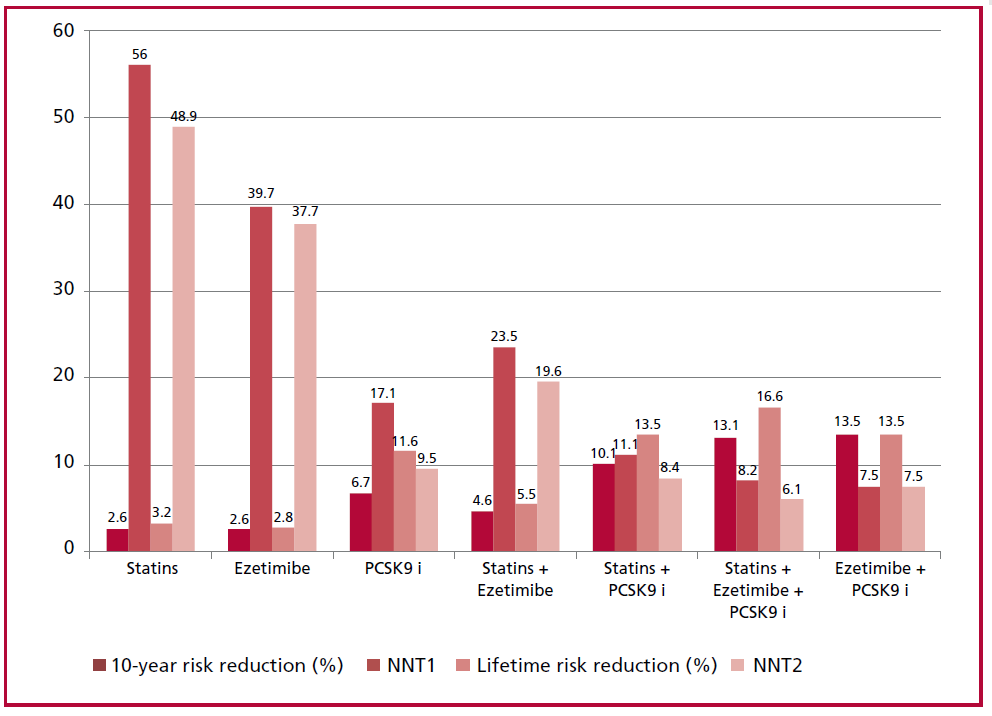
The 10-year and lifetime residual risks decreased on average by 2.8 ± 1.9% and 3.5 ± 2.9% after the first step in treatment optimization, respectively. The calculated NNT was 50.4 ± 28.4 in the first case and 42.5 ± 24.1 in the second case. After performing a second step for optimizing treatment, the 10-year and lifetime residual risks decreased on average by 6.1 ± 3% and 7.5 ± 3.6%, respectively. The calculated NNT was 19.7 ± 8.5 in the first case and 16.2 ± 6.5 in the second case. Finally, in the subgroup of patients in whom a third step for optimizing treatment could be performed, the 10-year and lifetime residual risks decreased on average by 12.8 ± 4.3% and 16.6 ± 2.5%, respectively. In this case, the calculated NNT were very low (8.2 ± 2.9 and 6.1 ± 1 for 10-year and lifetime risks, respectively). The impact of the different steps for optimizing lipid-lowering therapy on 10-year and lifetime residual risks are shown in Figure 2. The effect of the therapeutic regimens used on residual risk is shown in Figure 3.
Overall, 53.9% and 62.9% of the actions implemented to optimize treatment showed a 10-year or lifetime NNT < 30 to prevent an event, respectively. As expected, the lowest NNT values (greater impact on residual risk) were observed in patients with a higher baseline atherogenic lipid profile (Table 3).
Table 1. Baseline characteristics of the population
|
Continuous variables* |
Total population (n = 187) |
|
Age, years |
67.9 (9.3) |
|
Time from index event, years |
6 (3-11) |
|
Systolic blood pressure, mm Hg |
126.3 (14.1) |
|
Total
cholesterol, mg/dL |
151.4 (30.7) |
|
LDL-C, mg/dL |
81.4 (23.8) |
|
HDL-C, mg/dL |
44.2 (10.4) |
|
Triglycerides, mg/dL |
115 (88-153) |
|
non-HDL-cholesterol, mg/dL |
107.2 (29.4) |
|
Creatinine level, mg/dL |
1.1 (0.4) |
|
Glomerular filtration rate (mL/min) |
75.6 (20.2) |
|
Glycated hemoglobin (HbA1c), %** |
6.8 (1.2) |
|
C-reactive protein, mg/dL (n=57) |
1.5 (1.4) |
|
Lipoprotein(a), mg/dL (n=57) |
25 (14-63) |
|
Apolipoprotein B, mg/dL (n=51) |
67.2 (30.4) |
|
Categorical variables, % |
|
|
Men |
72.7 |
|
Type 2 diabetes |
27.3 |
|
Hypertension |
72.2 |
|
Active smoker |
10.7 |
|
Coronary artery disease |
83.1 |
|
Acute coronary syndrome |
55.1 |
|
Myocardial infarction |
36.4 |
|
Coronary revascularization |
75.4 |
|
Peripheral vascular disease |
13.9 |
|
Cerebrovascular disease |
15.5 |
|
Heart failure |
12.8 |
|
Atrial fibrillation |
12.3 |
|
Familial hypercholesterolemia |
8.6 |
*Mean or median (standard deviation or interquartile range)
**Patients with type 2 diabetes
Fig. 1 Stepwise sequence for optimizing treatment and proportion of patients who achieved LDL-C goal. PCSK9i: proprotein convertase subtilisin/ kexin type 9 inhibitors.

Fig. 2 The impact of the different steps for optimizing lipid-lowering therapy on 10-year and lifetime residual risks. NNT1: number needed to treat to reduce a recurrent event at 10 years. NNT2: number needed to treat to reduce a lifetime recurrent event.

Fig. 3 Impact of the different therapeutic schemes used to optimize treatment on residual risk. NNT1: number needed to treat to reduce a recurrent event at 10 years. NNT2: number needed to treat to reduce a lifetime recurrent event. PCSK9: proprotein convertase subtilisin/ kexin type 9.
DISCUSIÓN
In primary prevention, the current guidelines recommend the use of risk scores to stratify cardiovascular risk, since patients at higher cardiovascular risk are more likely to benefit from certain pharmacological treatments. (8,11,18,19) On the other hand, the traditional approach suggests that all patients with established vascular disease should be classified as "very high risk". However, this universal and simplified approach ignores the fact that the individual level of cardiovascular risk may vary in these patients and limits the possibility of a more personalized management in secondary prevention. (20)
In this context, predictive tools as the SMARTREACH model used in our study have emerged to estimate residual cardiovascular risk. The use of these tools could be very useful, since recurrent events are very common in the population with a history of cardiovascular disease. (21,22) Moreover, the clinical applicability of the risk scores developed to estimate residual cardiovascular risk has already been evaluated in our country. (23) Specifically, the present study evaluated a subpopulation in secondary prevention that did not reach the LDL-C goal ( < 55 mg/dL). As it would be expected in a subgroup with higher lipid values, our findings showed a significant estimated residual risk, not only for developing an event in a relatively short time interval (10 years), but also for experiencing recurrent events over a lifetime. This long-term time perspective could be especially useful in younger patients.
The use of high-intensity statins in patients with a history of cardiovascular disease is specifically recommended in the current guidelines. ((8),(11),(17),(18)) However, high-intensity statins are underutilized in routine practice. (24) These clinically relevant problems in the management of lipid-lowering medications have also been reported in our country. (25,26) In our study, approximately 4 out of 10 patients were receiving inadequate dose of statins. Consequently, the margin for improving statin therapy was relevant.
When the lipid-lowering goal is not achieved, addition of other non-statin drugs is also strongly recommended. (27) Combination therapy with statins and ezetimibe was associated with greater cardiovascular benefit compared to statin monotherapy in the IMPROVE-IT study. (28) Similarly, in patients with a history of cardiovascular disease, combination therapy with moderate-intensity statins and ezetimibe was not inferior to monotherapy with high-intensity statins in the RACING study. (29) Likewise, the addition of a PCSK9 inhibitor to secondary prevention patients treated with statins has demonstrated a reduction in residual cardiovascular risk in the FOURIER and ODYSSEY OUTCOMES trials. (3031) Nevertheless, the use of combination therapies is uncommon in daily practice. (24,32) In this regard, about 40% of the patients in our study were receiving ezetimibe and only two patients were receiving a PCSK9i. Again, after the simulation, treatment was optimized in 88 and 41 patients with the use of ezetimibe or PCSK9i, alone or in combination, respectively. The use of PCSK9i might even have been higher if we had not considered a threshold value of 70 mg/dL as an indication. Our findings are similar to those reported by Cannon et al. in a simulation model performed with patients in the United States. (13) In this case, 20%, 24%, and 26% of the population evaluated required optimizing therapy with statins alone, with combination therapy of statins and ezetimibe, or with a triple scheme (statins, ezetimibe, and PCSK9i) to achieve the LDL-C goal < 55 mg/dL, respectively. (33)
The NNT was introduced in the setting of clinical trials to assess the impact of a given treatment, (34) and is defined as the number of individuals needed to be treated with the experimental therapy to produce or prevent one additional event compared with those that would occur with the control treatment. The lower the NNT, the more effective the treatment. Although there is no cut-off value, there is some consensus among physicians and patients that a value below 30 is more than acceptable in the context of cholesterol- lowering treatments. (35) In our study, more than half of the individual interventions performed to optimize lipid-lowering treatment showed an NNT value < 30. The relevance of this finding is that optimizing lipid-lowering therapy produces a considerable reduction in the residual cardiovascular risk in many patients. Our findings are consistent with a simulation study based on the recently published DA VINCI study, (36) where the absolute and relative risk reduction with maximum possible treatment optimization in patients with a history of cardiovascular disease was 8.1% and 24%, respectively.
Finally, it is important to emphasize that the greatest impact in terms of residual risk reduction was observed in patients with a higher baseline atherogenic lipid profile and with very-high intensity LDL-C-lowering interventions. Undoubtedly, once again our findings demonstrate the direct association between the quantity and quality of lipid profile and the clinical complications of atherosclerosis. (37,38)
This study has some limitations. The possibility of bias cannot be ruled out due to the study design. Moreover, the score used in this study has not been validated in Argentina. Nevertheless, external validation of SMART-REACH model was performed in North America. (39) In addition, we did not consider the presence of statin intolerance in the simulation, a situation widely reported in the literature. (40) In other words, the simulation evaluated the optimization of treatment in an ideal scenario without adverse drug effects that could limit its use. Another remarkable aspect is that the incorporation of other lipid-lowering drugs as bempedoic acid or inclisiran were not considered by the simulation model because they were not available in our country at the time the study was conducted. On the other hand, our study only focused on optimizing lipid-lowering treatment. The model allows adjustment for other risk factors and incorporation of other drugs as antiplatelet agents or diabetes medications. Finally, the aim of the present study was to estimate residual cardiovascular risk using predictive tools. Further prospective research should be conducted to determine whether these scores calibrate and discriminate correctly in our population.
Table 2. Type and dose of statins received by the population
|
Statin |
n |
Total, n (%) |
|||||
|
|
2 mg |
5 mg |
10 mg |
20 mg |
40 mg |
80 mg |
|
|
Simvastatin |
|
1 |
4 |
|
|
5 (2.8) |
|
|
Atorvastatin |
|
15 |
26 |
31 |
12 |
84 (47.2) |
|
|
Rosuvastatin |
|
2 |
19 |
37 |
26 |
|
84 (47,2) |
|
Fluvastatin |
|
3 |
3 (1.7) |
||||
|
Pitavastatin |
2 |
|
2 (1.1) |
||||
Table 3. Baseline atherogenic particles and effect of treatment optimization on residual risk
|
Variable |
NNT < 30 (10-year risk) |
NNT ≥ 30 (10-year risk) |
p |
NNT < 30 (lifetime risk) |
NNT ≥ 30 (lifetime risk) |
p |
|
LDL-C, mg/dL |
94.4 (23.8) |
66.8 (10.1) |
<0.001 |
91.6 (23.8) |
64.2 (7.1) |
<0.001 |
|
non-HDL-C, mg/dL |
122.2 (30.7) |
91.6 (17.5) |
<0.001 |
119.3 (30) |
88.6 (17.2) |
<0.001 |
|
ApoB, mg/dL (n=51) |
77.1 (31.5) |
57.7 (28.1) |
0.027 |
74.2 (31.8) |
56.2 (27.2) |
0.047 |
ApoB: apolipoprotein B. NNT: number necessary to treat
CONCLUSION
Our study demonstrated that patients with a history of cardiovascular disease who do not reach LDL-C goal have a significant 10-year and lifetime residual cardiovascular risk. However, the simulation model showed a significant margin for optimizing treatment, which was also associated with a marked reduction in residual cardiovascular risk.
Conflicts of interest
None declared. (See authors' conflict of interests forms on the web/Additionalmaterial).
https://creativecommons.org/licenses/by-nc-sa/4.0/

©Revista Argentina de Cardiología
REFERENCES
- Roth GA, Abate D, Abate KH, Abay SM, Abbafati C, Abbasi N, et al. Global, regional, and national age-sex-specific mortality for 282 causes of death in 195 countries and territories, 1980-2017: a systematic analysis for the Global Burden of Disease Study 2017. Lancet. 2018;392:1736-1788. https://doi.org/10.1016/S0140-6736(18)32203-7.
- Dhindsa DS, Sandesara PB, Shapiro MD, Wong ND. The Evolving Understanding and Approach to Residual Cardiovascular Risk Management. Front Cardiovasc Med. 2020;7:88. https://doi.org/10.3389/fcvm.2020.00088.
- Chen D-Y, Li C-Y, Hsieh M-J, Chen C-C, Hsieh I-C, Chen T-H, et al. Predictors of subsequent myocardial infarction, stroke, and death in stable post-myocardial infarction patients: A nationwide cohort study. Eur Heart J Acute Cardiovasc Care. 2019;8:634-42. https://doi.org/10.1177/2048872617730037.
- Wong ND, Cupples LA, Ostfeld AM, Levy D, Kannel WB. Risk factors for long-term coronary prognosis after initial myocardial infarction: the Framingham Study. Am J Epidemiol. 1989;130:469-80. https://doi.org/10.1093/oxfordjournals.aje.a115360.
- D’Agostino RB, Russell MW, Huse DM, Ellison RC, Silbershatz H, Wilson PW, et al. Primary and subsequent coronary risk appraisal: new results from the Framingham Heart Study. Am Heart J 2000; 139: 272–81. https://doi.org/10.1067/mhj.2000.96469.
- Wong ND, Zhao Y, Xiang P, Coll B, López JAG. Five-Year Residual Atherosclerotic Cardiovascular Disease Risk Prediction Model for Statin Treated Patients With Known Cardiovascular Disease. Am J Cardiol. 2020;137:7-11. https://doi.org/10.1016/j.amjcard.2020.09.043.
- Wilson PWF, D’Agostino R, Bhatt DL, Eagle K, Pencina MJ, Smith SC, et al. Reach registry. An international model to predict recurrent cardiovascular disease. Am J Med. 2012;125:695-703. https://doi.org/10.1016/j.amjmed.2012.01.014.
- Visseren FLJ, Mach F, Smulders YM, Carballo D, Koskinas KC, Bäck M, et al. 2021 ESC Guidelines on cardiovascular disease prevention in clinical practice. Eur J Prev Cardiol. 2022;29:5-115. https://doi.org/10.1093/eurjpc/zwab154.
- Friedewald WT, Levy RI, Fredrickson DS. Estimation of the concentration of low-density lipoprotein cholesterol in plasma, without use of the preparative ultracentrifuge. Clin Chem 1972;18:499-502.
- Levey AS, Stevens LA. Estimating GFR using the CKD Epidemiology Collaboration (CKD-EPI) creatinine equation: more accurate GFR estimates, lower CKD prevalence estimates, and better risk predictions. Am J Kidney Dis 2010;55:622-27. https://doi.org/10.1053/j.ajkd.2010.02.337.
- Giunta G, Lavalle Cobo A, Brandani L, Lobo M, Forte E, Masson G, et al. Consenso de Prevención Cardiovascular. Rev Argent Cardiol 2023;91 (Suplemento 4): 1-190. http://dx.doi.org/10.7775/rac.es.v91.s4.
- Leitersdorf E. Cholesterol absorption inhibition: filling an unmet need in lipid-lowering management. Eur Heart J Suppl, 2001;3(Suppl E):E17-E23. https://doi.org/10.1016/S1520-765X(01)90108-7.
- Cannon CP, Khan I, Klimchak AC, Sánchez RJ, Sasiela WJ, Massaro JM, et al. Simulation of lipid-lowering therapy intensification in a population with atherosclerotic cardiovascular disease. Am Heart J. 2019;216:30-41. https://doi.org/10.1016/j.ahj.2019.06.005.
- Mikhailidis DP, Sibbring GC, Ballantyne CM, Davies GM, Catapano AL. Meta-analysis of the cholesterol-lowering effect of ezetimibe added to ongoing statin therapy. Curr Med Res Opin. 2007;23:2009-26. https://doi.org/10.1185/030079907x210507.
- Schmidt AF, Pearce LS, Wilkins JT, Overington JP, Hingorani AD, Casas JP. PCSK9 monoclonal antibodies for the primary and secondary prevention of cardiovascular disease. Cochrane Database Syst Rev. 2017;4:CD011748. https://doi.org/10.1002/14651858.CD011748.pub2.
- LaRosa JC, Grundy SM, Waters DD, Shear C, Barter P, Fruchart JC, et al. Intensive lipid lowering with atorvastatin in patients with stable coronary disease. N Engl J Med. 2005;352:1425–35. https://doi.org/10.1056/NEJMoa050461.
- Sadeq A, Elnour AA, Farah FH, Ramadan A, Baraka MA, Don J, et al. A Systematic Review of Randomized Clinical Trials on the Efficacy and Safety of Pitavastatin. Curr Rev Clin Exp Pharmacol. 2023;18:120-47. https://doi.org/10.2174/2772432817666220531115314.
- Mach F, Baigent C, Catapano AL, Koskinas KC, et al. ESC Scientific Document Group. 2019 ESC/EAS guidelines for the management of dyslipidaemias: lipid modification to reduce cardiovascular risk. Eur Heart J. 2020;41:111-88. https://doi.org/10.1093/eurheartj/ehz455.
- Grundy SM, Stone NJ, Bailey AL, et al. 2018 AHA/ACC/AACVPR/ AAPA/ABC/ACPM/ADA/AGS/APhA/ASPC/NLA/PCNA guideline on the management of blood cholesterol: a report of the American College of Cardiology/American Heart Association Task Force on Clinical Practice Guidelines. J Am Coll Cardiol. 2019;73:e285–e350. https://doi.org/10.1016/j.jacc.2018.11.003.
- Kaasenbrood L, Boekholdt SM, van der Graaf Y, et al. Distribution of estimated 10-year risk of recurrent vascular events and residual risk in a secondary prevention population. Circulation 2016;134:1419- 29. https://doi.org/10.1161/CIRCULATIONAHA.116.021314.
- Colantonio LD, Shannon ED, Orroth KK, Zaha R, Jackson EA, Rosenson RS, et al. Ischemic Event Rates in Very-High-Risk Adults. J Am Coll Cardiol. 2019;74:2496-507. https://doi.org/10.1016/j.jacc.2019.09.025.
- Steen DL, Khan I, Andrade K, Koumas A, Giugliano RP. Event Rates and Risk Factors for Recurrent Cardiovascular Events and Mortality in a Contemporary Post Acute Coronary Syndrome Population Representing 239 234 Patients During 2005 to 2018 in the United States. J Am Heart Assoc. 2022;11:e022198. https://doi.org/10.1161/JAHA.121.022198.
- Siniawski D, Masson G, Masson W, Barbagelata L, Destaville J, Lynch S, et al. Int J Cardiol Cardiovasc Risk Prev. 2023;18:200198. https://doi.org/10.1016/j.ijcrp.2023.200198.
- Navar AM, Kolkailah AA, Gupta A, Gillard KK, Israel MK, Wang Y, et al. Gaps in Guideline-Based Lipid-Lowering Therapy for Secondary Prevention in the United States: A Retrospective Cohort Study of 322 153 Patients. Circ Cardiovasc Qual Outcomes. 2023;16:533-43. https://doi.org/10.1161/CIRCOUTCOMES.122.009787.
- Rosende A, Caniggia C, Casuscelli J, Mirada M, Comtesse D, Charask A y cols. Conductas frente a la utilización de estatinas en una muestra de médicos de la Argentina. Estudio COFEMA. Rev Argent Cardiol 2016;84:558-64.
- Castillo Costa YB, Mauro V, Charask AA, Fairman E, Buhezo H, Barrero C. Uso de la estrategia de alta intensidad con estatinas ¿Se cumplen las pautas establecidas? Rev Argent Cardiol 2018;86:46-8. http://dx.doi.org/10.7775/rac.es.v86.i1.11430.
- Raschi E, Casula M, Cicero AFG, Corsini A, Borghi C, Catapano A. Beyond statins: New pharmacological targets to decrease LDLcholesterol and cardiovascular events. Pharmacol Ther. 2023:108507. https://doi.org/10.1016/j.pharmthera.2023.108507.
- Cannon CP, Blazing MA, Giugliano RP, McCagg A, White JA, Theroux P, et al. Ezetimibe added to statin therapy after acute coronary syndromes. N Engl J Med 2015;372: 2387-97. https://doi.org/10.1056/NEJMoa1410489.
- Kim BK, Hong SJ, Lee YJ, Hong SJ, Yun KH, Hong BK, et al. Long-term efficacy and safety of moderate-intensity statin with ezetimibe combination therapy versus high-intensity statin monotherapy in patients with atherosclerotic cardiovascular disease (RACING): a randomised, open-label, non-inferiority trial. Lancet. 2022;400:380- 90. https://doi.org/10.1016/S0140-6736(22)00916-3.
- Sabatine MS, Giugliano RP, Keech AC, Honarpour N, Wiviott SD, Murphy SA, et al. Evolocumab and clinical outcomes in patients with cardiovascular disease. N Engl J Med. 2017;376:1713-22. https://doi.org/10.1056/NEJMoa1615664.
- Schwartz GG, Steg PG, Szarek M, Bhatt DL, Bittner VA, Diaz R, et al. Alirocumab and cardiovascular outcomes after acute coronary syndrome. N Engl J Med. 2018;379:2097-107. https://doi.org10.1056/NEJMoa1801174.
- Bradley CK, Kolkailah AA, Shah NP, Page CB, Peterson ED, Navar AM. Uptake of non-statin lipid-lowering therapies for secondary prevention in community practice. J Clin Lipidol. 2023;17:412-4. https://doi.org/10.1016/j.jacl.2023.03.006.
- Cannon CP, Khan I, Klimchak AC, Reynolds MR, Sanchez RJ, Sasiela WJ. Simulation of Lipid-Lowering Therapy Intensification in a Population With Atherosclerotic Cardiovascular Disease. JAMA Cardiol. 2017;2:959-66. https://doi.org/10.1001/jamacardio.2017.2289.
- Mendes D, Alves C, Batel-Marques F. Number needed to treat (NNT) in clinical literature: an appraisal. BMC Med. 2017;15:112. https://doi.org/10.1186/s12916-017-0875-8.
- Robinson JG, Huijgen R, Ray K, Persons J, Kastelein JJP, Pencina MJ. Determining When to Add Nonstatin Therapy: A Quantitative Approach. J Am Coll Cardiol. 2016;68:2412-21. https://doi.org/10.1016/j.jacc.2016.09.928.
- Brandts J, Bray S, Villa G, Catapano AL, Poulter NR, Vallejo-Vaz AJ, et al. Optimal implementation of the 2019 ESC/EAS dyslipidaemia guidelines in patients with and without atherosclerotic cardiovascular disease across Europe: a simulation based on the DA VINCI study. Lancet Reg Health Eur. 2023;31:100665. https://doi.org/10.1016/j.lanepe.2023.100665.
- Qiao YN, Zou YL, Guo SD. Low-density lipoprotein particles in atherosclerosis. Front Physiol. 2022;13:931931. https://doi.org/10.3389/fphys.2022.931931.
- Lu Y, Cui X, Zhang L, Wang X, Xu Y, Qin Z, et al. The Functional Role of Lipoproteins in Atherosclerosis: Novel Directions for Diagnosis and Targeting Therapy. Aging Dis. 2022;13:491-520. https://doi.org/10.14336/AD.2021.0929.
- Kaasenbrood L, Bhatt DL, Dorresteijn JAN, Wilson PWF, D’Agostino Sr RB, Massaro JM. et al. Estimated Life Expectancy Without Recurrent Cardiovascular Events in Patients With Vascular Disease: The SMART-REACH Model. J Am Heart Assoc. 2018;7:e009217. https://doi.org/10.1161/JAHA.118.009217.
- Bytyçi I, Penson PE, Mikhailidis DP, Wong ND, Hernandez AV, Sahebkar A, et al. Prevalence of statin intolerance: a meta-analysis. Eur Heart J. 2022;43:3213-23. https://doi.org/10.1093/eurheartj/ehac015.