http://dx.doi.org/10.7775/rac.v91.i1.20592
REVIEW
ARTICLE
Alcohol Impact on the Heart and Cardiovascular System -
Hypertrophy, Remodeling and Strain Impairment - Contemporary State-of-the-Art
Efecto del alcohol sobre el corazón y el
sistema cardiovascular: hipertrofia, remodelamiento y disminución del strain.
Información actual
Piotr Hamala¹, Karina Wierzbowska-Drabik²
1 Ist Department and
Chair of Cardiology, Medical University of Lodz, Lodz Poland.
2 Internal Diseases
and Clinical Pharmacology Department, Medical University of Lodz, Lodz Poland
Address
for reprints: Piotr Hamala.
E-mail: piotrhamala@gmail.com
ABSTRACT
The recent data show that chronic overuse
of alcohol may lead to cardiovascular dysfunction, starting from traditionally
judged as low ethanol doses, since the burden of arrhythmias, including atrial
fibrillation, increases even in moderate alcohol consumers.
The other common mechanisms of the
disadvantageous impact of ethanol are related to the development of
hypertension and its direct aftermath, hypertrophy, fibrosis, and diastolic
dysfunction.
Since the chance of the reversibility of
cardiac remodeling depends on the early diagnosis of cardiac dysfunction, the
wider application of novel and sensitive methods of myocardial function
assessment, including longitudinal strain of the left and right ventricles, as
well as the adapted protocols for stress echocardiography, should be
recommended.
Keywords: Ethanol - Alcohol Cardiomyopathy - Toxic
Cardiomyopathy
RESUMEN
Datos recientes muestran que el abuso
crónico de alcohol puede conducir a disfunción cardiovascular, a partir de
dosis de etanol tradicionalmente consideradas bajas, ya que la aparición de
arritmias, incluyendo la fibrilación auricular, aumenta aún en consumidores de
alcohol moderados.
Los otros mecanismos comunes del impacto
negativo del etanol están relacionados con el desarrollo de hipertensión y su
consecuencia directa, la hipertrofia, fibrosis y disfunción diastólica.
Debido a que la probabilidad de
reversibilidad del remodelamiento cardíaco depende de un diagnóstico temprano
de disfunción cardíaca, se debería recomendar la aplicación más amplia de
métodos nuevos y más sensibles de evaluación de la función miocárdica,
incluyendo el strain longitudinal ventricular izquierdo y derecho, así como de
los protocolos adaptados a la ecocardiografía de estrés.
Palabras clave: Etanol - Miocardiopatía Alcohólica -
Miocardiopatía Tóxica
Received: 09/02/2022
Accepted: 11/08/2022
INTRODUCTION
According to recent WHO reports, 40% of the world’s adult
population consumes alcohol. In Europe this value is even higher and reaches
60%. To alcohol related diseases belong cardiac arrhythmias, especially atrial
fibrillation, alcohol cardiomyopathy, hypertension, stroke, epilepsy,
depression, hepatic steatosis, and cirrhosis, as well as pancreatitis and
numerous cancers. Alcohol related myocardial damage in early stages is believed
to be reversible after cessation of drinking, but the predictors of
reversibility are not well-defined. Similarly, the exact alcohol amount that
can be recommended, while still remaining safe for health, changes according to
different subjects, their sex, age, and general as well as cardiovascular
status. Nevertheless, the knowledge in this field has been recently intensively
widened with studies providing associations between clinical and laboratory
data, consumed amount of alcohol, and novel echocardiographic parameters
describing heart morphology and function. The present review shortly recalls
the epidemiology and pathophysiology of alcohol overuse, as well as summarizes
the current data concerning the impact of alcohol consumption on the
cardiovascular system and heart function.
Epidemiology of alcohol consumption
According to the World Health Organization (WHO) report from
2018, approximately 2.4 billion people in the world drink alcohol, 43% of the
world’s adult population over 15 years old. (1) The highest alcohol consumption burden is reported in Europe
and reaches 60% of the population. The second region with highest alcohol consumption
is South and North America, in which it achieves 54%. In comparison, tobacco
use is estimated in 1.33 billion persons (22%) in a world-wide scale. (2)
The world’s alcohol consumption, presented as liters of pure
ethanol per capita per year (APC - alcohol per capita, including persons older
then >15 years), has been increasing since 2000. According to the cited WHO
report, in 2000 APC reached 5.7 liters and it rose to 6.4 liters in 2016.
Interestingly, simultaneously with the increase of the APC index, the number of
alcohol drinkers decreased by about 683 million. (1)
As far as the sex-difference in consumption is concerned, in
Europe 69% of men and 50% of women drink alcohol, whereas in South and North America
alcohol is consumed by 67% of men and 42% of women. (1)
The alcohol impact on health
Genetic factors may have an influence on the level of alcohol
consumption as well as on ethanol tolerance. Holmes et al. investigated a group
of 260 000 subjects to evaluate the impact of the rs 1229984 mutation in the
gene encoding 1B alcohol dehydrogenase (ADH1B) on drinking behavior. The rs
1229984 mutation seems to be related to specific drinking behavior
characterized by the tendency to consume lower alcohol volumes (17% lower
compared with the group without this mutation). Since the mutation carriers
were exposed to more severe symptoms connected with alcohol metabolism, by
limiting the amount of consumed ethanol, they showed thereafter lower risk of
alcohol related complications. In this study, the lower risk of ischemic heart
disease was observed in the group with the mentioned mutation (Odds Ratio [OR]
0.90 and 95% Confidence Interval [95%CI] 0.84- 0.96). (3)
Nevertheless, the beneficial effect of low to moderate
alcohol consumption on the human body was also postulated. The beneficial
effect was jointed with documented reduction of the onset of ischemic heart
disease, stroke, and diabetes. (1) In a meta-analysis performer by Di
Castelnuovo and co-authors, based on 209 418 patients, cardiovascular risk
reduction was observed (relative ratio [RR] 0.68, 95%CI 0.59 – 0.77) in the
group consuming 150 mL wine per day, compared with teetotalers. (4) The beneficial effect of wine and
beer was connected with the presence of specific ingredients belonging to the
polyphenols, as resveratrol, whose molecular effect is based on triggering
signal tracks like Nrf2 (nuclear factor erythroid 2-related factor 2),
NF-κB (nuclear factor kappa-light-chain-enhancer of activated B cells),
Sirt1 (NAD-dependent deacetylase sirtuin-1), AMPK (5'AMP-activated protein
kinase), as well as the reduction of oxidative stress and apoptosis, which was
observed in experimental studies. (5,6) In a meta-analysis including 1 902
605 participants, low alcohol doses (10 - 14 grams per day) induced 18%
reduction of diabetes onset in women. (7) The postulated mechanism of this
effect was the higher insulin sensitivity related to alcohol consumption. The
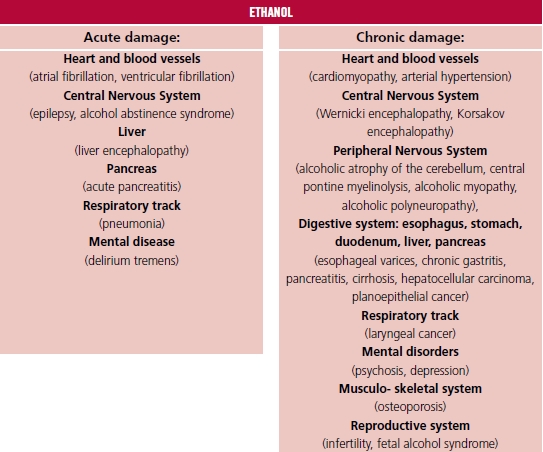
variety of harmful alcohol impacts on the human body is presented in Figure 1. (1,8,9)
Fig. 1. Target organs of
alcohol toxic impact.
ETHANOL
Alcohol metabolism
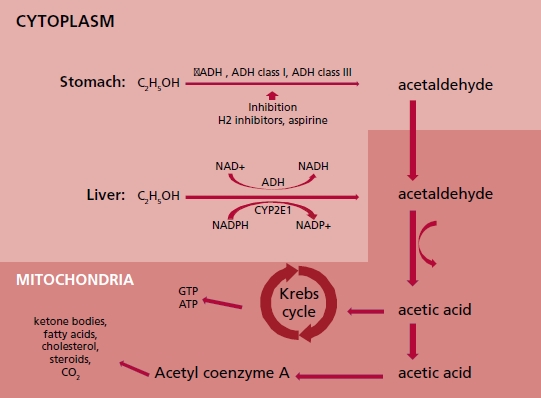
In the main pathway of alcohol metabolism two enzymes are
engaged: alcohol dehydrogenase (ADH) and P450 2E1 cytochrome (CYP450). The
intermediate product of this reaction is acetaldehyde, responsible for the toxic
influence on the human body. Further, acetaldehyde is metabolized by
acetaldehyde dehydrogenase and acetaldehyde oxidase to acetic acid, which
enters the Krebs cycle. A certain amount of the acetaldehyde leaves the liver
and is directed to peripheral tissues where it is metabolized to acetyl
coenzyme A (acetyl – CoA) as well as to cholesterol, steroids, fatty acids,
ketones and CO2 (Figure
2). Alcohol
metabolism starts in the gastric mucosa whose cells contain ADH. After absorption
in the gastrointestinal system through the portal vein, alcohol goes to the
hepatocytes where the main part of the metabolism is performed, since the
hepatocytes’ cytoplasm, mitochondria and microsomes contain the highest amounts
of ADH and CYP450. (10) A variety of factors can modify the
rate of alcohol metabolism. The afternoon body temperature increase, connected
with the daily cycle, accelerates alcohol metabolism. Physical exercise has
insignificant influence on alcohol metabolism acceleration, which is connected
rather with body temperature elevation. Young age is a factor reducing the pace
of alcohol metabolism due to lower expression of ADH and CYP450. The ADH Beta3
isoform characterizes higher efficiency of alcohol metabolism compared with
other isoforms. Cederbaum and co-authors observed the higher prevalence of ADH
Beta3 isoform among Afro-Americans (15%), and less frequent among Japanese,
South Americans (except indigenous peoples) and Europeans. The ADH Beta3 isoform
prevalence in this groups determines the rate of alcohol metabolism and the
risk of alcohol related complications. (10) According to lower content of body
water compared with men, women are more sensitive to alcohol toxic effects.
Other factors having impact on alcohol metabolism are nutrition level, and
exposition on other drugs like nicotine, cannabinoids, and cocaine. (11,12)
Fig. 2. Diagram
illustrating alcohol metabolism in stomach and liver cells. σADH: alcohol
dehydrogenase sigma isoform. ADH class I: alcohol dehydrogenase class one.ADH
class III: alcohol dehydrogenase class three. ATP: adenosine triphosphate.
CYP2E1: Cytochrome P450 2E1. ALDH2: mitochondrial aldehyde dehydrogenase 2/
acetaldehyde oxidase. C2H5OH: ethanol. CO2: carbon
dioxide. GTP: Guanosine triphosphate. H2: histamine receptor type 2. NAD+:
oxidized nicotinamide adenine dinucleotide. NADP+: oxidized nicotinamide
adenine dinucleotide phosphate. NADH: reduced nicotinamide adenine dinucleotide.
NADPH: reduced nicotinamide adenine dinucleotide phosphate
Alcohol consumption influence on the cardiovascular system
First studies
In 1861 Friedrich L. Goltz documented the relationship
between heart hypertrophy and persistent alcohol overuse. Further, in 1873
Walter H. Walshe as the author of the medical textbook entitled “The Physical
Diagnosis of Diseases of the Lungs” implemented the term “cirrhosis of the
heart” to describe phenomena of fibrosis observed simultaneously in both the
heart and the liver in patients with alcohol overuse history. (13,14) In 1884, in Munich, Otto von
Bollinger described “beer heart” cases (“Bierherz”) and linked this observation
to a history of beer overuse, reaching up to 432 liters of beer per year. The
“beer heart” in histopathology studies is characterized by hypotrophy,
fibrosis, and steatosis. (15,16) At the end of the 19th century
Graham Steell proved the relationship between heart failure and chronic
high-dose alcohol consumption. (17) In 1969, in Quebec, a series of patients
with cardiomyopathy related to beer drinking were observed. All mentioned cases
were reported in a seven-month period. Interestingly, all patients consumed
beer derived from the same origin, and ingredient analysis proved excess cobalt
concentration (in this time, cobalt was applied as foam stabilizer in beer). (18) The authors postulated alcohol and
cobalt as the etiology of cardiomyopathy in the above-mentioned cases.
Cardiotoxic alcohol dose
One of the first reports mentioning the relationship between
alcohol dose and its cardiotoxic impact was published by Tadashi Koide and
co-authors in 1974. The authors investigated the association between left
ventricular enlargement in chest radiograms and the dose of consumed alcohol.
The study showed that a pure ethanol dose ≥125 mL per day was related to
the increase of the cardio-thoracic index (CTI) above 0.5. Higher CTI rates
were observed in 33% of patients drinking ≥125 mL per day and in 4% of
patients drinking from 75 to 125 mL, as well as in 2.9 % of patients drinking <75
mL. The authors did not mention time duration of consumption in this group of
patients. (19) Other authors suggest that J or U –
shaped curves can describe the relationship between the alcohol dose and the
harmful cardiac effect. (20) For example, Ronksley et al., in a
meta-analysis including 84 studies, revealed that drinking 15 - 30 grams of
pure alcohol per day resulted in the decrease of cardiovascular mortality rate,
with hazard ratio (HR) 0.66 (95%CI 0.59 – 0.75), and in the group drinking 2.5-
14.9 grams per day, with HR 0.80 (95%CI 0.74 – 0.87). The decline in all-cause
mortality was observed in the drinking group compared with teetotalers, with HR
0.87 (95%CI 0.83 - 0.92). In this study the negative alcohol impact (ascending
J curve arm) was observed in groups drinking 30 - 60 grams per day (HR 1.15
[95% CI 0.98 – 1.35]). (21)
In the studies considering chronic alcohol consumption and
its relationship with cardiac toxicity, the alcohol dose is usually expressed
as the amount of alcohol and the time duration of alcoholism. In the
literature, the authors postulate a dose of 80 grams of pure ethanol drank every
day during 5 years as a toxic dose. (22-25) Other proposed cut-offs are 40 grams
per day, or on average of 14 grams per day during a minimum of 10 years. (20,26)
Alcohol-induced hypertension
The relationship between alcohol consumption and blood
pressure elevation can be temporary, and stop after alcohol drinking cessation.
(27) The hypertensive alcohol effect
seems to be independent of body mass index (BMI), tobacco use, physical
activity and coexisting arterial hypertension history. (20) This effect is caused by a few
mechanisms: the increased activity of the renin-angiotensin-aldosterone axis
and sympathetic nervous system, increased cortisol secretion, changed insulin
sensitivity, endothelial dysfunction and reduced nitric oxide production. (28) The modulation of the central
nervous system activity after alcohol exposure is an important reason for blood
pressure elevation. The alcohol vulnerable nervous tracks are the rostral
ventrolateral medulla and intermediolateral nucleus, as well as the
baroreceptor reflex, which passes through the nucleus solitarius, and causes a
hypertensive reaction. (20) According to the European Cardiac
Society guidelines for hypertension management, an alcohol dose above 14 alcohol
units (AU) per week (one AU contains 10 grams of ethanol) for men and 8 AU per
week for women is not recommended. (29) The rise of the consumed dose has a
linear positive correlation with the risk of arterial hypertension development.
(28) This relationship is sex
independent, but in women can be observed from a dose of 2 AU per week. (30) In
a meta-analysis performed by Roerecke and co-authors, mean reductions of 5 mmHg
in systolic blood pressure and 4 mmHg in diastolic blood pressure were observed
in patients who reduced the consumed dose from 6 AU per day to 0 AU. (31)
Dilated cardiomyopathy
The prevalence of dilated cardiomyopathy in the study
performed by Fernández-Solá and co-authors was higher among patients consuming
alcohol compared with the general population and reached 0.43% in males (mean pure
alcohol amount consumed during lifetime 30 ± 7 kg/kg body mass during an average
time of 29 ± 6 years), and 0.25% among women (mean pure alcohol amount consumed
during lifetime 17 ± 7 kg/kg body mass during an average time of 23 ± 7 years),
whereas in the general population dilated cardiomyopathy prevalence reaches
1:2500 individuals (0,4‰). (32-34) In the western countries, alcohol
overuse is the leading cause of non-ischemic cardiomyopathy. In the literature,
the estimated rate of alcohol overuse among patients diagnosed with dilated
cardiomyopathy ranges from 3.8% to 47%. (23,35) Factors as malnutrition, kwashiorkor, vitamins, electrolytes
and microelement deficiencies (sodium, potassium, calcium, magnesium,
phosphorus and selenium), as well as the exposition to other psychostimulants
like cocaine, amphetamine and nicotine, increase the risk of alcoholic
cardiomyopathy. (33,36)
There are no typical histopathological signs of alcoholic
cardiomyopathy. Similar to other dilated cardiomyopathy etiologies, myofibril
atrophy, mitochondriopathy (the abnormal differentiation of the mitochondria in
size and forms due to the chronic exposition to toxins including ethanol, and
the presence of megamitochondria), cardiomyocyte necrosis and fibrosis are
present. (11,14) Myocardial muscle exposition to
alcohol in the extracellular matrix leads to the activation of fibroblasts and
the excessive production of type I collagen. (37) Additionally, in fibroblasts exposed to alcohol, the
pro-inflammatory signal tracks are activated (the MAPK protein kinase groups,
the transcription factor STAT3 and the nuclear factor NF- κB) causing the
pro-inflammatory cytokine release from the fibroblast (IL-6, TNF-α,
IL-1β, IL-33), and cardiomyocyte dysfunction. (37,38) This process leads also to lower expression of genes
encoding contractile proteins, like Acta1, Actc1 (encoding actin) and Myh7
(encoding the myosin heavy chain alpha and myosin beta). Moreover, alcohol
exerts also a direct influence on cardiomyocytes (mediated by TNF-α,
without fibroblast as a mediator of the reaction), intensifying the processes
leading to a decrease of muscle fiber contractility. (37)
Acetaldehyde is an alcohol metabolite. Like ethanol,
acetaldehyde has a negative effect on the heart muscle, but the influence of
its cardiotoxic properties is even stronger than that of alcohol. (33) Both molecules have direct impact on
protein synthesis in the myocytes and lipid peroxidation, reduce the contractility
of myofibrils, and increase oxidative stress. (33,39) In magnetic resonance imaging, Liu S et al. observed shorter
native T1 values and greater extracellular volume in the myocardium (similarly
to images observed in heart amyloidosis and sarcoidosis) of a group of patients
consuming >28 grams of ethanol per day, during a minimum of 10 years,
compared with a control group (including persons drinking <100 grams per
month). Similar changes in magnetic resonance imaging are observed when the
content of fat molecules in the cell is increased. (26) Matyas and al. observed heart steatosis in the mice exposed
to ethanol compared with the control group receiving a calorie-adjusted diet
but without ethanol exposure. (40)
In images recorded by positron emission tomography with
carbon 11-labeled acetate, the decreased metabolic activity of the myocardium
has been observed in chronic alcohol overusers (26 years on average) consuming
high alcohol doses (167 grams of pure ethanol per day) compared with patients
drinking for a similar period a lower amount of alcohol (50 grams per day),
which suggests a decrease in mitochondrial function after chronic alcohol
exposure. (26)
The threshold of myocardial vulnerability to alcohol varies
from person to person and has not been clearly defined. The literature suggests
that the sensitivity can be also determined by genes. It is estimated that the
genetic background of susceptibility is present in 20% to 37% of dilated
cardiomyopathy cases. Currently, the possible mutations related to alcohol
cardiomyopathy occurrence are identified in over 50 genes, (41) the most common encoding proteins
being titin, posterophyllin - 2, myosin - binding protein C, desmoplakin,
ryanodine - 2 receptor, desmokolin - 2, desmoglein- 2 and SCN5A. (42) Ware and co-authors assessed the
effect of the presence of genes related to the occurrence of dilated
cardiomyopathy on cardiac function in patients consuming alcohol. The group
consisted of 715 patients aged 55 ± 14 years and included 70% men. In the
subgroup with the titin mutation (titin is the largest human protein, crucial
for the molecular basis of diastolic function), drinking excessive amounts of
alcohol (>21 AU/week in men or >14 AU/week in women) resulted in an 8.7%
reduction of left ventricular ejection fraction, LVEF (95%CI 2.3% - 15.1%; p
=0.007), compared with patients without this mutation. (43) The titin-encoding gene variants
associated with the development of dilated cardiomyopathy were more prevalent
in the alcoholic cardiomyopathy group compared with the control group. In the
group of patients with alcoholic cardiomyopathy, a relationship between the
amount of alcohol consumption and a decrease in LVEF was observed. This means
that in sensitive individuals, alcohol can also be a trigger in the dilated
cardiomyopathy development process. In addition, the finding of a greater
prevalence of titin gene mutations in the group with alcoholic cardiomyopathy
indicates the need for the active search of cardiomyopathy signs and symptoms
among the relatives of patients with this diagnosis. (44)
The toxic effect of alcohol metabolites on the heart results
in left and right ventricular muscle remodeling, atrium enlargement, and
secondary mitral and tricuspid valve regurgitation. (11) Based on the literature, it is
postulated that the diastolic dysfunction occurs first, followed by left ventricular
wall hypertrophy comparable to that observed in arterial hypertension. Further,
left ventricular systolic diameter (LVSD) and the left ventricular diastolic
diameter (LVDD) increase is observed, which parallels LVEF reduction. (11,25,33,45,46)
Current available data suggest that chronic alcohol exposure
also damages the right ventricle. (47) In a study of the cardiac effects of
a single exposure to alcohol, Cameli and co-authors observed a group of 64
volunteers (mean age 25 ± 4 years, 29 women) before and 60 minutes after 0.5
gram per kg alcohol consumption, and compared it with a control group. TAPSE
(tricuspid annulus plane excursion) was 22.1 ± 3.3 mm in the studied group vs.
24.0 ± 3.1 mm in controls, p = 0.003; and pulmonary artery systolic pressure
(PASP) amounted to 23.7 ± 3.2 mmHg in the exposed group vs. 20.2 ± 4.9 mmHg in
controls, p = 0.0002. (48)
This influence of alcohol on right ventricular hemodynamic
function can be related to increased pulmonary artery resistance due to
vasoconstriction caused by leukotrienes released from leukocytes after alcohol
exposure. (49)
Guzzo-Merello and co-authors postulated that the lack of
β-blocker treatment, the occurrence of atrial fibrillation and the
widening of the QRS complex are independent predictors of worse prognosis in
alcoholic cardiomyopathy, with an increased incidence of cardiac death and
heart transplantation. In patients who reduced their alcohol intake below 80
grams per day, the 59 - month risk of cardiac death or heart transplantation
was reduced to a level comparable with that of the general population. (23) The lack of abstinence in patients
with alcohol-related dilated cardiomyopathy resulted in very high mortality,
reaching 50% within 4 years, which poses alcoholic cardiomyopathy near to
patients with cancer diagnosis. (50) The pleiotropic effects of alcohol
on the heart are schematically shown in Figure 3.
Since the key issue related to early diagnosis of alcohol
related cardiomyopathy may be the usage of adequate diagnostic methods, it is
worth mentioning the still underused potential of the adapted protocols of
stress echocardiography in the early detection of diastolic dysfunction as
well as pulmonary hypertension. (51) Vriz and co-authors performed
exercise Doppler echocardiography in a group of 155 hypertensive patients and
in 145 healthy subjects, and documented that TAPSE during exercise was lower in
the hypertensive group, whereas PASP as well as its value indexed to the cardiac
output achieved during exercise was higher. (52)
In a recently published analysis of our group, the median and
interquartile range (IQR) of left ventricular mass index was 119 (91—155) g/m²
in patients drinking alcohol (median 30 (12–51) AU per week) vs. 93 (75–110)
g/m² in a control group (drinking up to 2 AU per week), p = 0.008; and the
relative wall thickness in the alcohol group was 0.5 (0.4–0.6) vs. 0.4
(0.4–0.4) in controls, p = 0.001. In the same study, global longitudinal strain
(GLS) and layer strains showed lower absolute values in alcohol overusers as
compared to controls. All abnormal strain parameters were associated with a
more frequent composite endpoint occurrence (higher clinical risk of death or
cardiovascular hospitalization). The best absolute cut-off values for outcome
prediction were: GLS <18%, layer endocardial strain <19%, and layer
epicardial strain <15%. (53)
Alcohol related arrhythmias
Atrial fibrillation (AF) is the most common arrhythmia
observed among alcohol abusers. Larsson and al. proved that during chronic
alcohol exposure, an increase in consumption of one AU per day increases the
risk of AF by 8%. (54) Most often, arrhythmias are observed
as a consequence of a single high alcohol dose intake in a short time and
numerous authors have mentioned this as “holiday heart syndrome”. This syndrome
was first reported in 1978 by Ettinger et al. (55,56) Chronic alcohol consumption is positively associated with
increasing risk of AF in the long-term follow-up. In the study performed by
Larson and al. (12 years of follow-up), AF RR (95%CI) for 7-14 AU/ week was
1.12 (1.02 – 1.23), and for >21 AU/week, 1.43 (1.25 – 1.65) compared with
controls. (54) Arrhythmias are more often observed
in patients diagnosed with cardiomyopathy than in those without structural
abnormalities. Moreover, episodes of abstinence may be triggers of arrhythmia
in chronically alcohol overusing patients, due to the increased adrenergic
activity observed at the beginning of abstinence and the coexisting deficiencies
of macro and micronutrients (sodium, potassium, magnesium, calcium, phosphorus,
selenium) and vitamins (thiamine) associated with previous alcohol abuse. (33,57,58)
Cirrhotic Cardiomyopathy
The disorders observed in the hepato-cardiac syndrome may
also contribute to the pathogenesis of alcoholic cardiomyopathy. (59) Systolic function is decreased both
at rest and during stress, the impairment associated with a decrease in the
activity of β1- receptors through a reduction in G protein expression in
the cytosol, related to liver impairment. (59-61) In parallel, diastolic dysfunction is observed, associated
with left ventricular hypertrophy, fibrosis, and endothelial dysfunction. (59,62) These factors lead to secondary
higher heart rate and increased cardiac output, which result in a hyperkinetic
cardiomyopathy. Diagnostic criteria of Cirrhotic Cardiomyopathy have been
established in 2005 during the World Congress of Gastroenterology and are
displayed in Table 1. (59,63)
Summary
The recent data show that chronic overuse of alcohol may lead
to cardiovascular dysfunction, starting from traditionally judged as low
ethanol doses, since the burden of arrhythmias, including atrial fibrillation,
increases even in moderate alcohol consumers.
The other common mechanisms of the disadvantageous impact of
ethanol are related to the development of hypertension and its direct
aftermath, hypertrophy, fibrosis, and diastolic dysfunction.
Since the chance of the reversibility of cardiac remodeling
depends on the early diagnosis of cardiac dysfunction, the wider application of
novel and sensitive methods of myocardial function assessment, including
longitudinal strain of the left and right ventricles, as well as the adapted
protocols for stress echocardiography, should be recommended.
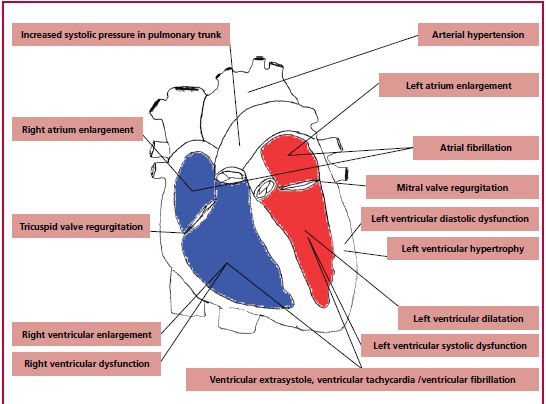
Fig. 1. D-dimer boxplot between
patients undergoing computed tomography pulmonary angiography diagnosed with
(1) or without (0) pulmonary embolism. a) pre- COVID-19 phase, b) COVID-19
phase
Table 1. Diagnostic
criteria of Cirrhotic Cardiomyopathy – World Congress of Gastroenterology,
Montreal 2005.
|
Definition:
A cardiac dysfunction in patients with cirrhosis characterized by impaired
contractile responsiveness to stress and/or altered diastolic relaxation with
electrophysiological abnormalities in the absence of other known cardiac
disease |
|
|
Diastolic dysfunction (one with above) |
-E/A ratio <1.0 (age - corrected) -Prolonged deceleration time (>200 msec) -Prolonged isovolumetric relaxation time (>80
msec) (4) |
|
Supportive
criterias |
-
inadequate chronotropic reaction to exercise,
assessed during tilt-test as heart rate increases over 22% in patients in B
Child’s – Pugh’s class and over 17% in patients in C Child’s – Pugh’s class
(64) |
Conflicts of interest
None declared.
(See authors' conflict of interests forms on the web/Additional
material.)
1.
World Health Organization. Global status report on alcohol and health. Geneva.
2019.
2.
World Health Organization. Global report on trends in prevalence of tobacco use
2000-2025, Geneva 2019.
3. Holme MV, Dale CE, Zuccolo L, Silverwood RW, Guo Y,
Ye Z, et al, Association between alcohol and cardiovascular disease: Mendelian
randomisation analysis based on individual participant data. Br Med J
2014;349:41-64.
4. DiCastelnuovo A, Rotondo S, Iacoviello L, Donati B,
DeGaetano G. Meta-analysis of wine and beer consumption in relation to vascular
risk. Circulation 2002;24:2836-44. https://doi.org/10.1161/01.CIR.0000018653.19696.01
5. Chiva-Blanch G, Arranz S, Lamuela-Raventos RM,
Estruch R. Effects of wine, alcohol and polyphenols on cardiovascular disease
risk factors: evidences from human studies. Alcohol
Alcohol 2013;48:270- 7. https://doi.org/10.1093/alcalc/agt007
6.
Zhuang Y, Wu H, Wang X, He J, He S, Yin Y. Resveratrol Attenuates Oxidative
Stress-Induced Intestinal Barrier Injury through PI3K/Akt-Mediated Nrf2
Signaling Pathway. Oxid Med Cell Longev 2019;2:7591840 https://doi.org/10.1155/2019/7591840
7. Knott C, Bell S, Britton A. Alcohol Consumption and
the Risk of Type 2 Diabetes: ASystematic Review and Dose-Response Meta-analysis
of More Than 1,9 Milion Individuals From 38 Observational Studies. Diabetes Care 2015;28:1804-12. https://doi.org/10.2337/dc15-0710
8. Kozela M, Doryńska A, Bobak M, Pająk A.
Alcohol use disorder increases the risk of nonfatal and fatal cardiovascular
disease: an 11-year follow-up of a Polish population-based cohort. The HAPIEE study. Pol Arch Intern Med 2020;130:960-6.
https://doi.org/10.20452/pamw.15616
9. Akhouri S, Kuhn J, Newton EJ. Wernicke-Korsakoff
Syndrome. s.l.: NCBI Bookshelf. A service of the National Library of Medicine,
National Institutes of Health. 2020;20:10.
10.
Cederbaum AI. Alcohol Metabolism. Clin Liver Disq 2012;16:667–
85. https://doi.org/10.1016/j.cld.2012.08.002
11.
Mirijello A, Tarli C, Vassallo GA, Sestito L, Antonelli M, d'Angelo C, et al. Alcoholic cardiomyopathy: What is
known and what is not known. Eur
J Intern Med 2017;17:1-5. https://doi.org/10.1016/j.ejim.2017.06.014
12. Mezey E. Influence of sex hormones on alcohol
metabolism. Alcohol Clin Exp Res 2000;4:421. https://doi.org/10.1111/j.1530-0277.2000.tb02005.x
13. Walshe WH. Diseases of the heart and great
vessels. 4th edit. London : Smith, Elder and Co, 1873.
14. Maisch, B. Alcoholic cardiomyopathy The result of
dosage and individual predisposition. Herz
2016;31:484-93. https://doi.org/10.1007/s00059-016-4469-6
15.
von Bollinger, Otto. Über die Häufigkeit und Ursachen der idiopathischen
Herzhypertrophie in München. DtschMedWochenschr 1884;180:80. https://doi.org/10.1055/s-0029-1209236
16. AL, Klatsky. Alcohol and cardiovascular diseases:
a historical overview. Ann
N Y Acad Sci 2002;957:7-15. https://doi.org/10.1111/j.1749-6632.2002.tb02901.x
17.
G. Steell. Heart failure as a result of chronic alcoholism. Med Chron 1893, 18,
pp. 1-22.
18. Morin Y, Têtu A, Mercier G. 'Quebec beer-drinkers'
cardiomyopathy: Clinical and hemodynamic aspects. Ann
N Y Acad Sci 1969;156:566–76. https://doi.org/10.1111/j.1749-6632.1969.
tb16751
19. Koide T, Ozeki K. The incidence of myocardial
abnormalities in man related to the level of ethanol consumption. A proposal of
a diagnostic criterion of alcoholic cardiomyopathy. Jpn Heart J 1974;15:337-48.
https://doi.org/10.1536/ihj.15.337
20.
El-Mas MM, Abdel-Rahman AA. Role of Alcohol Oxidative Metabolism in Its
Cardiovascular and Autonomic Effects. Adv Exp Med Biol 2019;1193:1-33.
https://doi.org/10.1007/978-981-13-6260-6_1
21. Ronksley PE, Brien SE, Turner BJ, Mukamal KJ,
Ghali WA. Association of alcohol consumption with selected cardiovascular disease
outcomes: a systematic review and meta-analysis. Br
Med J 2011;10:342. https://doi.org/10.1136/bmj.d671
22.
Ware JS, Amor-Salamanca A, Tayal U, Govind R, Serrano I, Salazar-Mendiguchía
J, García-Pinilla JM, Garcia-Pavia P et al. Genetic Etiology for Alcohol-Induced
Cardiac Toxicity. J Am Coll Cardiol 2018;71:2293-302. https://doi.org/10.1016/j.jacc.2018.03.462 https://doi.org/10.1016/j.jchf.2014.07.014
23.
Guzzo-Merello G, Segovia J, Dominguez F, Cobo-Marcos M, Gomez-Bueno M, Avellana
P, Millan I, et al. Natural history and prognostic factors in alcoholic
cardiomyopathy. JACC Heart Fail 2015;3:78-86. https://doi.org/10.1016/j.jchf.2014.07.014
24.
A Gavazzi, R De Maria, M Parolini, M Porcu. Alcohol abuse and dilated
cardiomyopathy in men. Am
J Cardiol 2000;9:1114-8. https://doi.org/10.1016/S0002-9149(00)00706-2
25.
Lazarević AM, Nakatani S, Nesković AN, Marinković J, Yasumura
Y, Stojicić D, Miyatake K, Bojić M, Popović AD. Early changes in left ventricular
function in chronic asymptomatic alcoholics: relation to the duration of heavy
drinking. J Am Coll Cardiol. 2000;35:1599- 606. https://doi.org/10.1016/S0735-1097(00)00565-9
26.
Liu S, Lin X, Shi X, Fang L, Huo L, Shang F et al. Myocardial tissue and
metabolism characterization in men with alcohol consumption by cardiovascular
magnetic resonance and 11C-acetate PET/CT. J Cardiovasc Magn Reson 2020;22:1. https://doi.org/10.1186/s12968-020-00614-2
27. Potter JF, Beevers DG. Pressor effect of alcohol
in hypertension. Lancet 1984;8369:119-22. https://doi.org/10.1016/S0140-6736(84)90060-6
28.
Ikehara S, Iso H. Alcohol consumption and risks of hypertension and
cardiovascular disease in Japanese men and women. Hypertens Res 2020;13:11. https://doi.org/10.1038/s41440-020-0417-1
29. Williams B, Mancia G, Spiering W, Rosei EA, Azizi
M, Desormais I, et al. 2018 ESC/ESH Guidelines for the management of arterial
hypertension. Kardiol Pol 2019;77:71-159. https://doi.org/10.5603/KP.2019.0018
30.
Roerecke M, Tobe SW, Kaczorowski J, Bacon SL, Vafaei A, Hasan OSM, et al.
Sex‐Specific Associations Between Alcohol Consumption and Incidence of
Hypertension: A Systematic Review and Meta‐ Analysis of Cohort Studies.
JAHA 2018;7:13. https://doi.org/10.1161/JAHA.117.008202
31. Roerecke M, Kaczorowski J, Tobe SW, Gmel G, Hasan
OSM, Rehm J. The effect of a reduction in alcohol consumption on blood
pressure: a systematic review and meta-analysis. Lancet
Public Health 2017;2:108-20. https://doi.org/10.1016/S2468-2667(17)30003-8
32.
Fernández-Solà J, Estruch R, Nicolásv JM, Paré JC, Sacanella E, Antúnez E, et
al. Comparison
of alcoholic cardiomyopathy in women versus men. Am J Cardiol 1997;80:4815. https://doi.org/10.1016/S0002-9149(97)00399-8
33.
Fernández-Solà, Joaquim. The Effects of Ethanol on the Heart: Alcoholic
Cardiomiopathy. Nutriens 2020;22:72. https://doi.org/10.3390/nu12020572
34. Maron BJ, Towbin JA, Thiene G, Antzelevitch C,
Corrado D, Arnett D, et al. Contemporary Definitions and Classification of the
Cardiomyopathies. Circulation 2006;113:1807-16. https://doi.org/10.1161/CIRCULATIONAHA.106.174287
35.
Guzzo-Merello G, Cobo-Marcos M, Gallego-Delgado M, Garcia- Pavia P. Alcoholic
cardiomyopathy. World J Cardiol 2014;8:771-81. https://doi.org/10.4330/wjc.v6.i8.771
36. Chan LN, Anderson GD. Pharmacokinetic and
pharmacodynamic drug interactions with ethanol (alcohol). Clin Pharmacokinet 2014;12:1115-36. https://doi.org/10.1007/s40262-014-0190-x
37. Mouton AJ, El Hajj EC, Ninh VK, Siggins RW,
Gardner JD. Inflammatory cardiac fibroblast phenotype underlies chronic
alcohol-induced cardiac atrophy and dysfunction. Life
Sci. 2020;15:245. https://doi.org/10.1016/j.lfs.2020.117330
38.
Chih-Chung L, Chih-Shuo P, Chen-Yu W, Shiau-Wen L, Li-Der H, Chuen-Mao Y. Tumor
necrosis factor-alpha induces VCAM-1-mediated inflammation via c-Src-dependent
transactivation of EGF receptors in human cardiac fibroblasts. J Biomed Sci
2015;22:1. https://doi.org/10.1186/s12929-015-0165-8
39. Rohde LE, Beck-da-Silva L. Alcohol and the heart:
the good, the bad and the worse in heart failure. Heart
2018;104:1641-12. https://doi.org/10.1136/heartjnl-2017-312924
40.
Matyas C, Varga ZV, Mukhopadhyay P, Paloczi J, Lajtos T, Erdelyi K, et al. Chronic plus binge ethanol feeding
induces myocardial oxidative stress, mitochondrial and cardiovascular
dysfunction, and steatosis. Am
J Physiol Heart Circ Physiol 2016;310:1658-70. https://doi.org/10.1152/ajpheart.00214.2016
41. Orphanou N, Papatheodorou E., Anastasakis A.
Dilated cardiomyopathy in the era of precision medicine: latest concepts and
developments. Heart Fail Rev 2021;12:1-19. https://doi.org/10.1007/s10741-021-10139-0
42. Haas J, Frese KS, Peil B, Kloos W, Keller A,
Nietsch R. Atlas of the clinical genetics of human dilated cardiomyopathy. Eur Heart J 2015;36:1123-35. https://doi.org/10.1093/eurheartj/ehu301
43. Ware JS, Cook SA. Role of titin in cardiomyopathy:
from DNA variants to patient stratification. Nat
Rev Cardiol. 2017;15:241-52. https://doi.org/10.1038/nrcardio.2017.190
44. Ware JS, Amor-Salamanca A, Tayal U, Govind R,
Garcia-Pavia P. Genetic Etiology for Alcohol-Induced Cardiac Toxicity. J Am Coll Cardiol2018;71:2293-302. https://doi.org/10.1016/j.jacc.2018.03.462
45. Park SK, Moon K, Ryoo JH, Oh CM, Choi JM, Kang JG,
et al. The association between alcohol consumption and left ventricular diastolic
function and geometry change in general Korean population. Eur Heart J Cardiovasc Imag 2018;3:271-8.
https://doi.org/10.1093/ehjci/jex091
46.
Kupari P, Koskinen A, Suokas M, Ventilä M. Left ventricular filling impairment
in asymptomatic chronic alcoholics. Am J Cardiol 1990;66:20. https://doi.org/10.1016/0002-9149(90)90537-B
47.
Meng S, Guo L. Li G. Early changes in right ventricular longitudinal function
in chronic asymptomatic alcoholics revealed by two-dimensional speckle tracking
echocardiography. Cardiovasc Ultrasound 2015;14:10. https://doi.org/10.1186/s12947-016-0058-3
48. Cameli M, Ballo P, Garzia A, Lisi M, Bocelli A,
Mondillo S. Acute effects of low doses of ethanol on left and right ventricular
function in young healthy subjects. Alcohol
Clin Exp Res 2011;35:1860-5. https://doi.org/10.1111/j.1530-0277.2011.01530.x
49.
Lu CY, Wang DX, Yu SB. Effects of acute ingestion of ethanol on hemodynamics
and hypoxic pulmonary vasoconstriction in dogs- -role of leukotrienes. J Tongji
Med Univ 1992; 12:253-6. https://doi.org/10.1007/BF02887861
50. Skotzko Ch, Vrinceanu A, Krueger L, Freudenberger R.
Alcohol use and congestive heart failure: incidence, importance, and approaches
to improved history taking. Heart
Failure Rev 2009;14:51- 5. https://doi.org/10.1007/s10741-007-9048-8
51.
Picano E, Ciampi Q, Cortigiani L, Arruda-Olson AM, Borguezan- Daros C, de
Castro E et al. The Stress Echo Study Group Of The Italian Society Of Echocardiography
And Cardiovascular Imaging Siecvi. Stress Echo 2030: The Novel ABCDE-(FGLPR)
Protocol to Define the Future of Imaging. J
Clin Med 2021;10:36-41. https://doi.org/10.3390/jcm10163641
52.
Vriz O, Palatini P, Rudski L, Frumento P, Kasprzak JD, Ferrara F, et al. Right Heart Pulmonary Circulation
Unit Response to Exercise in Patients with Controlled Systemic Arterial
Hypertension: Insights from the RIGHT Heart International NETwork (RIGHT-NET). J Clin Med 2022;11:451. https://doi.org/10.3390/jcm11020451
53.
Hamala P, Kasprzak JD, Bińkowska A, Zawitkowska-Witczak K, Broncel M,
Piekarska A, et al. The impact of chronic alcohol overuse on heart
function and prognosis: layer-specific longitudinal strain and mid-term outcome
analysis. Kardiol Pol 2021;7:781-8. https://doi.org/10.33963/KP.15986
54. Larsson SC, Nikola D, Wolk A. Alcohol Consumption
and Risk of Atrial Fibrillation: A Prospective Study and Dose-Response Meta-
Analysis. J Am Coll Cardiol 2014;64:281-9. https://doi.org/10.1016/j.jacc.2014.03.048
55. Ettinger PO, Wu CF, De La Cruz C Jr, Weisse AB,
Ahmed SS, Regan TJ. Arrhythmias and the "Holiday Heart":
alcohol-associated cardiac rhythm disorders. Am
Heart J 1978;95:555-62. https://doi.org/10.1016/0002-8703(78)90296-X
56. Tonelo D, Providência R, Gonçalves L. Holiday
heart syndrome revisited after 34 years. Arq
Bras Cardiol 2013;101:183-9. https://doi.org/10.5935/abc.20130153
57. Athen D, Beckmann H, Ackenheil M, Markianos M.
Biochemical Investigations into the Alcoholic Delirium: Alterations of Biogenic
Amines. Arch. Psychiat. Nervenkr.
1977;224:129-40. https://doi.org/10.1007/BF00346481
58. Fernández-Solà, J. Cardiovascular risks and
benefits of moderate and heavy alcohol consumption. Nat Rev Cardiol 2015;12:576-87. https://doi.org/10.1038/nrcardio.2015.91
59. Møller S, Henriksen JH. Cirrhotic cardiomyopathy. J Hepatol. 2010;53:179-90. https://doi.org/10.1016/j.jhep.2010.02.023
60. Wong F, Girgrah N, Graba J, Allidina Y, Liu P,
Blendis L. The cardiac response to exercise in cirrhosis. 2001;49:268-275. https://doi.org/10.1136/gut.49.2.268
61. Lee SS, Marty J, Mantz J, Samain E, Braillon A,
Lebrec D. Desensitization of myocardial beta-adrenergic receptors in cirrhotic
rats. Hepatology 1990;12:481-5. https://doi.org/10.1002/hep.1840120306
62. Ma Z, Lee SS. Cirrhotic cardiomyopathy: getting to
the heart of the matter. Hepatology
1996;24:451-9. https://doi.org/10.1002/hep.510240226
63. Møller S, Danielsen KV, Wiese S, Hove JD, Bendtsen
F. An update on cirrhotic cardiomyopathy. Expert
Rev Gastroenterol Hepatol 2019;13:497-505. https://doi.org/10.1080/17474124.2019.1587293
64. Møller S, Nørgaard A, Henriksen JH, Frandsen E,
Bendtsen F. Effects of tilting on central hemodynamics and homeostatic
mechanisms in cirrhosis. Hepatology
2004;40:811-9. https://doi.org/10.1002/hep.1840400410