SCIENTIFIC LETTERS
http://dx.doi.org/10.7775/rac.v91.i1.20606
Cardiac Autotransplantation
as Treatment Strategy for Malignant Heart Tumors
Primary malignant cardiac tumors, usually sarcomas,
represent an infrequent subgroup among cardiac masses. However, they constitute
processes with poor prognosis, and surgical treatment is the most favorable
therapeutic alternative in terms of survival.
The literature regarding left-sided cardiac sarcomas
reveals that patients are subjected to reinterventions
for local recurrence, generally related to incomplete resections, probably due
to a suboptimal anatomical exposure during surgery, which conditions inadequate
resections and technically difficult reconstructions.
Sometimes, to fulfill the objectives of a radical oncological
resection and facilitate the reconstruction of the resected cardiac structures,
it is necessary to explant the heart in order to resect
the tumor with adequate margins and reconstruct the cavities or involved
structures, finally reimplanting the heart in bench
surgery (cardiac autotransplantation).
This is the case is a female 73-year-old patient,
without relevant clinical history who was admitted to hospital due to
progressive dyspnea and anemia. In the diagnostic algorithm, the transthoracic
echocardiogram showed a dilated left atrium occupied by a 4.8 cm × 2.8 cm
immobile, heterogeneous mass, intimately associated with the mitral annulus,
which completely filled the left atrial appendage, and severe mitral valve
insufficiency with central jet.
A cardiac magnetic resonance performed to complete
the mass evaluation revealed the mentioned heterogeneous tumor in weighted T1
and T2 sequences, before and after contrast, as well as in perfusion and late
enhancement sequences. No contrast capture was evidenced in the tumor sector
protruding to the left atrium, which was interpreted as an added thrombotic
component (Figure 1). The same study showed absence of pericardial and
pulmonary vein involvement. Prophylactic anticoagulation was started, and a
positron-emission computed tomography (PET-CT) of the whole body was performed
for local evaluation and search of eventual metastasis.
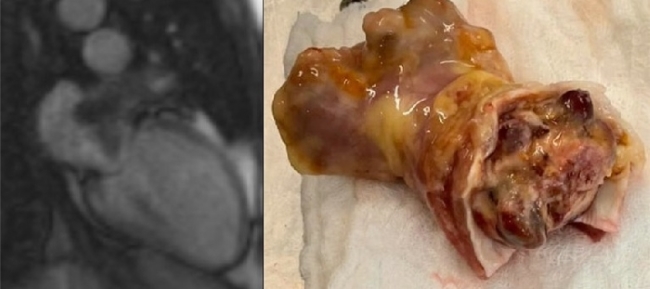
Fig. 1. A. Nuclear
magnetic resonance: left atrial sarcoma located in the left atrial appendage. B.
Surgical specimen with complete lesion resection. The resection margins are
evidenced on tissue not affected by the tumor.
The PET study showed a hypermetabolic
mass of 5.9 cm × 3.4 cm × 2.4 cm (SUV 8.5) in the already known location, focal
liver lesions compatible with hemosiderosis and
absence of secondaries.
A surgical treatment was decided due to the condition
and clinical characteristics of the patient, disease staging and prognosis
without resection. Owing to the location of the lesion to resect, in close
contact with the mitral annulus, the circumflex artery and the coronary sinus,
it was inferred that to perform an adequate oncological resection, the heart
should be explanted and reconstructed in bench surgery (ex situ) with
subsequent autotransplantation.
Surgery was performed with midline sternotomy
and cannulation of both venae cava and aorta
The tumor was explored entering the left atrium by the
interatrial sulcus as usually performed for a mitral
valve procedure. Absence of pulmonary vein involvement and tumor growth up to
the vicinity of the mitral annulus were verified.
Considering that the oncological resection would
involve resecting the mitral annulus and part of the mitral valve, and faced
with the difficulty to define, through the mentioned approach, the external
margin of the resection in relation to the interventricular
sulcus structures, it was decided to explant the heart and perform a bench
tumor resection.
The venae cava, aorta and pulmonary artery were
sectioned and the atriotomy was extended leaving a
hood that contained the pulmonary veins. The tumor was resected ex situ (bench
surgery), which implied resecting a section of the mitral annulus at the P1
level exposing the atrioventricular sulcus vessels
and the ventricular myocardium. The mitral annulus and the left atrium were reconstructed
with a bovine pericardial patch and the mitral valve was replaced with a #25
porcine biological prosthesis.
The organ was reimplanted
with autotransplantation technique (Figure 2).
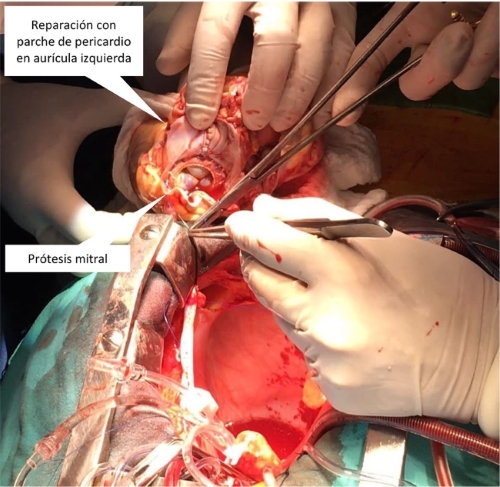
Fig. 2. Cardiac autotransplantation showing the left atrium repaired with
bovine pericardium and mitral prosthesis
Extracorporeal circulation time was 232 min and
cross-clamping time 175 min. The postoperative course was in accordance with
the magnitude of the procedure, requiring inotropic support for 72 h. Among
other events, the patient presented an episode of atrial flutter which was
controlled with amiodarone and isolated subfebrile records with negative cultures.
Anatomical pathology reported a grade III undifferentiated
pleomorphic sarcoma, which implies a maximum level of malignancy and undifferentiation.
The prevalence of primary cardiac tumors in autopsy
series is 0.02%. Among them, 25 % are malignant and 75 % of these are sarcomas.
(1) Median survival in published series ranges between 9
and 33 months. (2) Most are clinically silent until a very advanced
stage and are often considered nonresectable due to
the proximity to critical structures. However, surgical and imaging techniques
have improved allowing more aggressive interventions, which aim to achieve a
microscopically negative resection (R0), a situation in which there is clear
benefit of survival. (3)
Cardiac autotransplantation
is a procedure described many years ago for the resection of tumors with
difficult approach or complex intraoperative management. (4)
Along time, the technique was reproduced for the
management of this pathology in numerous patients, (5) and the initial results improved in terms of quality
of the oncological resection and survival. (6)
With adequate surgical training the technique is
reproducible and should be considered a valuable alternative in the therapeutic
arsenal to offer opportunities to patients with severe oncological disease and
poor prognosis without surgery.
Conflicts of interest
None declared.
(See authors' conflict of interests forms on the web/Additional material.)
Ethical considerations
Not applicable.
Ricardo G. Marenchino1, Edgar F.
Montalvo1, Juan C.
Climente1,
Diego E. Pinto1, Alejandra
Ferro2,
María E. González3
1 Department of Cardiovascular Surgery-
Hospital Privado de Comunidad.
2 Department of Cardiology - Hospital Privado
de Comunidad.
3 Intensive Care Unit-
Hospital Privado de Comunidad.
Address for reprints: Ricardo G. Marenchino.
Córdoba 4545 CP: 602CBM Mar del Plata
- Provincia de Buenos Aires - Argentina. Tel: + 54223- 4990000 - rmarenchino@gmail.com
1. Burazor I, Aviel-Ronen S, Imazio M, Markel
G, Grossman Y, Yosepovich A, et al. Primary
malignancies of the heart and pericardium. Clin Cardiol 2014;37:582-8. https://doi.org/10.1002/clc.22295
2. Chen TW, Loong HH, Srikanthan A. Primary cardiac sarcomas: a multinational
retrospective review. Cancer Med 2019;8:104-10.
https://doi.org/10.1002/cam4.1897
3. Putnam JB Jr,
Sweeney MS, Colon R. Primary cardiac sarcomas. Ann Thorac
Surg. 1991;51:906-10. https://doi.org/10.1016/0003-4975(91)91003-E
4. Cooley D, Reardon M, Fraizer
O, Angelini P. Human Cardiac Explantation
and Autotrasnplantation: Application in a Patient
with a Large Cardiac Pheocromocytoma. Tex Heart Inst J 1985;2:171-6.
5. Ranlawi B, Al-Jabbari O, Blau L, Davies M,
Bruckner B, Blackmon S et al. Autotransplantation
for the resection of complex left heart tumors. Ann Thorac
Surg 2014;98:863–8. https://doi.org/10.1016/j.athoracsur.2014.04.125
6. Hassan S, Witten J, Collier P, Tong M, Petterson G, Smedira N, et al.
Outcomes after resection of primary cardiac sarcoma. JTCVS Open 2021;8:384-90. https://doi.org/10.1016/j.xjon.2021.08.038
http://dx.doi.org/10.7775/rac.v91.i1.20603
Compressive Mass
in the Anterior Pericardium: Difficulty of the Differential Diagnosis
We report the case of a 79-year-old male patient,
smoker, with hypertension, type 2 diabetes, coronary artery
bypass surgery in 2009 due to chronic coronary syndrome, and prostatic
adenocarcinoma actively followed-up by the Urology Department. The
patient was referred to the Emergency Room due to dyspnea on moderate exertion
for three weeks and right pleural effusion seen in a chest X-ray. Upon arrival,
the patient was hemodynamically stable (blood
pressure 153/93 mmHg, heart rate 99 bpm), and oxygen
saturation at room air of 89%, with no tachypnea at rest. Physical examination
revealed jugular venous distention up to the middle third of the
sternocleidomastoid muscle, abolition of the vesicular murmur in the right
lung base, and bilateral pitting edema up to both knees.
ECG showed sinus rhythm (89 bpm)
with negative T-waves in V1-V4, already present in previous studies. Blood
screening showed normal renal function (urea 31 mg/dL,
creatinine 0.77 mg/dL,
glomerular filtration rate 86 ml/min/1.73 m2)
with all ions in range, C-reactive protein 25.98 mg/L, lactate dehydrogenase
950 U/L, creatinine kinase 55 U/L, NT-proBNP 950 pg. /mL, ultrasensitive troponin T 25 ng/L, hemoglobin 11.2 g/dL,
platelets 335000/mm3, white blood cells 11430/mm3,
and D-dimer 4860 ng/mL. To
complete the diagnosis, the unsynchronized computed tomography (CT) scan
revealed a hypodense lesion over the right cardiac
chambers (Figure 1). Neoplasia or hemopericardium due to cardiac rupture or bypass dehiscence
were suggested as first possible diagnosis. In view of these findings, evaluation
by the Cardiology Department was requested. Transthoracic echocardiography
(TTE) showed a heterogeneous, solid mass in the anterior pericardial sac, with
adhesions in the right chambers and compression of the right atrioventricular sulcus, which did not capture
echocardiographic contrast (Figure 2). The picture did not suggest a cardiac rupture, not
only because of the echocardiographic findings, but also because the patient
did not experience chest pain and was hemodynamically
stable, and ECG did not show abnormalities suggestive of acute ischemia, making
bypass dehiscence unlikely.
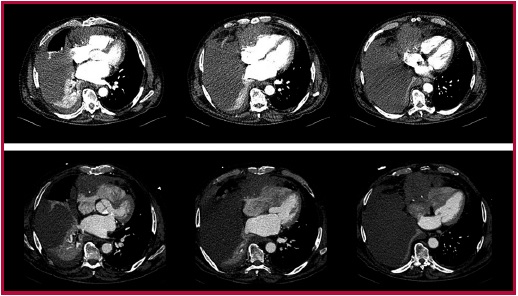
Fig.
1. CT scan. Top:
Unsynchronized CT scan, with a hypodense lesion over
the right cardiac chambers and severe right pleural effusion with associated
collapse. Bottom: Synchronized CT scan, with a right precardiac
mass of 8.5 x 10 cm, enhanced after contrast injection. The tumor exerts mass
effect and possibly infiltrates the right atrium, in contact with anterior
chest wall and possible infiltration of the right ventricle.
Fig.
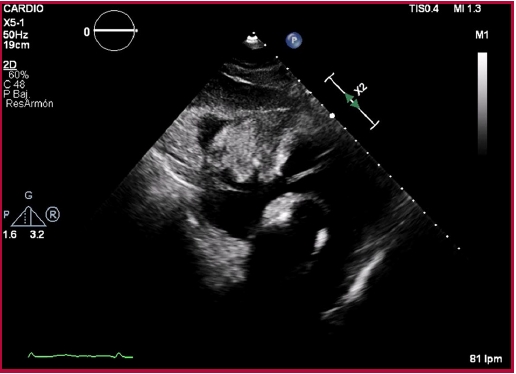
2. Transthoracic
echocardiography, subcostal access. Heterogeneous,
solid mass in the anterior pericardial sac, compressing the atrioventricular
groove and appearing to adhere to the ventricular wall.
Given the discrepancies, a synchronized CT scan was
performed for better characterization of the lesion, which showed an 8.5 x 10
cm right precardiac mass, apparently depending on the
pericardium, and showing enhancement after intravenous contrast (Figure 1). These findings raised the differential diagnosis of
metastasis or primary pericardial neoplasia.
The differential diagnosis of mediastinal
masses is primarily based on the location of the mass, its composition and the
age of the patient. (1) Different radiological techniques, including CT scan
and cardiac magnetic resonance (CMR), are of significant diagnostic value.
Considering that the lesion was located in the anterior wall of the heart, in
contact with the anterior chest wall, a broad differential diagnosis including
the different lesions at the level of the anterior mediastinum and the
tissue-dependent masses in the pericardium was proposed.
Unlike in our patient, thymomas
appear as an oval, homogeneous mass, with well-defined contours in CT scan.
Calcifications and cystic areas are usually present in thyroid goiters and teratomas, (1-2) and considering that CT findings showed no
calcifications in our patient, these two entities seemed unlikely. Lymphomas
account for 20% of mediastinal tumors in adults, and
Hodgkin's lymphomas are the most common subtype. (1-3) Within this subtype, mediastinal
large B-cell lymphoma constitutes an independent entity within the classification
of malignant lymphoid neoplasms, with a frequency estimated at 2-3% of
non-Hodgkin's lymphomas and between 6-10% of large B-cell lymphomas. This tumor
usually occurs as a rapidly expanding mediastinal
mass and may be associated with pleural or pericardial effusion. (4,
5)
The patient was admitted to the Cardiology Department
to complete assessment. An ultrasound-guided thoracentesis
of the pleural effusion was performed, and a serosanguineous
fluid consistent with an exudate (Light's criteria) was obtained, showing a hypercellular area in the cytological examination, with
characteristics indicative of a B lymphoproliferative
process. To complete the study, a core biopsy of the mediastinal
mass was performed, confirming the diagnosis of primary mediastinal
large B-cell lymphoma. Finally, the first cycle of chemotherapy was started
with rituximab, cyclophosphamide, non-pegylated
liposomal doxorubicin, vincristine, and prednisolone. He is still on treatment.
Conflicts of interest
None declared.
(See authors’ conflicts of interest
forms on the website/ Supplementary material).
Sources of funding: None.
Ethical considerations
Not applicable.
Uxue Idiazabal
Rodriguez1, Adrián
Costa Santos1,
Lara Ruiz Gómez2, Alain Laskibar Asua1, Iván Cano González1, Ana Ruiz Rodríguez1
1 Hospital Universitario
Basurto. Department of
Cardiology, Hospitalization Ward. Osakidetza. Bilbao, Spain.
2 Hospital Universitario
Basurto. Department of
Cardiology, Section of Cardiac Imaging. Osakidetza.
Bilbao, Spain
Address for reprints: Uxue Idiazabal
Rodriguez. E-mail: uxue_278@hotmail.com
1. Prosch H, Röhrich S, Tekin ZN, Ebner L. The role of radiological imaging
for masses in the prevascular mediastinum in clinical
practice. J Thorac Dis 2020;12:7591-7.
https://doi.org/10.21037/jtd-20-964
2. Nakazono T, Yamaguchi K, Egashira R, Mizuguchi M, Irie H. Anterior mediastinal
lesions: CT and MRI features and differential diagnosis. Jpn
J Radiol 2021;39:101-17. https://doi.org/10.1007/s11604-020-01031-2
3. Pfau D, Smith DA, Beck R,
Gilani KA, Gupta A, Caimi
P. Primary Mediastinal Large B-Cell Lymphoma: A Review
for Radiologists. AJR Am J Roentgenol 2019;213:W194-W210.
https://doi.org/10.2214/AJR.19.21225
4. Lees C, Keane C, Gandhi MK, Gunawardana
J. Biology and therapy of primary mediastinal B-cell
lymphoma: current status and future directions. Br J Haematol
2019;185:25-41. https://doi.org/10.1111/bjh.15778
5. Martelli M, Ferreri A, Di Rocco A, Ansuinelli
M, Johnson PW. Primary mediastinal
large B-cell lymphoma. Crit Rev Oncol Hematol 2017;113:318-27. https://doi.org/10.1016/j.critrevonc.2017.01.009
http://dx.doi.org/10.7775/rac.v91.i1.20604
Native Tricuspid Valve Infective Endocarditis
Right-sided infective endocarditis is a rare but potentially
fatal disease. It comprises 5-10% of the total number of infective
endocarditis events. It is most frequently associated with intravenous drug
use, and occurs less frequently in patients with venous access, intravascular
devices or underlying congenital heart disease, and exceptionally in non-addict
patients or in patients without cardiac malformations. (1)
We report the case of a 58-year-old male patient with
hypertension, dyslipidemia, and a history of aortic valve replacement with
mechanical prosthesis due to aortic stenosis in 2019.
He is admitted to the general ward for febrile syndrome
under study. Cardiac physical examination reveals neither changes in heart
sounds nor signs of congestive heart failure. ECG shows sinus tachycardia and
first-degree atrioventricular block (PR interval 220
msec).
Blood screening on admission shows white blood cells
31 840/mm3 (neutrophils 96%, lymphocytes 1.7%),
C-reactive protein 58.9 mg/L (normal range 0-5), procalcitonin
4.55 ng/mL (normal range 0-0.1), total bilirubin
1.33 mg/dL, indirect bilirubin 0.83 mg/dL, and direct bilirubin 0.50 mg/dL.
During hospitalization, methicillin-susceptible Staphylococcus
aureus is detected in blood cultures. Due to
suspicion of infective endocarditis, a transesophageal
echocardiogram is performed, that detects no vegetations,
and normal mechanical valve function. On the fourth day of antibiotics,
further blood cultures detect no bacterial growth; on the tenth day, transesophageal echocardiography shows no vegetations in the heart valves, ruling out infective endocarditis.
After receiving intravenous antibiotics during 14 days, patient is discharged.
A week later, he is readmitted for fever and general
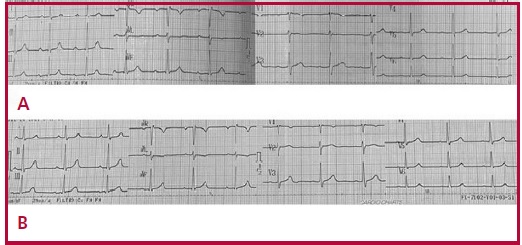
malaise; ECG reveals further PR interval prolongation (270 msec)
(Figure
1A). Blood screening
shows white blood cells 11 870/mm3 (neutrophils 87%, lymphocytes 4.7%),
C-reactive protein 25.6 mg/L, procalcitonin 0.19 ng/mL, erythrocyte sedimentation rate 32 mm/h.
Methicillin-susceptible Staphylococcus aureus is
isolated in follow-up blood cultures. Transthoracic echocardiography reveas a 0.6 cm x 0.6 cm mobile image at the tricuspid
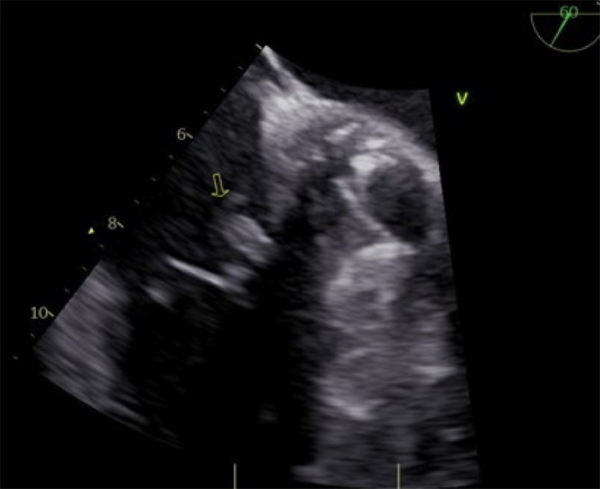
valve level. Transesophageal echocardiography (TEE)
confirms 0.9 x 0.6 cm vegetation at the level of the septal
leaflet, mild tricuspid regurgitation, and normal prosthetic valve function (Figure 2).
Fig. 1. A. ECG:
Prolonged PR interval (270 msec). B. ECG: Normal
PR interval (200 msec).
Fig.
2. TEE: View at 60°. Tricuspid septal leaflet vegetation is observed.
A conservative approach with intravenous cefazolin for 6 weeks is followed. First-degree AV block improves
by the fifth week of antibiotic treatment (Figure 1B). Follow-up transthoracic echocardiograms on the
second and sixth weeks of treatment show no evidence of tricuspid vegetation.
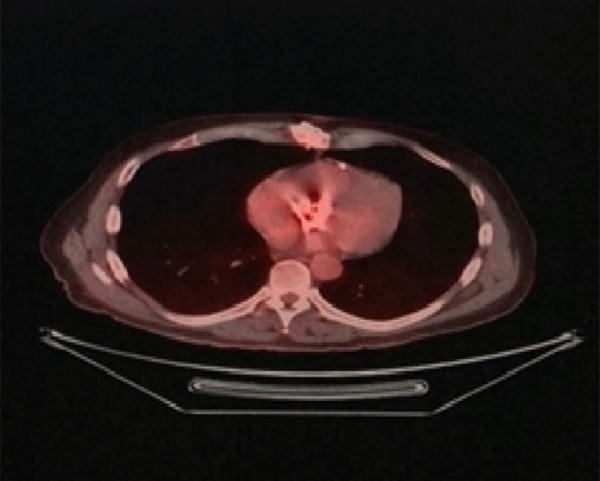
Outpatient positron-emission tomography/computed
tomography (PET/CT) for suspected prosthetic valve involvement reveals moderate
diffuse radiotracer uptake at the level of the replaced aortic valve,
suggesting the absence of an active infectious process, given the absence of a
dominant focus with increased concentration of the contrast material and SUVmax 3.5 (Figure 3).
Fig. 3. PET/CT with fluorine-18 deoxyglucose: transverse projection. Diffuse uptake in the
aortic prosthesis is observed.
Right-sided infective endocarditis is common in injecting
drug addicts and in patients with cardiac malformations; it is a potentially
serious condition, with a mortality rate between 23 and 31%. Simultaneous left
and right-sided endocarditis comprises 13% of the cases, whereas right-sided
endocarditis alone affects 10%. (1, 2)
Isolated native tricuspid valve endocarditis (NTVE)
usually occurs spontaneously, without evident history of dental or surgical
procedures; however, the skin is usually the most common portal of entry
(particularly in the case of S. Aureus). In
this clinical case, the predisposing factor could not be identified. Staphylococcus
aureus is the most commonly isolated infectious
agent (70% of cases), followed by Streptococcus and Enterococcus.
(3)
The clinical presentation invariably consists of
persistent fever associated to pulmonary events, anemia and microscopic
hematuria (tricuspid syndrome of Nandakumar and Raju). The absence of peripheral stigmata of endocarditis
or relevant murmurs in most cases is noteworthy. (4)
If fever is persistent (if it remains after a 2-week
course of antibiotics) it is usually associated with perivalvular
extension of infection, new septic emboli or superimposed nosocomial infection.
The clinical picture, positive findings on blood culture and echocardiography
are the main diagnostic tools in NTVE. (4)
The usefulness of PET/CT is significantly greater for
prosthetic valve endocarditis than for native valve infective endocarditis and
is an excellent alternative in case of negative or doubtful ultrasound scans.
Integrating PET/CT as a diagnostic tool in endocarditis allows for
reclassification of 76% of patients with prosthetic-valve infective
endocarditis from "possible" to "definite". (5)
Eighty percent of isolated NTVE patients are
successfully treated with medical therapy. However, surgery is recommended in
uncontrolled infection or right heart failure with tricuspid regurgitation refractory
to treatment. Surgical treatment repairs the valve dysfunction and eliminates
the infectious focus, thus contributing to reduce mortality associated with
heart failure. (6)
Regarding prognosis, a high success rate is achieved
with medical treatment (antibiotics), the development of heart failure is
uncommon, and only 25% of cases require valve replacement or surgery. (1) Mortality associated with isolated NTVE is lower than
that reported for endocarditis with a predisposing condition. (6)
This case suggests the need to consider isolated NTVE,
its clinical presentation, treatment, and prognosis, as well as the usefulness
of PET/CT to confirm prosthetic valve involvement.
Conflicts of interest
None declared.
(See authors’ conflicts of interest
forms on the website/ Supplementary material).
Ethical considerations
Not applicable.
Mauricio Tituana1, Diana Tituana1, Ramiro Ayala1, Gabriel Quiroga1, Andrea Trevisán1, Luis Mantilla1
Address for reprints: Department of Cardiology. Sanatorio Adventista del Plata, Entre Ríos, Argentina. Fax:
(0343) 4200-290. Email: manu2792@hotmail.com
1.
Zaldívar AÁ, Cardoso AA, Ramon RD. Endocarditis
Infecciosa Derecha. Presentación de un caso. Rev Cub Cardiol Cirug
Cardiovasc 2019;25(4).
2.
Pérez Domínguez JA, Aguilar Almaguer O, González Céspedes JC, Escandell Reyes A, Leyva Castro R, Rodríguez Peña MM.
Complicaciones sistémicas en endocarditis infecciosa de válvula tricúspide. Multimed 2019;23:543-51.
3.
Salamanca MA. Endocarditis tricuspídea secundaria a
infección asociada a catéter venoso central. Reporte de dos casos. In Anales de
la Facultad de Medicina 2020;81:330-32. UNMSM.
Facultad de Medicina.
4.
Alkan G, Emiroglu M, Sert A, Kartal A, Öc M. Endocarditis infecciosa de la válvula tricúspide
asociada con meningitis aséptica: presentación infrecuente en una niña. Arch Argent Pediatr 2020:e22-
e25.
5.
Ladrón-de-Guevara H, Canelo L, Bitar H, Ramón Soto J. Imágenes en endocarditis
infecciosa: No todo es ecocardiografía. Rev Chil infectol, 2021;38:260-70.
6.
Álvarez F, Torrez J, Galleguillos G, Saavedra J.
Endocarditis infecciosa cámaras derechas. Reporte de un caso. Rev Chil Anest
2021;50. Rev Argent Cardiol
2023;91:94-96. http://dx.doi.org/10.7775/rac.es.v91.i1.20604