José M. Bonorino1 MTSAC, Jorge A. Bilbao1 MTSAC, Nicolás A. Torres1, Mateo Iwanowski1, Emilia M. Spaini1, Agustina F. Gallegos1, Renzo Melchiori1, María E. Aris Cancela2, Horacio E. Fernández1 MTSAC, Sergio Baratta1 MTSAC
1 Institute of Cardiology, Hospital Universitario Austral
2 Hematology, Hemostasis and Thrombosis Department, Hospital Universitario Austral
Address for reprints: José M. Bonorino. Av. Presidente Perón 1500, Pilar, Buenos Aires, Argentina. Zip Code: B1629ODT. E-mail: jbonorin@cas.austral.edu.ar
Rev Argent Cardiol 2024;92:101-107. http://dx.doi.org/10.7775/rac.v92.i2.20744
SEE RELATED ARTICLE : Rev Argent Cardiol 2024;92:99-100. http://dx.doi.org/10.7775/rac.v92.i2.20755
ABSTRACT
Background: The Pulmonary Embolism Severity Index (PESI) is used to categorize the risk of death in acute pulmonary embolism (PE). By definition, cancer patients will always have a high simplified PESI score and will be considered at high risk. There is limited information regarding whether patients with an intermediate or high PESI score (≥86 points) and cancer are at greater risk of an unfavorable progression versus those without cancer.
Objectives: To determine whether the presence of active cancer in patients with a PESI score ≥86 points is associated with an unfavorable progression versus those without cancer.
Methods: A retrospective analysis in patients with PE and a PESI score ≥86, between 2008 and 2022. The occurrence of in-hospital mortality (IHM) the use of vasopressor drugs (VDs), and the need for mechanical ventilatory support (MVS) were evaluated in patients with vs. without cancer.
Results: 209 patients were analyzed. The population with cancer was younger than patients without cancer (65 vs 70 years; p=0.006), showed high simplified PESI values more frequently (100% vs 84%; p<0.001), had lower MVS requirement (9% vs 34%; p=0.005), and used fewer VDs (11% vs 23%; p=0.019). However, no difference was observed in IHM rates (12.7% vs 8%; p=NS).
Conclusions: Patients with PE and a PESI score ≥86 who have cancer did not show higher IHM and also had lower MVS and VDs requirement. Therefore, in the studied population, patients with PE and cancer had no greater risk of having an unfavorable progression.
Key words: Pulmonary Embolism – Cancer - Hospital Mortality - Risk Factors - PESI - Pulmonary Embolism Severity Index - Simplified PESI.
RESUMEN
Introducción: El Índice de Severidad del Embolismo Pulmonar (PESI) se utiliza para categorizar el riesgo de mortalidad en el tromboembolismo pulmonar agudo (TEP). Por definición, los pacientes con cáncer siempre presentarán un puntaje PESI simplificado alto y serán considerados de mayor riesgo. Existe información limitada respecto de si los pacientes con PESI intermedio o alto (≥86 puntos) y cáncer activo, tienen mayor riesgo de presentar una evolución desfavorable respecto de aquellos sin cáncer.
Objetivos: Determinar si, en pacientes con TEP y un puntaje PESI ≥86 puntos, la presencia de cáncer activo se asocia a una evolución desfavorable respecto de aquellos sin cáncer.
Material y métodos: Análisis retrospectivo en pacientes con TEP y un puntaje PESI ≥86, entre los años 2008 y 2022. Se evaluó la ocurrencia de muerte intrahospitalaria (MIH), uso de drogas vasopresoras (DV) y necesidad de asistencia respiratoria mecánica (ARM) en los pacientes con vs. sin cáncer.
Resultados: Se analizaron 209 pacientes. La población con cáncer, respecto de aquella sin cáncer, resultó ser más joven (65 vs. 70 años; p=0,006), presentó valores de PESI simplificado altos con mayor frecuencia (100 % vs. 84 %; p<0,001), tuvo menor requerimiento de ARM (9 % vs. 34 %; p=0,005) y menor uso de DV (11 % vs. 23 %; p=0,019), aunque no se observaron diferencias en las tasas de MIH (12,7 % vs. 8 %; p=NS).
Conclusiones: Los pacientes con TEP y un puntaje PESI ≥86 con cáncer no presentaron mayor MIH e incluso tuvieron menor requerimiento de ARM y DV. En la población estudiada, los pacientes con TEP y cáncer no tuvieron mayor riesgo de presentar una evolución desfavorable.
Palabras clave: Embolia pulmonar - Cáncer - Mortalidad hospitalaria - Puntuaciones de riesgo - PESI - Pulmonary Embolism Severity Index - PESI simplificado
Received: 09/30/2023
Accepted: 03/05/2024
INTRODUCTION
Acute pulmonary embolism (PE) is the second cause of death in cancer patients, only preceded by deaths resulting from cancer itself, (1) and it is estimated that up to 20% of patients with cancer will experience PE. (2,3)
Argentina has an intermediate to high cancer incidence, which, in addition to cardiovascular diseases, account for more than 40% of deaths based on the latest epidemiology registries. (4)
Cancer-associated PE (CA-PE) shows higher mortality in several registries, regardless of the characteristics of cancer (5-7) and any related symptoms. (8,9)
The Pulmonary Embolism Severity Index (PESI) is a useful prognostic method to estimate in-hospital death and 30-day mortality. Complications or fatal events in low or very low risk patients (<86 points) are lower than 1.9% and 1.1%, respectively. (10)
Based on the simplified version of PESI (sPESI), for individuals with a sPESI score <1, 30-day mortality was 1.1% (low risk), while for those with a score ≥1, it was 8.9% (high risk).(11) Both scores have been validated in Argentina and have shown good mortality correlation. (12)
Current guidelines recommend the use of any of these two scores (PESI or sPESI) for risk stratification. (13-15) Those with low PESI or sPESI scores may receive anticoagulants alone and, eventually, early hospital discharge. (16)
Patients with CA-PE will inevitably have a high sPESI score, as cancer history is considered a risk variable by definition and, therefore, will be considered at high risk when using this classification.
There is limited information on whether patients with CA-PE and an intermediate or high PESI (≥86 points) have a higher risk of an unfavorable progression versus those without cancer. It is also unknown if patients with CA-PE might have a better prognosis with a PESI <86 points.
METHODS
A retrospective analysis of a prospective single-center cohort from an Argentine University Hospital in patients hospitalized with PE between 2008 and 2022 and with a PESI score ≥86 points. The objective of this study is to determine whether in patients with PE and a PESI score ≥86 points the presence of active cancer is associated with an unfavorable progression versus those without cancer.
The incidence of in-hospital mortality (IHM), the use of vasopressor drugs (VDs), and the need for mechanical ventilatory support (MVS) were evaluated in patients with and without CA-PE. In addition, IHM and the incidence of BARC bleeding (17) were evaluated in patients with CA-PE and a PESI score <86 points.
Active cancer was defined as solid or hematologic malignancy treated with chemotherapy and/or radiotherapy within the past year, or with no active treatment but under palliative care. Patients with advanced cancer and brain neoplasms were included, whether anticoagulants or reperfusion therapy were contraindicated or not. Skin neoplasms, except for melanoma, were not considered active cancer. Right ventricular (RV) dilatation was defined as a RV/left ventricular diameter ratio >0.9 upon four-chamber view, and RV failure was defined as RV dilatation with at least one of the following: free wall hypokinesia, tricuspid annular plane systolic excursion (TAPSE) lower than 16 mm, and/or interventricular septal bulge.
Data were collected from the medical record and stored in an encrypted database approved by the Institutional Research Ethics Committee. All informed consents for the anonymous use of data were obtained, and the protocol was approved by our Institutional Bioethics Committee under regulation number 19-041.
Mean and standard deviation, or median and interquartile range (IQR) 25-75 were used to describe quantitative variables based on whether distribution was normal or not. Qualitative variables were displayed as frequency and percentage tables. For quantitative variables comparison, the t test or Wilcoxon test were used for single or paired samples, according to each case. For dichotomous variables comparison, the χ2 or McNemar tests were used. A multivariate logistic regression analysis was conducted to evaluate the stability of results on IHM, MVS, and use of VDs, adjusted by age, sex, hypertension (HT), diabetes, and active cancer. A two-tailed p value <0.05 was considered significant. The analysis was performed with Stata/SE v13.0 (StataCorp, United States).
RESULTS
Out of 456 patients hospitalized due to PE diagnosis and included in our registry, 209 patients with a PESI score ≥86 were evaluated. From this population, 126 (60.3%) patients had active cancer and 83 (39.7%) patients had no cancer (Figure 1). For the cancer population, 23% were incidental PE and 12.6% had limited therapeutic effort. An elevated prevalence of solid tumors was recorded: lung (17.4%), gastrointestinal (17.4%), breast (11.1%), biliopancreatic (10.3%), gynecological (9.5%), renal vesical (8.7%), prostatic (7.1%), central nervous system (4.7%), and other malignancies (6.3%), with a lower prevalence of hematologic malignancies (7.1%). (Figure 2). Baseline characteristics, prognostic markers, and reperfusion therapy prescribed for each population, with and without cancer, are shown in Table 1. The population with CA-PE was younger than the popula tion without (65 vs 70 years; p=0.006), respectively. Patients with CA-PE had a lower prevalence of HT (48% vs 72%; p<0.001) but a higher prevalence of diabetes (19% vs 8%; p= 0.045). By definition, all patients with CA-PE had a high sPESI score, with higher prevalence versus patients without cancer (100% vs 84%; p<0.001). There was an elevated rate of anticoagulant use in patients with CA-PE and in those without (97.6% vs 97.5%; p=NS). The population with CA-PE had lower RV dilatation versus the population without cancer (16.6% vs 50.1%; p<0.001) and lower RV dysfunction (13.4% vs 37.3%; p<0.001). CA-PE patients were prescribed some kind of reperfusion therapy less frequently (9.6% vs. 32.6%; p<0.001), with less fibrinolytic drugs indication. (Table 1). No differences were observed in terms of major bleeding rates (BARC ≥3) among patients with CA-PE versus patients without cancer (2.3% vs 4.8%; p=NS). There was no fatal bleeding (type 5 BARC), and a case of surgical bleeding was recorded (type 4 BARC) in each group.
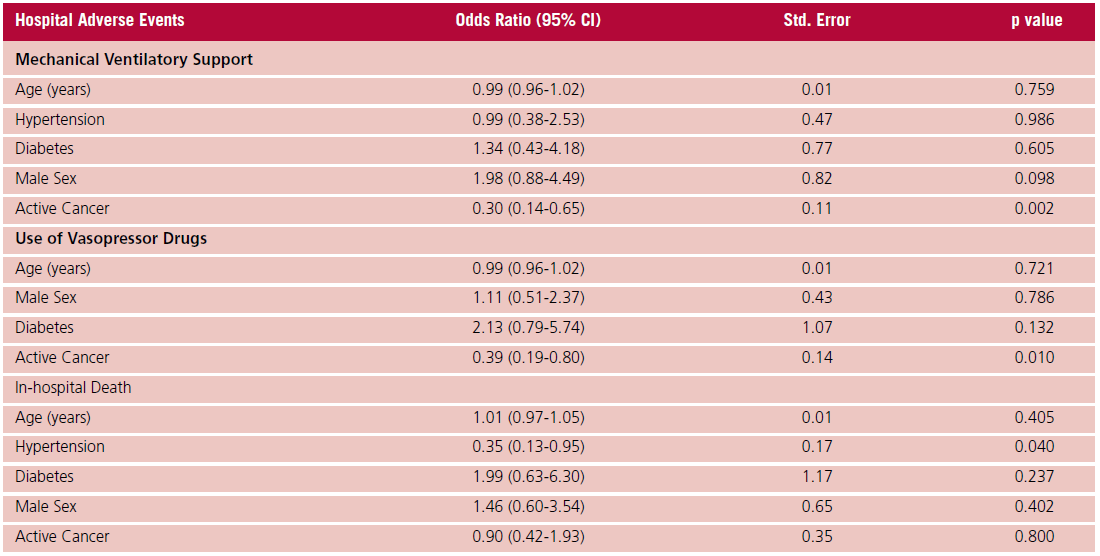
Patients with CA-PE did not show any differences in IHM rates versus patients without cancer (12.7% vs 8.4%; p=NS), even though they had a lower requirement of MVS (9.5% vs 24%; p=0.005) and use of VDs (11% vs 23%; p=0.019) (Table 2). Absence of active cancer was correlated with a higher risk of MVS, but not higher IHM, in multivariable analysis adjusting for age, sex, HT and diabetes (Table 3). The statistical model had good graphic and statistical calibration, with a ROC area under the curve of 0.70 (95% CI 0.65- 0.73) and a Hosmer-Lemeshow test p value = 0.521.
None of the patients with a PESI score <86 points, either with cancer (n=24) or without cancer (n=151), died during hospitalization. In cancer patients, a PESI score <86 vs ≥86 points was not useful to predict IHD (0% vs 12%; p=NS). No patient with CA-PE and PESI <86 had bleeding or right ventricular dysfunction, and only 8.3% had mild RV dilatation.
Fig. 1. Flow diagram. Patients hospitalized due to acute pulmonary embolism with a PESI score ≥86 points, with active cancer vs with no active cancer.
Fig. 2. Patients with acute pulmonary embolism and active cancer (n=126). Types of malignancy.
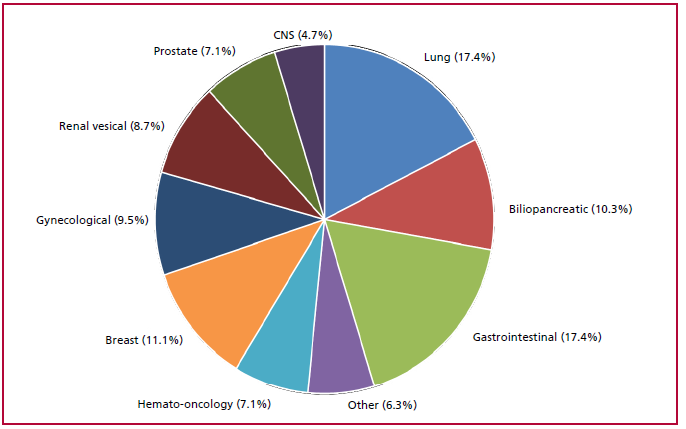
CNS = central nervous system
Table 1. Clinical characteristics, prognostic markers, and reperfusion therapy prescribed for patients with pulmonary embolism (PE) and PESI ≥86 points.
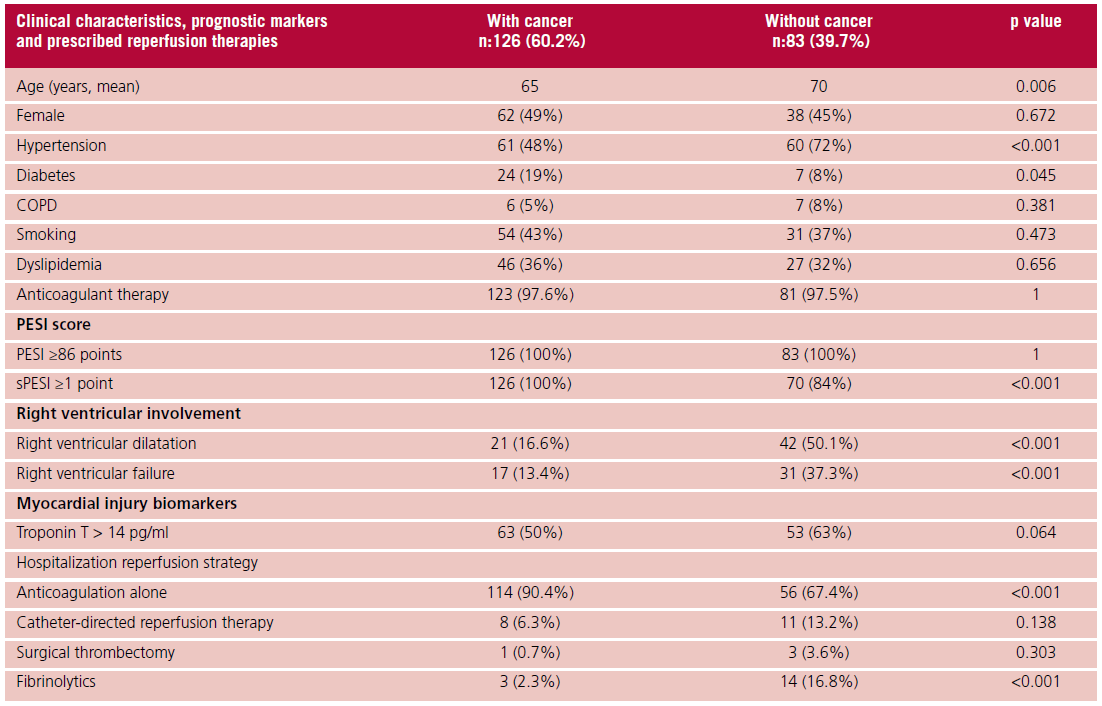
COPD: chronic obstructive pulmonary disease; sPESI: simplified PE Severity Index. Right ventricular (RV) failure: RV dilatation with at least one of the following: free wall hypokinesia, TAPSE < 16 mm, interventricular septal bulge.
Table 2. Incidence of in-hospital adverse events in patients with PESI score ≥ 86, with vs. without cancer
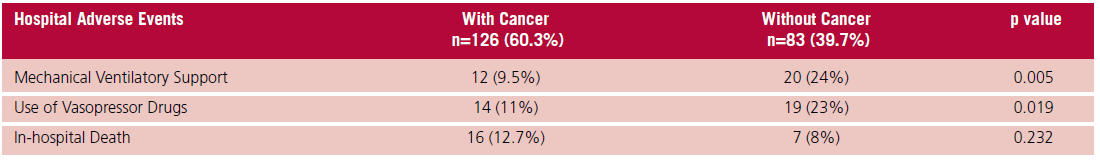
Table 3. Multivariate analysis for the prediction of hospital adverse events.
DISCUSSION
PESI score assessment is part of the initial stratification of EP patients, whether they have active cancer or not. In this sense, it is assumed that patients with CA-PE have a higher risk than those with no active cancer. Patients with a high PESI score, RV dilatation, and elevated troponin are considered intermediate to high risk patients in terms of an unfavorable progression. Use of some kind of reperfusion therapy is considered for these patients, as well as patients with hemodynamic instability. Thus, the importance of establishing whether the sole presence of active cancer might have an impact on prognosis for PE patients. Several models attempt to identify patients with CA-PE and a low risk of complications who might be offered a therapeutic alternative that may avoid or reduce hospitalization. (18)
As observed in other registries, (19,20) IHM in patients with PE and a PESI score ≥86 points was high, even though no differences were observed in the population without cancer vs patients with CA-PE (8% vs 12.7%, p=NS). Unlike other studies, (21,22) where CA-PE patients are older than those without cancer, the CA-PE population was younger in our study. The multivariate analysis for IHM, adjusted for age, sex, HT, and diabetes, failed to show that cancer is an independent adverse prognostic marker. However, note that patients with cancer had lower HT prevalence, which may have affected the result. It is also likely that a higher risk of MVS and VDs requirement in patients without cancer is related to a higher prevalence in this population with adverse prognostic predictors, with greater dilatation and RV dysfunction. Also, reperfusion therapies were more commonly prescribed in the population without cancer, suggesting a more severe condition for these patients. These findings suggest that the PESI has mainly a negative predictive value for adverse prognosis; a high PESI score requires concomitant RV dilatation and/or dysfunction, and elevated troponin to predict IHM.
While this study did not evaluate other predictive models useful for CA-PE, there are various potentially helpful prognostic scores. The POMPE-C predictive model has adequate prognostic accuracy for 30-day mortality and a better performance than PESI. According to Kline et al., the ROC area under the curve for POMPE-C was 0.84 (95% CI 0.82-0.88). No patient with POMPE-C ≤5% died within 30 days, while 77% of patients with POMPE-C >50% died during 30-day follow-up. (23) RIETE-VTE and the modified Ottawa score are risk models that might be useful to identify patients with CA-PE and a low risk of IHD. In a study by Pfaundler et al., no patient classified as low risk according to RIETE-VTE models or the modified Ottawa score died within 30 days. (24) The HESTIA model showed that a total zero score was consistent with a very low rate of adverse events, even though it has not been sufficiently validated in patients with active cancer. (25) Roy et al. compared the HESTIA model and sPESI to determine which of them might be more accurate to identify patients adequate for outpatient treatment. In the HESTIA model arm, the primary endpoint at 90 days occurred in 2.96% vs 1.40%; p=NS. Both scores showed similar efficacy and safety. (26)
Prognosis for CA-PE patients might be more affected by advanced cancer and overall patient condition than by risk scores such as PESI. Li et al. showed that, in a CA-PE population, patients who died within 30 days had a higher rate of metastatic cancer and an Eastern Cooperative Oncology Group performance status ≥2. This study showed that the incidence of death and adverse events was associated with more specific models for CA-PE patients (RIETE and POMPE-C), rather than with more general models (PESI and HESTIA) or the modified Ottawa score. Except for the HESTIA score, with a 73.5% sensitivity, the other four scores were highly sensitive (>94%) for mortality at 30 days. All these models showed an excellent negative predictive value (>92%) for mortality at 30 days, especially, for RIETE and POMPE-C scores (98.6% and 96.5%, respectively). (18) Other models such as EPIPHANY and COMMAND VTE are also useful for patients with CA-PE, even though they have not been sufficiently validated. (27,28)
Among the multiple limitations of this study, the single-center nature must be noted. Therefore, prevalence of certain types of tumors could differ from other sites. There was no 30-day follow-up, incidental PE occurrences were included, no outpatients were enrolled, and neither RV dilatation nor RV dysfunction were considered for the multivariate analysis.
The present study shows that, in our setting, patients with CA-PE and PESI ≥86 points have high IHM, even though not higher than in patients without cancer. In addition, it is suggested that the sPESI might overestimate the risk in patients with CA-PE, as no patient with PESI <86 points either with or without cancer died during hospitalization. No further studies are needed to check these findings and evaluate the use of PESI and other prognostic models for patients with CA-PE.
CONCLUSIONS
Conflicts of interest
None declared.
(See conflicts of interest forms on the website).
Funding
Support Sources: CEDECAFO, Hospital Universitario Austral Institute of Cardiology.
https://creativecommons.org/licenses/by-nc-sa/4.0/

©Revista Argentina de Cardiología
REFERENCES
- Khorana AA, Francis CW, Culakova E, Kuderer NM, Lyman GH. Thromboembolism is a leading cause of death in cancer patients receiving outpatient chemotherapy. J Thromb Haemost. 2007;5:632-4. https://doi.org/10.1111/j.1538-7836.2007.02374.x
- Lyman GH. Venous thromboembolism in the patient with cancer: focus on burden of disease and benefits of thromboprophylaxis. Cancer. 2011;117:1334-49. https://doi.org/10.1002/cncr.25714
- Mulder FI, Horváth-Puhó E, van Es N, van Laarhoven HWM, Pedersen L, Moik F, et al. Venous thromboembolism in cancer patients: a population-based cohort study. Blood. 2021;137:1959-69. https://doi.org/10.1182/blood.2020007338
- https://www.argentina.gob.ar/salud/instituto-nacional-del-cancer/estadisticas/mortalidad
- Brunson AM, Keegan THM, Mahajan A, White RH, Wun T. Cancer associated venous thromboembolism: incidence and impact on survival. Thromb Res.2018;164:178-9. https://doi.org/10.1016/j.thromres.2018.02.012
- Chew HK, Wun T, Harvey D, Zhou H, White RH. Incidence of venous thromboembolism and its effect on survival among patients with common cancers. Arch Intern Med.2006;166:458-64. https://doi.org/10.1001/archinte.166.4.458
- Sørensen HT, Mellemkjær L, Olsen JH, Baron JA. Prognosis of cancers associated with venous thromboembolism. N Engl J Med.2000;343:1846-50. https://doi.org/10.1056/nejm200012213432504
- Dentali F, Ageno W, Giorgi Pierfranceschi M, Imberti D, Malato A, Nitti C, et al. Prognostic relevance of an asymptomatic venous thromboembolism in patients with cancer. J Thromb Haemost. 2011;9:1081-3. https://doi.org/10.1111/j.1538-7836.2011.04259.x
- den Exter PL, Hooijer J, Dekkers OM, Huisman MV. Risk of recurrent venous thromboembolism and mortality in patients with cancer incidentally diagnosed with pulmonary embolism: a comparison with symptomatic patients. J Clin Oncol. 2011;29:2405-9. https://doi.org/10.1200/JCO.2010.34.0984
- Aujesky D, Obrosky DS, Stone RA, Auble TE, Perrier A, Cornuz J, et al. Derivation and validation of a prognostic model for pulmonary embolism. Am J Respir Crit Care Med.2005;172:1041-6. https://doi.org/10.1164/rccm.200506-862OC
- Jiménez D, Aujesky D, Moores L, Gómez V, Lobo JL, Uresandi F, et al; RIETE Investigators. Simplification of the pulmonary embolism severity index for prognostication in patients with acute symptomatic pulmonary embolism. Arch Intern Med.2010;170:1383-9. https://doi.org/10.1001/archinternmed.2010.199
- Burgos LM, Scatularo CE, Cigalini IM, Jauregui JC, Bernal MI, Bonorino JM, et al. External validation of prognostic scores for in-hospital and 30-day mortality in patients with pulmonary embolism in Argentina. MEDICINA (B AIRES).2020;80:462-72
- Konstantinides S, Meyer G, Becattini C, Bueno H, Geersing G, Harjola V y col. Guıía ESC 2019 para el diagnóstico y tratamiento de la embolia pulmonar aguda. Rev Esp Cardiol. 2020;73:497.e1-497. e58. https://doi.org/10.1016/j.recesp.2019.12.030
- Ubaldini J, Bilbao J, Spenatto M, Bonorino J et al. Consenso de enfermedad tromboembólica aguda. SAC 2016. Rev Argent Cardiol 2016;84:74-91.
- Flores LA, Ubaldini J, Kenar MR, Bilbao J, Bonorino J, Ceresetto J, et al. Guías de Manejo y Tratamiento de la Enfermedad Tromboembólica Aguda. Rev Arg de Ter Int. [Internet]. 7 de octubre de 2019 [citado 18 de septiembre de 2023];36(2). Disponible en: https://revista.sati.org.ar/index.php/MI/article/view/642
- Roy PM, Penaloza A, Hugli O, Klok FA, Arnoux A, Elias A, et al; HOME-PE Study Group. Triaging acute pulmonary embolism for home treatment by Hestia or simplified PESI criteria: the HOME-PE randomized trial. Eur Heart J. 2021;42:3146-57. https://doi.org/10.1093/eurheartj/ehab373
- Mehran R, Rao SV, Bhatt DL, Gibson CM, Caixeta A, Eikelboom J, et al. Standardized bleeding definitions for cardiovascular clinical trials: a consensus report from the Bleeding Academic Research Consortium. Circulation. 2011;123:2736-47. https://doi.org/10.1161/CIRCULATIONAHA.110.009449
- Li X, Hu Y, Lin P, Zhang J, Tang Y, Yi Q, et al. Comparison of Different Clinical Prognostic Scores in Patients with Pulmonary Embolism and Active Cancer. Thromb Haemost. 2021;121:834-44. https://doi.org/10.1055/a-1355-3549
- Pollack CV, Schreiber D, Goldhaber SZ, Slattery D, Fanikos J, O'Neil BJ, et al. Clinical characteristics, management, and outcomes of patients diagnosed with acute pulmonary embolism in the emergency department: initial report of EMPEROR (Multicenter Emergency Medicine Pulmonary Embolism in the Real World Registry). J Am Coll Cardiol. 2011;57:700-6. https://doi.org/10.1016/j.jacc.2010.05.071
- Cigalini I, Igolnikof D, Scatularo C, et al. Tromboembolismo pulmonar agudo en la Argentina. Registro CONAREC XX. Rev Argent Cardiol 2019;87:137-45.
- Cohen AT, Katholing A, Rietbrock S, Bamber L, Martinez C. Epidemiology of first and recurrent venous thromboembolism in patients with active cancer. A population-based cohort study. Thromb Haemost. 2017;117:57-65. https://doi.org/10.1160/TH15-08-0686
- Cai B, Bedayat A, George E, Hunsaker AR, Dill KE, Rybicki FJ, et al. Malignancy and Acute Pulmonary Embolism: Risk Stratification Including the Right to Left Ventricle Diameter Ratio in 1596 Subjects. J. Thorac. Imaging. 2013;28:196-201. https://doi.org/10.1097/RTI.0b013e3182870bf9
- Kline JA, Roy PM, Than MP, Hernandez J, Courtney DM, Jones AE, et al. Derivation and validation of a multivariate model to predict mortality from pulmonary embolism with cancer: The POMPE-C tool. Thromb Res.2012;129:e194-9. https://doi.org/10.1016/j.thromres.2012.03.015
- Pfaundler N, Limacher A, Stalder O, Méan M, Rodondi N, Baumgartner C, et al. Prognosis in patients with cancer-associated venous thromboembolism: Comparison of the RIETE-VTE and modified Ottawa score. J Thromb Haemost.2020;18:1154-61. https://doi.org/10.1111/jth.14783
- Zondag W, Mos IC, Creemers-Schild D, Hoogerbrugge AD, Dekkers OM, Dolsma J, et al; Hestia Study Investigators. Outpatient treatment in patients with acute pulmonary embolism: the Hestia Study. J Thromb Haemost.2011;9:1500-7. https://doi.org/10.1111/j.1538-7836.2011.04388.x
- Roy PM, Penaloza A, Hugli O, Klok FA, Arnoux A, Elias A, et al; HOME-PE Study Group. Triaging acute pulmonary embolism for home treatment by Hestia or simplified PESI criteria: the HOME-PE randomized trial. Eur Heart J. 2021;42:3146-57. https://doi.org/10.1093/eurheartj/ehab373
- Font C, Carmona-Bayonas A, Beato C, Reig Ò, Sáez A, Jiménez- Fonseca P, et al; Asociación para la Investigación de la Enfermedad Tromboembólica de la región de Murcia. Clinical features and short-term outcomes of cancer patients with suspected and unsuspected pulmonary embolism: the EPIPHANY study. Eur Respir J. 2017;49:1600282. https://doi.org/10.1183/13993003.00282-2016
- Yamashita Y, Morimoto T, Amano H, Takase T, Hiramori S, Kim K, et al; COMMAND VTE Registry Investigators. Usefulness of Simplified Pulmonary Embolism Severity Index Score for Identification of Patients With Low-Risk Pulmonary Embolism and Active Cancer: From the COMMAND VTE Registry. Chest. 2020;157:636-44. https://doi.org/10.1016/j.chest.2019.08.2206s