SCIENTIFIC LETTERS
http://dx.doi.org/10.7775/rac.v91.i3.20638
Acute Myocardial Infarction Due to Coronary Embolism
in a Young Woman with Mechanical Aortic Valve Prosthesis and Anomalous Origin
of Two Coronary Vessels: A Case Report
Heart valve disease affects approximately
2.5% of adults in developed countries. Since 1960, valve replacement with
mechanical prostheses is one of the therapeutic alternatives for the management
of valve disease. Its main complication is the development of thrombosis or
embolic phenomena, with an estimated annual incidence of 0.3-1.3% and 0.7-6%,
respectively. (1) The risk is increased in the first months
of implantation, depending on its anatomical position and its association with
other thromboembolic risk factors (e.g., atrial fibrillation).
We present a case of ST-segment elevation
acute myocardial infarction in a young woman with a prosthetic aortic valve who
had voluntarily discontinued anticoagulation.
A 23-year-old woman, from the Colombian
Pacific region, with a history of mitral regurgitation and mechanical valve
prosthesis implantation at 8 years of age, was anticoagulated
with warfarin until 2 years ago when she discontinued medical treatment. She
consulted the emergency department due to 8 hours of high-intensity oppressive
chest pain radiating to the right upper limb, with no other associated symptoms.
On physical examination, she was afebrile, with blood pressure of 121/76 mmHg,
heart rate 82 bpm, and respiratory rate 19 rpm.
Auscultation revealed a grade III/VI holosystolic
murmur in the mitral focus, and a grade III/VI diastolic
murmur in the aortic focus, with no signs of acute heart failure and no other
relevant findings. The electrocardiogram showed sinus rhythm with ST-segment
elevation from V1 to V3 and inferior ST-segment depression, with the presence
of pathological Q waves in leads I and aVL, and signs of left ventricular enlargement. Emergency
coronary angiography was performed 12 hours after admission, which documented a
total occlusion of chronic appearance in the mid-proximal segment of the left
anterior descending artery (Figure 1) and a thrombotic lesion in the first obtuse marginal
of the circumflex artery, with 90% stenosis (Figure 2), without other angiographically
significant lesions. Loss of mobility of one of the hemidiscs
of the double-disc mechanical valve prosthesis was evidenced, due to in situ
thrombus.
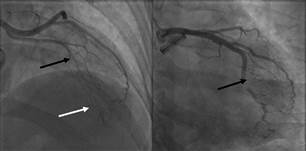
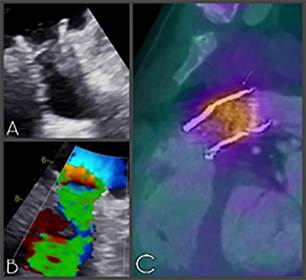
Fig. 1. Left: Chronic
occlusion of the left anterior descending artery at the junction of the
proximal to middle segment (black arrow) with heterocoronary
and homocoronary collateral circulation (white
arrow). Right: Obtuse
marginal artery with filling defect compatible with thrombus generating subocclusion and TIMI 2 flow (black arrow).
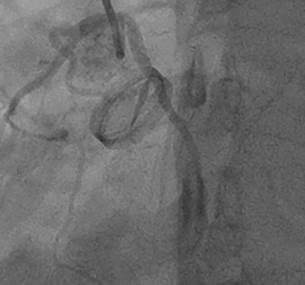
Fig. 2. Loss of
mobility of one of the hemidiscs of a mechanical double disc prosthesis due to in situ thrombus
An anomalous origin of the right coronary
artery and a second marginal obtuse artery, independently from the left coronary
sinus were demonstrated as incidental findings. Due to the high risk of
prosthetic thrombosis, it was initially decided to anticoagulate
the patient with low molecular weight heparin and warfarin until international
normalized ratio (INR) goals were reached. Laboratory tests showed positive
cardiac troponin I (6.53 ng/mL for a normal upper limit
of 0.12 ng/mL). Transesophageal echocardiogram revealed akinesia
without thinning of the anterolateral and inferolateral
walls, with a left ventricular ejection fraction of 47% by Simpson's method. It
also showed mechanical aortic prosthesis in adequate position with restriction
of the posterior leaflet movement, and presence of pannus
and marked turbulence in the antegrade flow, with
maximum velocity of 3.2 m/s and a mean gradient of 22.5 mm Hg, as well as
severe mitral regurgitation secondary to perforation of the anterior leaflet.
With these findings, she underwent aortic
valve prosthesis replacement using a n° 23 Medtronic
mechanical prosthesis. Enlargement of the aortic annulus with a heterologous
pericardial patch using Manougian´s technique, and
repair of the mitral valve with closure of the anterior leaflet orifice were
performed. Fresh thrombi in the aortic mechanical prosthesis at the hinge level
of both discs and severe subvalvular pannus were found. It was not possible to perform
revascularization of the anterior descending artery as its course could not be
visualized due to the presence of epicardial-pericardial
adhesions from the previous surgery, so coronary angioplasty was indicated. The
second coronary angiography performed 25 days after admission revealed complete
resolution of the thrombotic lesion in the obtuse marginal artery. The total
occlusion of the left anterior descending artery persisted, but it was not
possible to perform percutaneous revascularization as it was a vessel with a
small caliber. It was decided to continue medical treatment, accompaniment by
the Psychology and Education service, and she was discharged after 41 days of
hospitalization, without complications, with an INR of 3.2 and indications for
strict medical control.
Ischemic heart disease is the leading
cause of death worldwide, mainly associated with atherosclerosis. Significant
atherosclerotic lesions are not found in up to 7% of cases. Coronary embolism
is a cause of non-atherosclerotic infarction, and it is estimated that it
represents 3% of all myocardial infarctions. It generally affects the left
coronary circulation, (1) as in the case of our patient.
The main associated causes are atrial
fibrillation, cardiomyopathies, presence of prosthetic valves, endocarditis,
tumors, and prothrombotic conditions. Coronary
thrombosis associated with acute infection by SARS-CoV-2 during the pandemic
has been reported for this entity. (2) Before the use of prosthetic valves, endocarditis was
the main cause of death; now atrial fibrillation is mainly considered. (1) Currently, prosthetic valve replacement
is the gold standard for the management of severe valve disease in patients
with low or intermediate surgical risk. Mechanical valves have a longer life,
but are prothrombotic, which requires indefinite
anticoagulation to prevent valve thrombosis and embolic events.
There are three types of coronary
embolism: direct, paradoxical, and iatrogenic. Direct coronary embolism occurs
when an embolus enters the coronary circulation from the left ventricle, left
atrial appendage, pulmonary veins, and the aortic or mitral valve. (3)
The clinical, electrocardiographic, and
echocardiographic manifestations of myocardial infarction due to coronary
embolism are indistinguishable from infarction of atherosclerotic origin, and
it should be suspected in patients with prothrombotic
risk factors who present with sudden chest pain. (4)
There are currently no guidelines for the
management of coronary embolism. Intracoronary thrombus aspiration vs. angioplasty
alone has been tested in patients with ST-segment-elevation myocardial infarction,
without demonstrating an additional benefit in mortality. However, patients
with a high thrombotic burden, such as patients with coronary embolism, could
benefit more from this measure. (5) In cases of coronary embolism, systemic thrombolysis
with tissue plasminogen activator (t-PA) has been reported to be successful in
restoring coronary flow. Karakoyun et al (5) effectively and safely treated three
patients with coronary embolism associated with prosthetic valves with low-dose
intravenous t-PA. Similarly, intravenous infusion of bivalirudin
for 48 hours in coronary embolism of the distal right coronary artery has been
described, with complete resolution of the thrombus without major bleeding. (6) Other therapies include balloon
angioplasty, which has been shown to be successful in restoring blood flow,
both as isolated treatment and as adjunctive therapy to thrombotic aspiration. (5)
In conclusion, we describe the case of a
young woman with mechanical aortic valve prosthesis who voluntarily
discontinued anticoagulation and who presented an acute myocardial infarction
due to coronary embolism. This condition is potentially fatal, so adherence to
pharmacological treatment and education about the disease is essential in a
patient at high risk of thrombosis. Permanent anticoagulation, strict clinical
monitoring and education are the most important measures to prevent new events.
Conflicts of interest
None declared.
(See authors' conflict of interests forms on
the web/Additional material.)
Ethical considerations
Not applicable.
Fernando Araque-Villaquirán, Raúl Vallejo-Serna, Mónica Fernandes
Pineda, Álvaro Herrera-Escandón
Department of
Internal Medicine, Universidad del Valle. Cali,
Colombia.
Hospital
Universitario del Valle, Santiago de Cali - Colombia
Address for reprints: Mónica Fernandes Pineda Mailing
address: 760032 Email: monica.fernandes.pineda@gmail.com
1. Roudaut R, Serri K, Lafitte S.
Thrombosis of prosthetic heart valves: diagnosis and therapeutic
considerations. Heart (British Cardiac Society) 2007 ;93
:137-42. https://doi.org/10.1136/hrt.2005.071183
2. Prizel KR, Hutchins GM, Bulkley
BH. Coronary artery embolism and myocardial infarction.
Ann Intern Med 1978;88:155-61. https://doi.org/10.7326/0003-4819-88-2-155.
3. Chikkabasavaiah N, Rajendran R.
Percutaneous coronary intervention for coronary thrombo
embolism during balloon mitral valvuloplasty in a
pregnant woman. Heart Lung Circ 2016;25:e29-31.
4. Lacey MJ, Raza S, Rehman H, Puri R, Bhatt DL, Kalra A. Coronary Embolism: A Systematic Review. Cardiovasc Revasc Med. 2020;21:367-74. https://doi.org/10.1016/j.carrev.2019.05.012.
5. Karakoyun S, Gürsoy MO, Kalçık M, Yesin M, Özkan M. A case series of prosthetic
heart valve thrombosis-derived coronary embolism. Turk Kardiyol Dern Ars. 2014;42:467-71.
https://doi.org/10.5543/tkda.2014.05031
6. Steinwender C, Hofmann R, Hartenthaler
B, Leisch F. Resolution of a coronary embolus by intravenous
application of bivalirudin. Int
J Cardiol. 2009;132:e115-6. https://doi.org/10.1016/j.ijcard.2007.08.032.
http://dx.doi.org/10.7775/rac.v91.i3.20643
Extracorporeal Ventricular Assistance in In-hospital
Cardiac Arrest: A Feasible Reality in Our Setting?
Extracorporeal cardiopulmonary resuscitation (ECPR) is
the use of extracorporeal membrane oxygenation (ECMO) in patients in whom
standard cardiopulmonary resuscitation (SCPR) measures do not achieve a
sustained return of spontaneous circulation after cardiac arrest (CA). (1) Patients undergoing ECMO implantation during or
immediately after CA have a particularly unfavorable prognosis. (2)
Despite there are no current systematic recommendations
on the indication of ECMO in CA, it could be considered an emerging therapy in
selected cases when SCPR fails. (3) At present, no randomized controlled trials have been reported
comparing the results of ECPR versus SCPR in in-hospital CA (IHCA). (1) Though numerous cohort studies have shown that this
therapy is associated with a higher survival rate until discharge, and with
favorable neurological results, (4) to our knowledge, limited information has been published in our
setting.
The aim of this study was to analyze and report the
characteristics and clinical results of a retrospective and consecutive cohort
of adult patients treated with ECPR after IHCA in a high complexity center of
Argentina.
Patients over 18 years of age treated with venoarterial (VA) ECMO for IHCA between 2014 and 2022 were
analyzed. The study included patients with witnessed IHCA, possibly of cardiac
origin (mainly ventricular tachycardia or ventricular fibrillation as initial
rhythm, extending for more than 20 minutes), (1) even with adequate CPR since its onset. Patients with CA during cardiac
surgery were excluded from the study. Table 1 summarizes the inclusion criteria
for ECPR at our center.
An analysis of the ventricular assistance database,
which is prospectively completed, including among its main variables, demographic
characteristics, information on the type of ventricular assistance, complications,
relevant clinical events and clinical evolution, biochemical and
echocardiographic predictors was performed. Regarding relevant clinical events,
two types of survival were evaluated:
- Survival in ECMO: It assesses survival in ECMO and
up to 24 hours from ventricular assistance weaning. In this case, the reasons
for weaning from ECMO are cardiac function recovery or heart transplantation.
- Survival at discharge. It evaluates survival at hospital
discharge, either by release from hospital or referral to another healthcare
center (e.g., third level of rehabilitation).
In addition, neurological complications, brain death
(irreversible loss of consciousness and neurovegetative
functions, including breathing capacity) and stroke (acute neurological focus
and new ischemic or hemorrhagic changes in brain computed tomography
) were analyzed.
The analysis included 8 patients, representing 11.9%
of VA ECMO implanted during this period in the center. Median (interquartile
range, IQR) age was 46 years (IQR 30-58) and 66% were women. Three patients
had history of hypertension and dyslipidemia and one of diabetes. No patient
presented with previous history of obstructive pulmonary disease, chronic
kidney disease, stroke, peripheral vascular disease, atrial fibrillation, or
anemia.
Three patients presented with acute coronary syndrome,
two with electrical storm and the remaining causes were peripartum
cardiomyopathy, myocarditis, and unidentified restrictive cardiomyopathy.
Cannulation was peripheral in 87.5% of cases (7 patients). The
same number of patients required use of intra-aortic balloon pump, and 2 cases
needed surgical left ventricular decompression, through pulmonary vein venting.
In all the cases, ECMO was implanted as bridge to recovery.
Median circulatory assistance duration was 5 days (IQR
2-8). Successful VA ECMO weaning was achieved in 5 patients.
The rate of survival in VA ECMO was 62.5% (n=5) and at
discharge 37.5% (n=3). The cause of death was non-cardiovascular in 4 of the 5
deaths.
Complications included major hemorrhage (66%), non-dialytic acute kidney failure (66%), infection (33%),
seizures (11%) and thromboembolic complications (33%). No brain death was
reported, and one patient suffered an ischemic stroke.
Median follow-up after discharge was 14 months (IQR
7-30). One of the 3 surviving patients is on the waiting list for elective
heart transplantation, and 2 are followed-up with preserved biventricular
function.
There is an increasing worldwide use of ECPR as a
rescue technique in patients with refractory CA. Although controlled randomized
trials are still missing demonstrating its efficacy in this setting, observational
studies have reported 20% to 40% survival. (5) Currently, there is no sufficient data available to identify patients
who could benefit from ECPR. It is internationally recommended to establish
agreed inclusion criteria in each center to guide physicians on how to balance
the intelligent use of resources among patients who are believed to have a
better probability of survival after CA. (2) In our center, inclusion criteria were standardized since the creation
of the multidisciplinary “ECMO team” (Table 1), considering that decision making for ECPR is often time critical and
influenced by external factors such as hours and day of the week. It is
therefore essential to present with adequate logistics, 24/7 trained staff for cannulation (as it is recommended that ECMO is functioning
within 60 minutes after CA) and for the fast assembly and purge of the device
in the emergency, and healthcare professionals who can detect within 10 minutes
of CA the possible ECPR candidates.
Table 1. Inclusion criteria for ECPR
|
Age <70 years |
|
In-hospital CA |
|
Time of first CA onset <5 minutes |
|
Initial cardiac rhythm: ventricular fibrillation,
ventricular tachycardia, or pulseless electrical activity |
|
Estimated time from CA to ECMO flow <60 minutes |
|
Recovery from intermittent spontaneous circulation
or from recurrent ventricular fibrillation |
|
Absence of previously known life-limiting
comorbidities |
CA: cardiac arrest; ECMO: extracorporeal membrane
oxygenation
Protocols and algorithms endeavor to quickly identify
the cases with higher probability of survival with a favorable neurological
outcome, as well as patients with witnessed CRA in whom high-quality CPR was
quickly administered, and also cardiac arrests with a presumably reversible
disorder, such as acute coronary obstructions. (2) Other factors which may influence ECPR indication are age, cause of CA,
time, comorbidities and cardiac rhythm at CA onset. (3) Recently, the RESCUE-IHCA survival predictive score derived from 1075
patients was published, showing 28% survival at discharge, and identifying 6
variables associated with in-hospital mortality: age, time of day, initial
rhythm, history of kidney failure, type of patient (cardiac vs. non-cardiac and
clinical vs. surgical) and duration of cardiac arrest. (5) The greatest probability of success occurs in a young patient (in some
working teams 50 years of age is considered the limit for ECPR), with few
comorbidities, with a witnessed CA, preferably during daytime (when logistics
is easier and there is more access to trained staff), with adequate CPR maneuvers
performed immediately (preferably in intensive care units), and of cardiac origin,
with a shockable initial rhythm.
Our results are comparable to those reported by the
ELSO (Extracorporeal Life Support Organization) international multicenter
registry, in which ECMO survival was 41%, and at hospital discharge 30% at an
international level, (6) and to the results of the RESCUE-IHCA. (5)
In our center, VA ECMO as treatment for IHCA presented
an acceptable survival at hospital discharge, and it can be considered an
effective treatment in highly selected patients when conventional therapies
fail, being useful and applicable in a country with low and medium income and
limited access to circulatory assist devices. Probably these results cannot be
extrapolated to other centers of the region, as our institution is a referral
VA ECMO high complexity monovalent cardiovascular center, with a developed care
program, more than 7-year experience and currently, with more than 15 implants
per year. Although the number of patients included in this series was low, it
is still a novelty, as it would be the first experience published analyzing the
results of VA ECMO in refractory IHCA in our country.
Conflicts of interest
None declared.
(See authors’ conflicts of interest forms on the
website/ Supplementary material).
Ethical considerations
The study was conducted according to research
principles (Declaration of Helsinki) and was approved by the institutional
Ethics Committee.
Lucrecia María Burgos1, Ana Spaccavento,
Leonardo Seoane1, Juan
Francisco Furmento1, Mariano
Vrancic1, Mirta
Diez1
1 Instituto Cardiovascular de Buenos Aires. Ciudad de Buenos
Aires. Argentina.
Address for reprints: Email: insuficienciacardiaca@icba.com.ar
1. Jacobs I, Nadkarni
V, Bahr J, Berg RA, Billi JE, Bossaert
L, et al; International Liaison Committee on Resuscitation; American Heart
Association; European Resuscitation Council; Australian Resuscitation Council;
New Zealand Resuscitation Council; Heart and Stroke Foundation of Canada; InterAmerican Heart Foundation; Resuscitation Councils of
Southern Africa; ILCOR Task Force on Cardiac Arrest and Cardiopulmonary
Resuscitation Outcomes: Cardiac arrest and cardiopulmonary resuscitation
outcome reports: Update and simplification of the Utstein
templates for resuscitation registries: A statement for healthcare
professionals from a task force of the International Liaison Committee on
Resuscitation (American Heart Association, European Resuscitation Council,
Australian Resuscitation Council, New Zealand Resuscitation Council, Heart and
Stroke Foundation of Canada, InterAmerican Heart
Foundation, Resuscitation Councils of Southern Africa). Circulation 2004;110:3385–97. http://doi.org/10.1016/j.resuscitation.2004.09.008
2. Keebler ME, Haddad EV, Choi CW, McGrane S, Zalawadiya S, Schlendorf KH, et al. Venoarterial
Extracorporeal Membrane Oxygenation in Cardiogenic Shock. JACC Heart Fail 2018;6:503-16. http://doi.org/10.1016/j.jchf.2017.11.017.
3. Richardson ASC, Tonna
JE, Nanjayya V, Nixon P, Abrams DC, Raman L, et al. Extracorporeal Cardiopulmonary Resuscitation in Adults. Interim Guideline Consensus Statement From the Extracorporeal Life Support Organization. ASAIO J
2021;67:221-8. http://doi.org/10.1097/MAT.0000000000001344
4. Klee TE, Kern KB. A
review of ECMO for cardiac arrest. Resusc Plus
2021;5:100083.
5. Tonna JE, Selzman
CH, Girotra S, Presson AP, Thiagarajan RR, Becker LB, et al; American Heart
Association Get With the Guidelines–Resuscitation Investigators. Resuscitation
Using ECPR During In-Hospital Cardiac Arrest
(RESCUE-IHCA) Mortality Prediction Score and External Validation. JACC Cardiovasc Interv 2022;15:237-47. http://doi.org/10.1016/j.jcin.2021.09.032
6. Registro ELSO. Extracorporeal
Life Support Organization. Revisado 17/2/2022.
Extracorporeal Life Support Registry Report. Available
online: https://www.elso.org/Registry/Statistics/InternationalSummary.aspx.
http://dx.doi.org/10.7775/rac.v91.i3.20637
Posterior Embolic Stroke Secondary to Subclavian Artery Thrombosis